Abstract
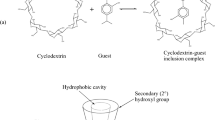
Host-guest interactions of permethylated β-cyclodextrin (PM-β-CD) with methyl mandelate enantiomers ((R/S)-MMA) were simulated using semiempirical PM3 and ONIOM (B3LYP/6-31G(d):PM3) method. The chiral recognition mechanism of (R/S)-MMA enantiomers on PM-β-CD was investigated. The binding energies for all orientations considered in this research are reported. The most stable geometry structures of the two complexes are different. The benzene ring of (R)-MMA locates horizontally approximately on the wider edge of the PM-β-CD cavity, but the aromatic ring of (S)-MMA is deeply included into the hydrophobic cavity. Furthermore, the results of NBO analysis show that the main driving forces in the inclusion process of PM-β-CD with (R/S)-MMA are hydrogen bonding interaction, dipole-dipole interaction, charge-transfer and hydrophobic interaction. The stabilization energy of the (R)-MMA/PM-β-CD complex is lower than that of the (S)-MMA/PM-β-CD complex. Moreover, the chiral carbon in MMA of (R/S)-MMA/PM-β-CD complexes are close to the C2 and C3 in the glucose unit. The chiral recognition mechanism is thus closely related to the chiral environment provided by C2 and C3 in the glucose unit and the degree of (R/S)-MMA and PM-β-CD inclusion.

The results show that the main driving forces in the inclusion process of PM-β-CD with (R/S)-MMA are hydrogen bonding interaction, dipole-dipole interaction, charge-transfer and hydrophobic interaction.


Similar content being viewed by others
References
Saenger W, Jacob J, Gessler K, Steiner T, Hoffmann D, Sanbe H, Koizumi K, Smith SM, Takaha T (1998) Structures of the common cyclodextrins and their larger analogues beyond the doughnut. Chem Rev 98:1755–1786
French AD, Johnson GP (2007) Linkage and pyranosyl ring twisting in cyclodextrins. Carbohydr Res 342:1223–1237
Rekharsky MV, Inoue Y (1998) Complexation thermodynamics of cyclodextrins. Chem Rev 98:1875–1917
Lipkowitz KB (1998) Applications of computational chemistry to the study of cyclodextrins. Chem Rev 98:1829–1873
Xing SK, Zhang C, Ai HQ, Zhao Q, Zhang Q, Sun DZ (2009) Theoretical study of the interactions of β-cyclodextrin with 2′-hydroxyl-5′-methoxyacetophone and two of its isomers. J Mol Liq 146:15–22
Beier T, Höltje HD (1998) Modified cyclodextrins as chiral selectors: molecular modelling investigations on the enantioselective binding properties of heptakis(2,3-di-O-methyl-6-O-tert-butyldimethylsilyl)-β-cyclodextrin. J Chromatogr B 708:1–20
Shahgaldian P, Pieles U (2006) Cyclodextrin derivatives as chiral supramolecular receptors for enantioselective sensing. Sensors 6:593–615
Juvancz Z, Kendrovics RB, Iványi R, Szente L (2008) The role of cyclodextrins in chiral capillary electrophoresis. Electrophoresis 29:1701–1712
Shi JH, Cheng XW, Yan W (2009) Enantiomeric separation of methylmandelate by gas chromatography and discussion the chiral recognition mechanism. Chin J Pharm Anal 29:1681–1684
Grandeury A, Petit S, Gouhier G, Agasse V, Coquerel G (2003) Enantioseparation of 1-(p-bromophenyl)ethanol by crystallization of host–guest complexes with permethylated β-cyclodextrin: crystal structures and mechanisms of chiral recognition. Tetrahedron: Asymmetry 14:2143–2152
Harata K (1998) Structural aspects of stereodifferentiation in the solid state. Chem Rev 98:1803–1827
Kano K, Kato Y, Kodera M (1996) Mechanism for chiral recognition of binaphthyl derivatives by cyclodextrins. J Chem Soc Perkin Trans 2:1211–1217
Ding HY, Chao JB, Zhang GM, Shuang SM, Pan JH (2003) Preparation and spectral investigation on inclusion complex of b-cyclodextrin with rutin. Spectrochim Acta A 59:3421–3429
Lipkowitz KB, Stoehr CM (1996) Detailed experimental and theoretical analysis of chiral discrimination: enantioselective binding of R/S methyl mandelate by β-cyclodextrin. Chirality 8:341–350
Dos Santos HF, Duarte HA, Sinisterra RD, De Melo Mattos SV, De Oliveira LFC, De Almeida WB (2000) Quantum-mechanical study of the interaction of α-cyclodextrin with methyl mercury chloride. Chem Phys Lett 319:569–575
Liu L, Guo QX (2004) Use of quantum chemical methods to study cyclodextrin chemistry. J Incl Phenom Macro 50:95–103
Yan CL, Xiu ZL, Li XH, Hao C (2007) Molecular modeling study of β-cyclodextrin complexes with (+)-catechin and (−)-epicatechin. J Mol Graph Model 26:420–428
Maseras F, Morokuma K (1995) Imomm - a new integrated ab initio plus molecular mechanics geometry optimization scheme of equilibrium structures and transition-states. J Comput Chem 16:1170–1179
Svensson M, Humbel S, Froese RDT, Matsubara T, Sieber S, Morokuma K (1996) ONIOM: a multilayered integrated MO + MM method for geometry optimizations and single point energy predictions. a test for diels-alder reactions and Pt(P(t-Bu)3)2 + H2 oxidative addition. J Phys Chem 100:19357–19363
Dapprich S, Komaromi I, Byun KS, Morokuma K (1999) A new ONIOM implementation in Gaussian98. I. The calculation of energies, gradients, vibrational frequencies and electric field derivatives. J Mol Struct THEOCHEM 461(462):1–21
Holt JS (2010) Structural characterization of the Brooker's merocyanine/β-cyclodextrin complex using NMR spectroscopy and molecular modeling. J Mol Struct 965:31–38
Yan CL, Xiu ZL, Li XH, Teng H, Hao C (2007) Theoretical study for quercetin/β-cyclodextrin complexes: quantum chemical calculations based on the PM3 and ONIOM2 method. J Incl Phenom Macro Chem 58:337–344
Tafazzoli M, Ghiasi M (2009) Structure and conformation of α-, β- and γ-cyclodextrin in solution: Theoretical approaches and experimental validation. Carbohyd Polym 78:10–15
Chen HY, Ji HB (2010) Alkaline hydrolysis of cinnamaldehyde to benzaldehyde in the presence of β-Cyclodextrin. AIChE J 56:466–476
Wang LF (2009) Halogenation effects of pheniramines on the complexation with β-cyclodextrin. J Pharm Biomed Anal 50:392–396
Nie MY, Zhou LM, Wang QH, Zhu DQ (2000) Gas chromatographic enantiomer separation of mandelates and its analogs on permethylated cyclodextrin chiral stationary phases. Chin J Anal Chem 28:1366–1370
Foresman JB, Frisch A (1996) Exploring chemistry with electronic structure methods, 2nd edn. Pittsburgh, PA, USA
Shi JH, Xiao KK, Lü YY (2009) Host-guest interactions of β-cyclodextrin with enantiomers of ethyl α-chloropropionates. Acta Phys Chim Sin 25:1273–1278
Yahia OA, Khatmi DE (2009) Theoretical study of the inclusion processes of Venlafaxine with β-cyclodextrin. J Mol Struct THEOCHEM 912:38–43
Li XH, Yin GX, Zhang XZ (2010) Natural bond orbital (NBO) population analysis of some benzyl nitrites. J Mol Struct (THEOCHEM) 957:61–65
Chocholoušová J, Špirko V, Hobza P (2004) First local minimum of the formic acid dimer exhibits simultaneously red-shifted O–H…O and improper blue-shifted C–H…O hydrogen bonds. Phys Chem Chem Phys 6:37–41
Yan L, Li XH, Xiu ZL, Hao C (2006) A quantum-mechanical study on the complexation of β-cyclodextrin with quercetin. J Mol Struct THEOCHEM 764:95–100
Saenger W (1980) Cyclodextrin inclusion compounds in research and industry. Angew Chem Int Edn Engl 19:344–362
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
Mukherjee V, Singh NP, Yadav RA (2011) Optimized geometry and vibrational spectra and NBO analysis of solid state 2,4,6-tri-fluorobenzoic acid hydrogen bonded dimer. J Mol Struct 988:24–34
Madi F, Khatmi D, Dhaoui N, Bouzitouna A, Abdaoui M, Boucekkine A (2009) Molecular model of CENS piperidine β-CD inclusion complex: DFT study. C R Chimie 12:1305–1312
Uccello-Barretta G, Balzano F, Sicoli G, Fríglola C, Aldana I, Monge A, Paolino D, Guccione S (2004) Combining NMR and molecular modelling in a drug delivery context: investigation of the multi-mode inclusion of a new NPY-5 antagonist bromobenzenesulfonamide into β-cyclodextrin. Bioorgan Med Chem 12:447–458
Van den Worm E, Beukelman CJ, van den Berg AJJ, Kroes BH, Labadie RP, van Dijk H (2001) Effects of methoxylation of apocynin and analogs on the inhibition of reactive oxygen species production by stimulated human neutrophils. Eur J Pharmacol 433:225–230
Chocholousova J, Spirko V, Hobza P (2004) First local minimum of the formic acid dimer exhibits simultaneously red-shifted O-H center dot center dot center dot O and improper blue-shifted C-H center dot center dot center dot O hydrogen bonds. Phys Chem Chem Phys 4:37–41
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, Jh., Ding, Zj. & Hu, Y. Theoretical study on chiral recognition mechanism of methyl mandelate enantiomers on permethylated β-cyclodextrin. J Mol Model 18, 803–813 (2012). https://doi.org/10.1007/s00894-011-1118-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-1118-0