Abstract
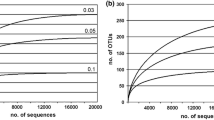
Surveys of Champagne Pool, one of New Zealand’s largest terrestrial hot springs and rich in arsenic ions and compounds, have been restricted to geological and geochemical descriptions, and a few microbiological studies applying culture-independent methods. In the current investigation, a combination of culture and culture-independent approaches were chosen to determine microbial density and diversity in Champagne Pool. Recovered total DNA and adenosine 5′-triphosphate (ATP) content of spring water revealed relatively low values compared to other geothermal springs within New Zealand and are in good agreement with low cell numbers of 5.6 ± 0.5 × 106 cells/ml obtained for Champagne Pool water samples by 4′,6-diamidino-2-phenylindole (DAPI) staining. Denaturing Gradient Gel Electrophoresis (DGGE) and 16S rRNA (small-subunit ribosomal nucleic acid) gene clone library analyses of environmental DNA indicated the abundance of Sulfurihydrogenibium, Sulfolobus, and Thermofilum-like populations in Champagne Pool. From these results, media were selected to target the enrichment of hydrogen-oxidizing and sulfur-dependent microorganisms. Three isolates were successfully obtained having 16S rRNA gene sequences with similarities of ∼98% to Thermoanaerobacter tengcongensis, 94% to Sulfurihydrogenibium azorense, and 99% to Thermococcus waiotapuensis, respectively.





Similar content being viewed by others
References
Aguiar P, Beveridge TJ, Reysenbach AL (2004) Sulfurihydrogenibium azorense, sp. nov., a thermophilic hydrogen-oxidizing microaerophile from terrestrial hot springs in the Azores. Int J Syst Evol Microbiol 54:33–39
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Brock TD, Brock KM, Belly RT, Weiss RL (1972) Sulfolobus–New genus of sulfur-oxidizing bacteria living at low pH and high-temperature. Arch Mikrobiol 84:54–68
Dempster EL, Pryor KV, Francis D, Young JE, Rogers HJ (1999) Rapid DNA extraction from ferns for PCR-based analyses. Biotechniques 27:66–68
Ellis DG, Bizzoco RLW, Maezato Y, Baggett JN, Kelley ST (2005) Microscopic examination of acidic hot springs of Waiotapu, North Island, New Zealand. NZ J Mar Freshw Res 39:1001–1011
Felsenstein J (1993) PHYLIP (Phylogeny Inference Package) version 3.5c. Distributed by the author. Department of Genetics, University of Washington, Seattle
Ferris MJ, Muyzer G, Ward DM (1996) Denaturing gradient gel electrophoresis profiles of 16S rRNA-defined populations inhabiting a hot spring microbial mat community. Appl Environ Microbiol 62:340–346
Giggenbach WF, Sheppard DS, Robinson BW, Stewart MK, Lyon GL (1994) Geochemical structure and position of the Waiotapu geothermal field, New Zealand. Geothermics 23:599–644
Gonzalez JM, Sheckells D, Viebahn M, Krupatkina D, Borges KM, Robb FT (1999) Thermococcus waiotapuensis sp. nov., an extremely thermophilic archaeon isolated from a freshwater hot spring. Arch Microbiol 172:95–101
Hedenquist JW, Henley RW (1985) Hydrothermal eruptions in the Waiotapu geothermal system, New Zealand: their origin, associated breccias and relation to precious metal mineralization. Econ Geol 80:1640–1668
Hedenquist JW (1986a) Waiotapu geothermal field. In: Henley RW, Hedenquist JW, Roberts PJ (eds) Guide to the active epithermal (geothermal) systems and precious metal deposits of New Zealand, Monograph Series on Mineral Deposits. Gebrüder Borntraeger, Berlin-Stuttgart, pp 65–79
Hedenquist JW (1986b) Geothermal systems in the Taupo Volcanic Zone: their characteristics and relation to volcanism and mineralisation. R Soc NZ Bull 23:134–168
Hirner AV, Feldmann J, Krupp E, Grumping R, Goguel R, Cullen WR (1998) Metal(loid)organic compounds in geothermal gases and waters. Org Geochem 29:1765–1778
Huber H, Hohn MJ, Rachel R, Fuchs T, Wimmer VC, Stetter KO (2002) A new phylum of Archaea represented by a nanosized hyperthermophilic symbiont. Nature 417:63–67
Johnson JL (1994) Similarity analysis of rRNAs. In: Gerhardt P, Murray RGE, Wood WA, Krieg NR (eds) Methods for general and molecular bacteriology. American Society for Microbiology, Washington, DC, pp 683–700
Jones B, Renaut RW, Rosen MR (1999) Actively growing siliceous oncoids in the Waiotapu geothermal area, North Island, New Zealand. J Geol Soc 156:89–103
Jones B, Renaut RW, Rosen MR (2001) Biogenicity of gold- and silver-bearing siliceous sinters forming in hot (75 degrees C) anaerobic spring-waters of Champagne Pool, Waiotapu, North Island, New Zealand. J Geol Soc 158:895–911
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Willy, New York, NY, pp 115–175
Lloyd EF (1959) The hot springs and hydrothermal eruptions of Waiotapu. NZ J Geol Geophys 2:141–176
Ludwig W, Strunk O, Westram R, et al (2004) ARB: a software environment for sequence data. Nucleic Acids Res 32:1363–1371
Marmur J (1961) A procedure for the isolation of deoxyribonucleic acid from microorganisms. J Mol Biol 3:208–218
Mountain BW, Benning LG, Boerema JA (2003) Experimental studies on New Zealand hot spring sinters: rates of growth and textural development. Can J Earth Sci 40:1643–1667
Muyzer G, Dewaal EC, Uitterlinden AG (1993) Profiling of complex microbial-populations by denaturing gradient gel-electrophoresis analysis of polymerase chain reaction-amplified genes-coding for 16S ribosomal-RNA. Appl Environ Microbiol 59:695–700
Niederberger TD (2005) The microbial ecology and colonisation of surfaces in selected hot springs–a molecular- and culture-based approach. Ph.D. thesis, Thermophile Research Unit, University of Waikato, Hamilton, New Zealand
Niederberger TD, Gotz DK, McDonald IR, Ronimus RS, Morgan HW (2006) Ignisphaera aggregans gen. nov., sp. nov., a novel hyperthermorphilic crenarchaeote isolated from hot springs in Rotorua and Tokaanu, New Zealand. Int J Syst Evol Microbiol 56:965–971
Phoenix VR, Renaut RW, Jones B, Ferris FG (2005) Bacterial S-layer preservation and rare arsenic-antimony-sulphide bioimmobilization in siliceous sediments from Champagne Pool hot spring, Waiotapu, New Zealand. J Geol Soc 162:323–331
Pope JG, Brown KL, McConchie DM (2005) Gold concentrations in springs at Waiotapu, New Zealand: implications for precious metal deposition in geothermal systems. Econ Geol 100:677–687
Porter KG, Feig YS (1980) The use of DAPI for identifying and counting aquatic microflora. Limnol Oceanogr 25:943–948
Sierra-Alvarez R, Cortinas I, Yenal U, Field JA (2004) Methanogenic inhibition by arsenic compounds. Appl Environ Microbiol 70:5688–5691
Stetter KO (1986) Diversity of extremely thermophilic archaebacteria. In: Brock TD (ed) Thermophiles: general, molecular and applied microbiology. Wiley, New York, pp 39–74
Stolz JF, Oremland RS (1999) Bacterial respiration of arsenic and selenium. FEMS Microbiol Rev 23:615–627
Wetzel RG, Likens GE (2000) Limnological analysis. Springer, New York
Xue YF, Xu Y, Liu Y, Ma YH, Zhou PJ (2001) Thermoanaerobacter tengcongensis sp. nov., a novel anaerobic, saccharolytic, thermophilic bacterium isolated from a hot spring in Tengcong, China. Int J Syst Evol Microbiol 51:1335–1341
Acknowledgments
Research funding was provided by the New Zealand Foundation of Research, Science and Technology (Contract Number C05X0303: Extremophilic Microorganisms for Metal Sequestration from Aqueous Solutions). The authors are grateful to Richard Fulton for permission to collect samples from Champagne Pool and Helen M. Turner who helped to take the SEM micrographs used in this paper. A. Hetzer is indebted to his beloved wife for her encouragement and support. The editor and the two reviewers offered comments and suggestions that greatly improved this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D. A. Cowan.
Rights and permissions
About this article
Cite this article
Hetzer, A., Morgan, H.W., McDonald, I.R. et al. Microbial life in Champagne Pool, a geothermal spring in Waiotapu, New Zealand. Extremophiles 11, 605–614 (2007). https://doi.org/10.1007/s00792-007-0073-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-007-0073-2