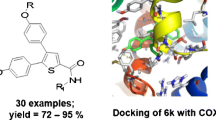
Abstract
Stilbene analogs are a new class of anti-inflammatory compounds that effectively inhibit COX-2, which is the major target in the treatment of inflammation and pain. In this study, docking simulations were conducted using AutoDock 4 software that focused on the binding of this class of compounds to COX-2 protein. Our aim was to better understand the structural and chemical features responsible for the recognition mechanism of these compounds, and to explore their binding modes of interaction at the active site by comparing them with COX-2 co-crystallized with SC-558. The docking results allowed us to provide a plausible explanation for the different binding affinities observed experimentally. These results show that important conserved residues, in particular Arg513, Phe518, Trp387, Leu352, Leu531 and Arg120, could be essential for the binding of the ligands to COX-2 protein. The quality of the docking model was estimated based on the binding energies of the studied compounds. A good correlation was obtained between experimental logAr values and the predicted binding energies of the studied compounds.

The interaction of thirty stilbene analogs to COX-2 protein was studied using the automated docking simulation. The results of this study showed that the analogs can adopt two different binding modes in the active site of the COX-2 protein and that their predicted binding energies are in agreement with their experimental logAr values.









Similar content being viewed by others
References
Smith WL, DeWitt DL, Garavito RM (2000) Annu Rev Biochem 69:145–182
Smith WL, DeWitt DL (1996) Adv Immunol 62:167–215
Marnett LJ, Rowlinson SW, Goodwin DC, Kalgutkar AS, Lanzo CA (1999) J Biol Chem 274(33):22903–22906
Picot D, Loll PJ, Garavito RM (1994) Nature 367:243–249
Luong C, Miller A, Barnett J, Chow J, Ramesha C, Browner MF (1996) Nat Struct Biol 3:927–933
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D, Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings WC (1996) Nature 384:644–648
Zhang V, O’Sullivan M, Hussain H, Roswit WT, Holtzman MJ (1996) Biochem Biophys Res Commun 227:499–506
Sirois J, Richards JS (1993) J Biol Chem 268:21931–21938
Yamagata K, Andreasson KI, Kaufmann WE, Barnes CA, Worley PF (1993) Neuron 11:371–386
Seibert K, Zhang Y, Leahy K, Hauser S, Masferrer J, Perkins W, Lee L, Isakson P (1994) Proc Natl Acad Sci USA 91:12013–12017
Turini ME, DuBois RN (2002) Ann Rev Med 53:35–57
Singh R, Kumar R, Singh DP (2009) J Med Food 12(1):208–218
Prasit P, Wang Z, Brideau C, Chan CC, Charleson S, Cromlish W, Ethier D, Evans JF, Ford–Hutchinson AW, Gauthier JY, Gordon R, Guay J, Gresser M, Kargman S, Kennedy B, Leblanc Y, Léger S, Mancini J, O’Neill GP, Ouellet M, Percival MD, Perrier H, Riendeau D, Rodger I, Tagari P, Thérien M, Vickers P, Wong E, Xu LJ, Young RN, Zambouni R (1999) Bioorg Med Chem Lett 9:1773–1778
Li S, Zheng Y (2006) Int J Mol Sci 7:220–229
Biava M, Porretta GC, Cappelli A, Vomero S, Manetti F, Botta M, Sautebin L, Rossi A, Makovec F, Anzini M (2005) J Med Chem 48:3428–3432
Bosch J, Roca T, Catena JL, Llorens O, Pérez JJ, Lagunas C, Fernàndez AG, Miquel I, Fernàndez-Serrat A, Farrerons C (2000) Bioorg Med Chem Lett 10:1745–1748
FitzGerald GA (2004) N Engl J Med 351:1709–1911
Solomon DH, Schneeweiss S, Glynn RJ, Kiyota Y, Levin R, Mogun H, Avorn J (2004) Circulation 109:2068–2073
Finckh A, Aronson MD (2005) Ann Intern Med 142:212–214
Dogné JM, Supuran CT, Pratico D (2005) J Med Chem 48:2251–2257
Chavatte P, Yous S, Marot C, Baurin N, Lesieur D (2001) J Med Chem 44:3223–3230
Desiraju GR, Gopalkrishnan B, Jetti RK, Nagaraju A, Raveendra D, Sarma JA, Sobhia ME, Thilagavathi R (2002) J Med Chem 45:4847–4857
Liu H, Huang X, Shen J, Luo X, Li M, Xiong B, Chen G, Shen J, Yang Y, Jiang H, Chen K (2002) J Med Chem 45:4816–4827
Soliva R, Almansa C, Kalko SG, Luque FJ, Orozco M (2003) J Med Chem 46:1372–1382
Garg R, Kurup A, Mekapati SB, Hansch C (2003) Chem Rev 103:703–732
Murias M, Handler N, Erker T, Pleban K, Ecker G, Saiko P, Szekeres T, Jäger W (2004) Bioorg Med Chem 12:5571–5578
Baurin N, Mozziconacci JC, Arnoult E, Chavatte P, Marot C, Morin-Allory L (2004) J Chem Inf Comput Sci 44:276–285
Chen YC, Chen KT (2007) Acta Pharmacol Sin 12:2027–2032
Dilber SP, Dobric SLJ, Juranic ZD, Markovic BD, Vladimirov SM, Juranic IO (2008) Molecules 13:603–615
Ermondi G, Caron G, Lawrence R, Longo D (2004) J Comput Aided Mol Des 18:683–696
Uddin MJ, Praveen Rao PN, Knaus EE (2005) Bioorg Med Chem 13:417–424
Uddin MJ, Rao PN, Knaus EE (2004) Bioorg Med Chem Lett 14:1953–1956
Uddin MJ, Rao PN, Knaus EE (2004) Bioorg Med Chem 12:5929–5940
Moreau A, Chen QH, Praveen Rao PN, Knaus EE (2006) Bioorg Med Chem 14:7716–7727
MDL Information Systems (2002) ISIS/Draw 2.5. MDL Information Systems, San Leandro
Cambridge Scientific Computing (2003) ChemOffice Ultra. Cambridge Scientific Computing, Cambridge
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) J Comput Chem 25(13):1605–1612
Sanner MF (1999) J Mol Graphics Mod 17:57–61
Morris Garrett M, Goodsell David S, Halliday Robert S, Huey R, Hart WE, Belew RK, Olson AJ (1998) J Comput Chem 19:1639–1662
Solis FJ, Wets RJB (1981) Math Operations Res 6:19–30
Greig GM, Francis DA, Falgueyret JP, Ouellet M, Percival MD, Roy P, Bayly C, Mancini JA, O’Neill GP (1997) Mol Pharmacol 52:829–838
Rao PN, Uddin MJ, Knaus EE (2004) J Med Chem 47:3972–3990
Zebardast T, Zarghi A, Daraie B, Hedayati M, Dadrass OG (2009) Bioorg Med Chem Lett 19:3162–3165
Kozak KR, Prusakiewicz JJ, Rowlinson SW, Schneider C, Marnett LJ (2001) J Biol Chem 276:30072–30077
Selinsky BS, Gupta K, Sharkey CT, Loll PJ (2001) Biochemistry 40:5172–5180
Rowlinson SW, Kiefer JR, Prusakiewicz JJ, Pawlitz JL, Kozak KR, Kalgutkar AS, Stallings WC, Kurumbail RG, Marnett LJ (2003) J Biol Chem 278:45763–45769
Kiefer JR, Pawlitz JL, Moreland KT, Stegeman RA, Hood WF, Gierse JK, Stevens AM, Goodwin DC, Rowlinson SW, Marnett LJ, Stallings WC, Kurumbail RG (2000) Nature 405:97–101
Teague SJ (2003) Nat Rev Drug Discov 2:527–541
Gunasekaran K, Nussinov R (2007) J Mol Biol 365:257–273
Acknowledgments
Souhila Bouaziz-Terrachet wishes to warmly thank Lehtihet Abdelhalim El Amine and Redouane Terrachet of the EMP (Ecole Militaire Polytechnique) of Algeria for their help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bouaziz-Terrachet, S., Toumi-Maouche, A., Maouche, B. et al. Modeling the binding modes of stilbene analogs to cyclooxygenase-2: a molecular docking study. J Mol Model 16, 1919–1929 (2010). https://doi.org/10.1007/s00894-010-0679-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-010-0679-7