Abstract.
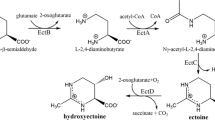
The role of K+ in osmoregulation of the halophilic bacterium Halomonas elongata was investigated. At lower salinities (0.51 M NaCl), K+ was the predominant cytoplasmic solute (1.25 µmol mg protein–1). At higher salinities (1.03 M NaCl) ectoine became the main cytoplasmic solute (1.57 µmol mg protein–1), while the K+ content remained unchanged. In response to osmotic upshock, cells of H. elongata simultaneously accumulated ectoine and K+ glutamate. The ectoine and K+ glutamate levels in osmotically stressed cells exceeded the level of cells adapted to high salinities. The increase in K+ glutamate was long lasting (>120 min) and not transient, as described for non-halophiles. Regulation of the synthesis of ectoine and glutamate was proven to occur mainly at the level of enzyme activity. Limitation of K+ inhibited the growth of salt-adapted H. elongata cells, especially at high salinities, and caused a decrease of the intracellular organic solute content, inhibition of respiration, and an abolition of the cell's ability to respond to osmotic stress. The saturation constant K S for K+ was estimated to be 105 µM at a salinity of 0.51 M NaCl, indicating that an uptake system of medium affinity is responsible for K+ accumulation in H. elongata.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Kraegeloh, A., Kunte, H. Novel insights into the role of potassium for osmoregulation in Halomonas elongata . Extremophiles 6, 453–462 (2002). https://doi.org/10.1007/s00792-002-0277-4
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00792-002-0277-4