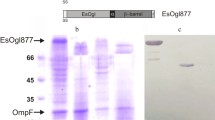
Abstract
Cell surface display is a popular approach for the construction of whole-cell biocatalysts, live vaccines, and screening of combinatorial libraries. To develop a novel surface display system for the popular scaffold protein 10th human fibronectin type III domain (10Fn3) in Escherichia coli cells, we have used an α-helical linker and a C-terminal translocator domain from previously characterized autotransporter from Psychrobacter cryohalolentis K5T. The level of 10Fn3 passenger exposure at the cell surface provided by the hybrid autotransporter Fn877 and its C-terminal variants was low. To improve it, the fusion proteins containing 10Fn3 and the native autotransporter passenger Est877 or the cold-active esterase EstPc in different orientations were constructed and expressed as passenger domains. Using the whole-cell ELISA and activity assays, we have demonstrated that N-terminal position of EstPc in the passenger significantly improves the efficiency of the surface display of 10Fn3 in E. coli cells.



Similar content being viewed by others
References
a Nijeholt JAL, Driessen AJ (2012) The bacterial Sec-translocase: structure and mechanism. Philos Trans R Soc B 367:1016–1028
Bakermans C, Ayala-del-Rio HL, Ponder MA, Vishnivetskaya T, Gilichinsky D, Thomashow MF, Tiedje JM (2006) Psychrobacter cryohalolentis sp. nov. and Psychrobacter arcticus sp. nov., isolated from Siberian permafrost. Int J Syst Evol Microbiol 56:1285–1291. https://doi.org/10.1099/ijs.0.64043-0
Barnard TJ, Dautin N, Lukacik P, Bernstein HD, Buchanan SK (2007) Autotransporter structure reveals intra-barrel cleavage followed by conformational changes. Nat Struct Mol Biol 14:1214–1220
Becker S, Theile S, Heppeler N, Michalczyk A, Wentzel A, Wilhelm S, Jaeger KE, Kolmar H (2005) A generic system for the Escherichia coli cell-surface display of lipolytic enzymes. FEBS Lett 579:1177–1182. https://doi.org/10.1016/j.febslet.2004.12.087
Binder U, Matschiner G, Theobald I, Skerra A (2010) High-throughput sorting of an Anticalin library via EspP-mediated functional display on the Escherichia coli cell surface. J Mol Biol 400:783–802. https://doi.org/10.1016/j.jmb.2010.05.049
Braselmann E, Clark PL (2012) Autotransporters: the cellular environment reshapes a folding mechanism to promote protein transport. J Phys Chem Lett 3:1063–1071. https://doi.org/10.1021/jz201654k
Cavicchioli R, Siddiqui KS, Andrews D, Sowers KR (2002) Low-temperature extremophiles and their applications. Curr Opin Biotechnol 13:253–261
Celik N, Webb CT, Leyton DL, Holt KE, Heinz E, Gorrell R, Kwok T, Naderer T, Strugnell RA, Speed TP, Teasdale RD (2012) A bioinformatic strategy for the detection, classification and analysis of bacterial autotransporters. PLoS One 7:e43245
Chen TF, de Picciotto S, Hackel BJ, Wittrup KD (2013) Engineering fibronectin-based binding proteins by yeast surface display. Methods Enzymol 523:303–326. https://doi.org/10.1016/B978-0-12-394292-0.00014-X
Costa TRD, Felisberto-Rodrigues C, Meir A, Prevost MS, Redzej A, Trokter M, Waksman G (2015) Secretion systems in gram-negative bacteria: structural and mechanistic insights. Nat Rev Microbiol 13:343–359. https://doi.org/10.1038/nrmicro3456
D’Amico S, Claverie P, Collins T, Georlette D, Gratia E, Hoyoux A, Meuwis MA, Feller G, Gerday C (2002) Molecular basis of cold adaptation. Philos Trans R Soc B 357:917–925
Deyev S, Lebedenko E, Petrovskaya L, Dolgikh D, Gabibov A, Kirpichnikov M (2015) Man-made antibodies and immunoconjugates with desired properties: function optimization using structural engineering. Russ Chem Rev 84:1–26
Drobnak I, Braselmann E, Clark PL (2015) Multiple driving forces required for efficient secretion of autotransporter virulence proteins. J Biol Chem 290:10104–10116. https://doi.org/10.1074/jbc.M114.629170
Elleuche S, Schröder C, Sahm K, Antranikian G (2014) Extremozymes—biocatalysts with unique properties from extremophilic microorganisms. Curr Opin Biotechnol 29:116–123
Feller G (2003) Molecular adaptations to cold in psychrophilic enzymes. Cell Mol Life Sci 60:648–662. https://doi.org/10.1007/s00018-003-2155-3
Gawarzewski I, DiMaio F, Winterer E, Tschapek B, Smits SH, Jose J, Schmitt L (2014) Crystal structure of the transport unit of the autotransporter adhesin involved in diffuse adherence from Escherichia coli. J Struct Biol 187:20–29. https://doi.org/10.1016/j.jsb.2014.05.003
Georlette D, Blaise V, Collins T, D’Amico S, Gratia E, Hoyoux A, Marx JC, Sonan G, Feller G, Gerday C (2004) Some like it cold: biocatalysis at low temperatures. FEMS Microbiol Rev 28:25–42
Grijpstra J, Arenas J, Rutten L, Tommassen J (2013) Autotransporter secretion: varying on a theme. Res Microbiol 164:562–582. https://doi.org/10.1016/j.resmic.2013.03.010
Jong WS, Hagen-Jongman T, Corinne M, Den Blaauwen T, Jan Slotboom D, Tame JR, Wickström D, De Gier JW, Otto BR, Luirink J (2007) Limited tolerance towards folded elements during secretion of the autotransporter Hbp. Mol Microbiol 63:1524–1536
Jose J, Maas RM, Teese MG (2012) Autodisplay of enzymes–molecular basis and perspectives. J Biotechnol 161:92–103. https://doi.org/10.1016/j.jbiotec.2012.04.001
Junker M, Besingi RN, Clark PL (2009) Vectorial transport and folding of an autotransporter virulence protein during outer membrane secretion. Mol Microbiol 71:1323–1332. https://doi.org/10.1111/j.1365-2958.2009.06607.x
Kjaergaard K, Hasman H, Schembri MA, Klemm P (2002) Antigen 43-mediated autotransporter display, a versatile bacterial cell surface presentation system. J Bacteriol 184:4197–4204. https://doi.org/10.1128/jb.184.15.4197-4204.2002
Koide A, Bailey CW, Huang X, Koide S (1998) The fibronectin type III domain as a scaffold for novel binding proteins. J Mol Biol 284:1141–1151. https://doi.org/10.1006/jmbi.1998.2238
Koide S, Koide A, Lipovsek D (2012) Target-binding proteins based on the 10th human fibronectin type III domain (10Fn3). Methods Enzymol 503:135–156. https://doi.org/10.1016/b978-0-12-396962-0.00006-9
Leo JC, Grin I, Linke D (2012) Type V secretion: mechanism(s) of autotransport through the bacterial outer membrane. Philos Trans R Soc B 367:1088–1101. https://doi.org/10.1098/rstb.2011.0208
Leyton DL, Sevastsyanovich YR, Browning DF, Rossiter AE, Wells TJ, Fitzpatrick RE, Overduin M, Cunningham AF, Henderson IR (2011) Size and conformation limits to secretion of disulfide-bonded loops in autotransporter proteins. J Biol Chem 286:42283–42291
Leyton DL, Rossiter AE, Henderson IR (2012) From self sufficiency to dependence: mechanisms and factors important for autotransporter biogenesis. Nat Rev Microbiol 10:213–225. https://doi.org/10.1038/nrmicro2733
Lipovsek D (2011) Adnectins: engineered target-binding protein therapeutics. Protein Eng Des Sel 24:3–9. https://doi.org/10.1093/protein/gzq097
Littlechild JA (2015) Enzymes from extreme environments and their industrial applications. Front Bioeng Biotechnol 3:161. https://doi.org/10.3389/fbioe.2015.00161
Lofblom J (2011) Bacterial display in combinatorial protein engineering. Biotechnol J 6:1115–1129. https://doi.org/10.1002/biot.201100129
Lum M, Morona R (2012) IcsA autotransporter passenger promotes increased fusion protein expression on the cell surface. Microb Cell Factories 11:20. https://doi.org/10.1186/1475-2859-11-20
Maayer PD, Anderson D, Cary C, Cowan DA (2014) Some like it cold: understanding the survival strategies of psychrophiles. EMBO Rep 15:508–517. https://doi.org/10.1002/embr.201338170
Margesin R, Feller G, Gerday C, Russell NJ (2002) Cold-adapted microorganisms: adaptation strategies and biotechnological potential. Encycl Environ Microbiol. https://doi.org/10.1002/0471263397.env150
Miroux B, Walker JE (1996) Over-production of proteins in Escherichia coli: mutant hosts that allow synthesis of some membrane proteins and globular proteins at high levels. J Mol Biol 260:289–298
Nicolay T, Vanderleyden J, Spaepen S (2015) Autotransporter-based cell surface display in gram-negative bacteria. Crit Rev Microbiol 41:109–123. https://doi.org/10.3109/1040841X.2013.804032
Noinaj N, Kuszak AJ, Gumbart JC, Lukacik P, Chang H, Easley NC, Lithgow T, Buchanan SK (2013) Structural insight into the biogenesis of β-barrel membrane proteins. Nature 501:385–390
Novototskaya-Vlasova K, Petrovskaya L, Yakimov S, Gilichinsky D (2012) Cloning, purification, and characterization of a cold adapted esterase produced by Psychrobacter cryohalolentis K5T from Siberian cryopeg. FEMS Microbiol Ecol 82:367–375. https://doi.org/10.1111/j.1574-6941.2012.01385.x
Novototskaya-Vlasova K, Petrovskaya L, Kryukova E, Rivkina E, Dolgikh D, Kirpichnikov M (2013a) Expression and chaperone-assisted refolding of a new cold-active lipase from Psychrobacter cryohalolentis K5T. Protein Expr Purif 91:96–103
Novototskaya-Vlasova K, Petrovskaya L, Rivkina E, Dolgikh D, Kirpichnikov M (2013b) Characterization of a cold-active lipase from Psychrobacter cryohalolentis K5T and its deletion mutants. Biochemistry (Moscow) 78:385–394
Oomen CJ, van Ulsen P, Van Gelder P, Feijen M, Tommassen J, Gros P (2004) Structure of the translocator domain of a bacterial autotransporter. EMBO J 23:1257–1266
Pakharukova N, Roy S, Tuittila M, Rahman MM, Paavilainen S, Ingars AK, Skaldin M, Lamminmäki U, Härd T, Teneberg S, Zavialov AV (2016) Structural basis for Myf and Psa fimbriae-mediated tropism of pathogenic strains of Yersinia for host tissues. Mol Microbiol 102:593–610. https://doi.org/10.1111/mmi.13481
Park M, Bong JH, Yoo G, Jose J, Kang MJ, Pyun JC (2013) Flow cytometric immunoassay using E. coli with autodisplayed Z-domains. Enzym Microb Technol 53:181–188. https://doi.org/10.1016/j.enzmictec.2013.03.018
Peterson JH, Tian P, Ieva R, Dautin N, Bernstein HD (2010) Secretion of a bacterial virulence factor is driven by the folding of a C-terminal segment. Proc Natl Acad Sci USA 107:17739–17744
Petrovskaya LE, Shingarova LN, Kryukova EA, Boldyreva EF, Yakimov SA, Guryanova SV, Novoseletsky VN, Dolgikh DA, Kirpichnikov MP (2012) Construction of TNF-binding proteins by grafting hypervariable regions of F10 antibody on human fibronectin domain scaffold. Biochemistry (Moscow) 77:62–70. https://doi.org/10.1134/s0006297912010075
Petrovskaya L, Novototskaya-Vlasova K, Kryukova E, Rivkina E, Dolgikh D, Kirpichnikov M (2015) Cell surface display of cold-active esterase EstPc with the use of a new autotransporter from Psychrobacter cryohalolentis K5T. Extremophiles 19:161–170
Plaxco KW, Spitzfaden C, Campbell ID, Dobson CM (1997) A comparison of the folding kinetics and thermodynamics of two homologous fibronectin type III modules. J Mol Biol 270:763–770. https://doi.org/10.1006/jmbi.1997.1148
Renn JP, Junker M, Besingi RN, Braselmann E, Clark PL (2012) ATP-independent control of autotransporter virulence protein transport via the folding properties of the secreted protein. Chem Biol 19:287–296
Roussel-Jazédé V, Gelder PV, Sijbrandi R, Rutten L, Otto BR, Luirink J, Gros P, Tommassen J, Ulsen PV (2011) Channel properties of the translocator domain of the autotransporter Hbp of Escherichia coli. Mol Membr Biol 28:158–170
Rutherford N, Mourez M (2006) Surface display of proteins by gram-negative bacterial autotransporters. Microb Cell Factories 5:22. https://doi.org/10.1186/1475-2859-5-22
Rutherford N, Charbonneau M-È, Berthiaume F, Betton J-M, Mourez M (2006) The periplasmic folding of a cysteineless autotransporter passenger domain interferes with its outer membrane translocation. J Bacteriol 188:4111–4116
Siddiqui KS, Cavicchioli R (2006) Cold-adapted enzymes. Annu Rev Biochem 75:403–433
Ståhl S, Kronqvist N, Jonsson A, Löfblom J (2013) Affinity proteins and their generation. J Chem Technol Biotechnol 88:25–38. https://doi.org/10.1002/jctb.3929
Struyvé M, Moons M, Tommassen J (1991) Carboxy-terminal phenylalanine is essential for the correct assembly of a bacterial outer membrane protein. J Mol Biol 218:141–148
Thanassi DG, Stathopoulos C, Karkal A, Li H (2005) Protein secretion in the absence of ATP: the autotransporter, two-partner secretion and chaperone/usher pathways of gram-negative bacteria. Mol Membr Biol 22:63–72
Trainor K, Gingras Z, Shillingford C, Malakian H, Gosselin M, Lipovšek D, Meiering EM (2016) Ensemble modeling and intracellular aggregation of an engineered immunoglobulin-like domain. J Mol Biol 428:1365–1374. https://doi.org/10.1016/j.jmb.2016.02.016
van Bloois E, Winter RT, Kolmar H, Fraaije MW (2011) Decorating microbes: surface display of proteins on Escherichia coli. Trends Biotechnol 29:79–86. https://doi.org/10.1016/j.tibtech.2010.11.003
van Ulsen P, ur Rahman S, Jong WS, Daleke-Schermerhorn MH, Luirink J (2014) Type V secretion: from biogenesis to biotechnology. Biochim Biophys Acta (BBA) Mol. Cell Res 1843:1592–1611
Veiga E, de Lorenzo V, Fernández LA (2003) Neutralization of enteric coronaviruses with Escherichia coli cells expressing single-chain Fv-autotransporter fusions. J Virol 77:13396–13398
Veiga E, de Lorenzo V, Fernandez LA (2004) Structural tolerance of bacterial autotransporters for folded passenger protein domains. Mol Microbiol 52:1069–1080. https://doi.org/10.1111/j.1365-2958.2004.04014.x
Wang L-X, Mellon M, Bowder D, Quinn M, Shea D, Wood C, Xiang S-H (2015) Escherichia coli surface display of single-chain antibody VRC01 against HIV-1 infection. Virology 475:179–186
Webb CT, Lithgow T (2015) Identification of BamC on the surface of E. coli. In: The BAM complex: methods and protocols, pp 215–225
Wilhelm S, Tommassen J, Jaeger K-E (1999) A novel lipolytic enzyme located in the outer membrane of Pseudomonas aeruginosa. J Bacteriol 181:6977–6986
Wilhelm S, Kolmar H, Rosenau F (2010) Bacterial secretion systems for use in biotechnology: autotransporter-based ultra-high throughput cell-surface display and screening of large protein libraries. In: Handbook of hydrocarbon and lipid microbiology, pp 4587–4600 https://doi.org/10.1007/978-3-540-77587-4_361
Acknowledgements
The work was supported by Russian Academy of Sciences program “Molecular and Cellular Biology”, RF President Grant for Scientific Schools Grant SS-8384.2016.4 and RFBR Grants nos. 16-04-00717a, 15-04-07772a.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by H. Atomi.
Rights and permissions
About this article
Cite this article
Petrovskaya, L.E., Zlobinov, A.V., Shingarova, L.N. et al. Fusion with the cold-active esterase facilitates autotransporter-based surface display of the 10th human fibronectin domain in Escherichia coli . Extremophiles 22, 141–150 (2018). https://doi.org/10.1007/s00792-017-0990-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-017-0990-7