Abstract
Three-dimensional quantitative structure-activity relationship (3D-QSAR) and molecular docking studies were carried out to explore the binding of 73 inhibitors to dipeptidyl peptidase IV (DPP-IV), and to construct highly predictive 3D-QSAR models using comparative molecular field analysis (CoMFA) and comparative molecular similarity indices analysis (CoMSIA). The negative logarithm of IC50 (pIC50) was used as the biological activity in the 3D-QSAR study. The CoMFA model was developed by steric and electrostatic field methods, and leave-one-out cross-validated partial least squares analysis yielded a cross-validated value (\( {\hbox{r}}_{{\rm{cv}}}^{\rm{2}} \)) of 0.759. Three CoMSIA models developed by different combinations of steric, electrostatic, hydrophobic and hydrogen-bond fields yielded significant \( {\hbox{r}}_{{\rm{cv}}}^{\rm{2}} \) values of 0.750, 0.708 and 0.694, respectively. The CoMFA and CoMSIA models were validated by a structurally diversified test set of 18 compounds. All of the test compounds were predicted accurately using these models. The mean and standard deviation of prediction errors were within 0.33 and 0.26 for all models. Analysis of CoMFA and CoMSIA contour maps helped identify the structural requirements of inhibitors, with implications for the design of the next generation of DPP-IV inhibitors for the treatment of type 2 diabetes.

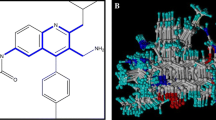
Binding conformations of docked compounds at the active site of dipeptidyl peptidase IV



Similar content being viewed by others
References
Mentlein R (1999) Regul Pept 85:9–24
Hopsu-Havu VKGG (1966) Histochemie 7:197–201
Holst JJ, Deacon CF (2004) Curr Opin Pharmacol 4:589–596
Drucker DJ (2001) Curr Pharm Des 7:1399–1412
Zander M, Madsbad S, Madsen JL, Holst JJ (2002) Lancet 359:824–830
Weber AE (2004) J Med Chem 47:4135–4141
Deacon CF, Ahren B, Holst J (2004) J Exp Opin Investig Drugs 13:1091–1102
Drucker D (2003) J Exp Opin Investig Drugs 12:87–100
Augustyns K, Van der Veken P, Senten K, Haemers A (2003) Exp Opin Ther Patents 13:499–510
Wiedeman PE, Trevillyan JM (2003) Curr Opin Investig Drugs 4:412–420
Barlocco D (2004) Curr Opin Investig Drugs 5:1094–1100
McIntyre JA, Castaner J (2004) Drugs Fut 29:887–891
Holst JJ, Deacon CF (1998) Diabetes 47:1663–1670
Augustyns K, Bal G, Thonus G, Belyaev A, Zhang XM, Bollaert W, Lambeir AM, Durinx C, Goossens F, Haemers A (1999) Curr Med Chem 6:311–327
Villhauer EB, Coppola GM, Hughes TE (2001) Annu Rep Med Chem 36:191–200
Drucker DJ (2003) Exp Opin Investig Drugs 12:87–100
Wiedeman PE, Trevillyan JM (2003) Curr Opin Investig Drugs 4:412–420
Villhauer EB, Brinkman JA, Naderi GB, Dunning BE, Mangold BL, Mone MD, Russell ME, Weldon SC, Hughes TE (2002) J Med Chem 45:2362–2365
Hughes TE, Mone MD, Russell ME, Weldon SC, Villhauer EB (1999) Biochemistry 38:11597–11603
Kanstrup AB, Christiansen LB, Lundbeck JM, Sams CK, Kristiansen M (2002) PCT Int Appl: WO2002002560
Sorbera LA, Revel L, Castaner J (2001) Drugs Fut 26:859–864
Feng J, Gwaltney SL, Stafford JA, Zang Z (2005) Jpn Kokai Tokkyo Koho: JP 2005263780
Kim D, Wang L, Beconi M, Eiermann GJ, Fisher MH, He H, Hickey GJ, Kowalchick JE, Leiting B, Lyons K, Marsilio F, McCann ME, Patel RA, Petrov A, Scapin G, Patel SB, Roy RS, Wu JK, Wyvratt MJ, Zhang BB, Zhu L, Thornberry NA, Weber AE (2005) J Med Chem 48:141–151
Mcintyre JA, Castaner J (2004) Drugs Fut 29:887–891
Barlocco D (2004) Curr Opin Invest Drugs 5:1094–1100
Xu JY, Ok HO, Gonzalez EJ, Colwell LF, Habulihaz JB, He HB, Leiting B, Lyons KA, Marsilio F, Patel RA, Wu JK, Thornberry NA, Weber AE, Parmee ER (2004) Bioorg Med Chem Lett 14:4759–4762
Brockunier LL, He JF, Colwell LF Jr, Habulihaz JB, He HB, Leiting B, Lyons KA, Marsilio F, Patel RA, Teffera Y, Wu JK, Thornberry NA, Weber AE, Parmee ER (2004) Bioorg Med Chem Lett 14:4763–4766
Brandt W, Lehmann T, Barth A, Fittkaui S (1993) J Mol Graphics 11:277–278
BrandtW LT, Thondor I, Born I, Schutkowski M, Rahfeld JU, Neubert K, Barth A (1995) Int J Peptide Prot Res 46:494–507
Zeng J, Liu GX, Tang Y, Jiang HL (2007) J Mol Model 13:993–1000
Pissurlenkar RRS, Shaikh MS, Coutinho EC (2007) J Mol Model 13:1047–1071
Ashton WT, Sisco RM, Dong H, Lyons KA, He HB, Doss GA, Leiting B, Patel RA, Wu JK, Marsilio F, Thornberry NA, Weber AE (2005) Bioorg Med Chem Lett 15:2253–2258
Kowalchick JE, Leiting B, Pryor KD, Marsilio F, Wu JK, He HB, Lyons KA, Eiermann GJ, Petrov A, Scapin G, Patel RA, Thornberry NA, Weber AE, Kim D (2007) Bioorg Med Chem Lett 17:5934–5939
Chen P, Caldwell CG, Mathvink RJ, Leiting B, Marsilio F, Patel RA, Wu JK, He HB, Lyons KA, Thornberry NA, Weber AE (2007) Bioorg Med Chem Lett 17:5853–5857
Kim D, Kowalchick JE, Edmondson SD, Mastracchio A, Xu J, Eiermann GJ, Leiting B, Wu JK, Pryor KD, Patel RA, He HB, Lyons KA, Thornberry NA, Weber AE (2007) Bioorg Med Chem Lett 17:3373–3377
Kim D, Kowalchick JE, Brockunier LL, Parmee ER, Eiermann GJ, Fisher MH, He HB, Leiting B, Lyons KA, Scapin G, Patel SB, Petrov A, Pryor KD, Roy RS, Wu JK, Zhang XP, Wyvratt MJ, Zhang BB, Zhu L, Thornberry NA, Weber AE (2008) J Med Chem 51:589–602
Sybyl Version 8.1 (2009) Tripos, St. Louis, MO
Clark M, Cramer RDI, Opdenbosch NV (1989) J Comput Chem 10:982–1012
Gasteiger J, Marsili M (1980) Tetrahedron 36:3219–3228
Weiner SJ, Kollman PA, Case DA, Singh C, Ghio G, Alagona S, Profeta P, Weiner P (1984) J Am Chem Soc 106:765–784
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) J Comput Chem 19:1639–1662
Auffinger P, Hays FA, Westhof E, Ho PS (2004) Proc Natl Acad Sci USA 101:16789–16794
Howard EI, Sanishvili R, Cachau RE, Mitschler A, Chevrier B, Barth P, Lamour V, Zandt MV, Sibley E, Bon C, Moras D, Schneider TR, Joachimiak A, Podjarny A (2004) Proteins 55:792–804
Cramer RD, Patterson IIIDE, Bunce JD (1988) J Am Chem Soc 110:5959–5967
Klebe G, Abraham U, Mietzner T (1994) J Med Chem 37:4130–4146
Kuhn B, Hennig M, Mattei P (2007) Curr Top Med Chem 7:609–619
Xu M, Zhang AQ, Han SK, Wang LS (2002) Chemosphere 48:707–715
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, YK. Molecular docking and 3D-QSAR studies on β-phenylalanine derivatives as dipeptidyl peptidase IV inhibitors. J Mol Model 16, 1239–1249 (2010). https://doi.org/10.1007/s00894-009-0637-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-009-0637-4