Abstract
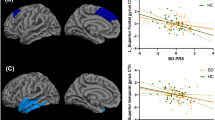
Children of parents diagnosed with bipolar disorder (BD), termed high-risk offspring (HRO), are at greater risk of developing psychiatric disorders compared to healthy children of healthy parents (HCO). Gray matter volume (GMV) abnormalities have been observed in HRO, however, these reports are inconsistent. We posit that this variability may be attributed to differences in methodology among offspring studies; in particular, the presence of psychiatric symptoms in HRO. Here, we directly compared GMVs between symptomatic and asymptomatic HRO, and HCO. High-resolution T1-weighted MR images were collected from 31 HRO (18 symptomatic and 13 asymptomatic) and 20 age- and sex-matched HCO. HRO had at least one parent diagnosed with BD. Symptomatic HRO were defined as having a psychiatric diagnosis other than BD, while asymptomatic HRO were required to be free of any psychiatric diagnosis. Scans were processed using voxel-based morphometry methods and between group analyses were performed in SPM. Compared to HCO, the HRO group showed decreased GMV in the right inferior orbitofrontal, right middle frontal, and bilateral superior and middle temporal regions. Both symptomatic and asymptomatic HRO groups showed decreased GMV in these regions separately when compared to HCO. When comparing symptomatic and asymptomatic HRO, GMVs were comparable in all regions except the lateral occipital cortex. Our study compared symptomatic and asymptomatic HRO directly. In doing so, we provided further support for the presence of discrete GMV deficits in HRO, and confirmed that these deficits are present irrespective of the presence of symptoms in HRO.

Similar content being viewed by others
References
McGuffin P, Rijsdijk F, Andrew M (2003) The heritability of bipolar affective disorder and the genetic relationship to unipolar depression. Arch Gen Psychiatry 60(5):497–502
Rasic D, Hajek T, Alda M, Uher R (2014) Risk of mental illness in offspring of parents with schizophrenia, bipolar disorder, and major depressive disorder: a meta-analysis of family high-risk studies. Schizophr Bull 40(1):28–38
Birmaher B, Axelson D, Monk K, Kalas C, Goldstein B, Hickey MB, Obreja M, Ehmann M, Iyengar S, Shamseddeen W (2009) Lifetime psychiatric disorders in school-aged offspring of parents with bipolar disorder: the Pittsburgh Bipolar Offspring study. Arch Gen Psychiatry 66(3):287–296
DelBello MP, Geller B (2001) Review of studies of child and adolescent offspring of bipolar parents. Bipolar Disord 3(6):325–334. doi:10.1034/j.1399-5618.2001.30607.x
Wray NR, Gottesman II (2012) Using summary data from the danish national registers to estimate heritabilities for schizophrenia, bipolar disorder, and major depressive disorder. Front Genet 3(118):1–12. doi:10.3389/fgene.2012.00118
Craddock N, O’Donovan M, Owen M (2005) The genetics of schizophrenia and bipolar disorder: dissecting psychosis. J Med Genet 42(3):193–204
Kieseppa T, Partonen T, Haukka J, Kaprio J, Lonnqvist J (2004) High concordance of bipolar I disorder in a nationwide sample of twins. Am J Psychiatry 161(10):1814–1821. doi:10.1176/appi.ajp.161.10.1814
Dean K, Stevens H, Mortensen PB, Murray RM, Walsh E, Pedersen CB (2010) Full spectrum of psychiatric outcomes among offspring with parental history of mental disorder. Arch Gen Psychiatry 67(8):822–829. doi:10.1001/archgenpsychiatry.2010.86
Hillegers MHJ, Reichart CG, Wals M, Verhulst FC, Ormel J, Nolen WA (2005) Five-year prospective outcome of psychopathology in the adolescent offspring of bipolar parents. Bipolar Disord 7(4):344–350. doi:10.1111/j.1399-5618.2005.00215.x
Chang KD, Howe M, Gallelli KIM, Miklowitz D (2006) Prevention of pediatric bipolar disorder. Ann N Y Acad Sci 1094(1):235–247. doi:10.1196/annals.1376.026
Johnson JG, Cohen P, Brook JS (2000) Associations between bipolar disorder and other psychiatric disorders during adolescence and early adulthood: a community-based longitudinal investigation. Am J Psychiatry 157(10):1679–1681
Brückl T, Wittchen H, Höfler M, Pfister H, Schneider S, Lieb R (2007) Childhood separation anxiety and the risk of subsequent psychopathology: results from a community study. Psychother Psychosom 76(1):47–56
Strakowski SM, Adler CM, Almeida J, Altshuler LL, Blumberg HP, Chang KD, DelBello MP, Frangou S, McIntosh A, Phillips ML, Sussman JE, Townsend JD (2012) The functional neuroanatomy of bipolar disorder: a consensus model. Bipolar Disord 14(4):313–325. doi:10.1111/j.1399-5618.2012.01022.x
Strakowski SM, Delbello MP, Adler CM (2005) The functional neuroanatomy of bipolar disorder: a review of neuroimaging findings. Mol Psychiatry 10(1):105–116. doi:10.1038/sj.mp.4001585
Phillips ML, Ladouceur CD, Drevets WC (2008) A neural model of voluntary and automatic emotion regulation: implications for understanding the pathophysiology and neurodevelopment of bipolar disorder. Mol Psychiatry 13(9):833–857
Pavuluri MN, O‘Connor MM, Harral E, Sweeney JA (2007) Affective neural circuitry during facial emotion processing in pediatric bipolar disorder. Biol Psychiatry 62(2):158–167
Blond BN, Fredericks CA, Blumberg HP (2012) Functional neuroanatomy of bipolar disorder: structure, function, and connectivity in an amygdala-anterior paralimbic neural system. Bipolar Disord 14(4):340–355. doi:10.1111/j.1399-5618.2012.01015.x
Townsend JD, Altshuler LL (2012) Emotion processing and regulation in bipolar disorder: a review. Bipolar Disord 14(4):326–339. doi:10.1111/j.1399-5618.2012.01021.x
Chen C-H, Suckling J, Lennox BR, Ooi C, Bullmore ET (2011) A quantitative meta-analysis of fMRI studies in bipolar disorder. Bipolar Disord 13(1):1–15. doi:10.1111/j.1399-5618.2011.00893.x
Hajek T, Gunde E, Slaney C, Propper L, MacQueen G, Duffy A, Alda M (2009) Amygdala and hippocampal volumes in relatives of patients with bipolar disorder: a high-risk study. Can J Psychiatry—Revue Canadienne de Psychiatrie 54(11):726–733
Singh MK, DelBello MP, Adler CM, Stanford KE, Strakowski SM (2008) Neuroanatomical characterization of child offspring of bipolar parents. J Am Acad Child Adolesc Psychiatry 47(5):526–531
Hajek T, Kozeny J, Kopecek M, Alda M, Hoschl C (2008) Reduced subgenual cingulate volumes in mood disorders: a meta-analysis. J Psychiatry Neurosci 33(2):91–99
Hajek T, Novak T, Kopecek M, Gunde E, Alda M, Höschl C (2010) Subgenual cingulate volumes in offspring of bipolar parents and in sporadic bipolar patients. Eur Arch Psychiatry Clin Neurosci 260(4):297–304. doi:10.1007/s00406-009-0077-2
Hajek T, Gunde E, Bernier D, Slaney C, Propper L, MacQueen G, Duffy A, Alda M (2008) Pituitary volumes in relatives of bipolar patients. Eur Arch Psychiatry Clin Neurosci 258(6):357–362
Ladouceur CD, Almeida JRC, Birmaher B, Axelson DA, Nau S, Kalas C, Monk K, Kupfer DJ, Phillips ML (2008) Subcortical gray matter volume abnormalities in healthy bipolar offspring: potential neuroanatomical risk marker for bipolar disorder? J Am Acad Child Adolesc Psychiatry 47(5):532–539. doi:10.1097/CHI.0b013e318167656e
Bauer IE, Sanches M, Suchting R, Green CE, El Fangary NM, Zunta-Soares GB, Soares JC (2014) Amygdala enlargement in unaffected offspring of bipolar parents. J Psychiatr Res 59:200–205
Hajek T, Cullis J, Novak T, Kopecek M, Blagdon R, Propper L, Stopkova P, Duffy A, Hoschl C, Uher R, Paus T, Young LT, Alda M (2013) Brain structural signature of familial predisposition for bipolar disorder: replicable evidence for involvement of the right inferior frontal gyrus. Biol Psychiatry 73(2):144–152. doi:10.1016/j.biopsych.2012.06.015
Karchemskiy A, Garrett A, Howe M, Adleman N, Simeonova DI, Alegria D, Reiss A, Chang K (2011) Amygdalar, hippocampal, and thalamic volumes in youth at high risk for development of bipolar disorder. Psychiatry Res 194(3):319–325. doi:10.1016/j.pscychresns.2011.03.006
Sugranyes G, de la Serna E, Romero S, Sanchez-Gistau V, Calvo A, Moreno D, Baeza I, Diaz-Caneja CM, Sanchez-Gutierrez T, Janssen J (2015) Grey matter volume decrease distinguishes schizophrenia from bipolar offspring during childhood and adolescence. J Am Acad Child Adolesc Psychiatry 54(8):677
Chang KD, Karchemskiy A, Barnea-Goraly N, Garrett A, Simeonova DI, Reiss A (2005) Reduced amygdalar gray matter volume in familial pediatric bipolar disorder. J Am Acad Child Adolesc Psychiatry 44(6):565–573. doi:10.1097/01.chi.0000159948.75136.0d
Hajek T, Gunde E, Slaney C, Propper L, MacQueen G, Duffy A, Alda M (2009) Striatal volumes in affected and unaffected relatives of bipolar patients—high-risk study. J Psychiatr Res 43(7):724–729. doi:10.1016/j.jpsychires.2008.10.008
Simeonova DI, Jackson V, Attalla A, Karchemskiy A, Howe M, Adleman N, Chang K (2009) Subcortical volumetric correlates of anxiety in familial pediatric bipolar disorder: a preliminary investigation. Psychiatry Research: Neuroimaging 173(2):113–120
Stanfield AC, Moorhead TWJ, Job DE, McKirdy J, Sussmann JE, Hall J, Giles S, Johnstone EC, Lawrie SM, McIntosh AM (2009) Structural abnormalities of ventrolateral and orbitofrontal cortex in patients with familial bipolar disorder. Bipolar Disorders 11(2):135–144
Chen HH, Nicoletti MA, Hatch JP, Sassi RB, Axelson D, Brambilla P, Monkul ES, Keshavan MS, Ryan ND, Birmaher B (2004) Abnormal left superior temporal gyrus volumes in children and adolescents with bipolar disorder: a magnetic resonance imaging study. Neurosci Lett 363(1):65–68
Nugent AC, Milham MP, Bain EE, Mah L, Cannon DM, Marrett S, Zarate CA, Pine DS, Price JL, Drevets WC (2006) Cortical abnormalities in bipolar disorder investigated with MRI and voxel-based morphometry. Neuroimage 30(2):485–497
Frangou S (2005) The Maudsley bipolar disorder project. Epilepsia 46(s4):19–25
Narita K, Suda M, Takei Y, Aoyama Y, Majima T, Kameyama M, Kosaka H, Amanuma M, Fukuda M, Mikuni M (2011) Volume reduction of ventromedial prefrontal cortex in bipolar II patients with rapid cycling: a voxel-based morphometric study. Progr Neuro-Psychopharmacol Biol Psychiatry 35(2):439–445
Wilke M, Kowatch RA, DelBello MP, Mills NP, Holland SK (2004) Voxel-based morphometry in adolescents with bipolar disorder: first results. Psychiatry Res: Neuroimaging 131(1):57–69
James A, Hough M, James S, Burge L, Winmill L, Nijhawan S, Matthews PM, Zarei M (2011) Structural brain and neuropsychometric changes associated with pediatric bipolar disorder with psychosis. Bipolar Disorders 13(1):16–27. doi:10.1111/j.1399-5618.2011.00891.x
Dickstein DP, Milham MP, Nugent AC et al (2005) Frontotemporal alterations in pediatric bipolar disorder: results of a voxel-based morphometry study. Arch Gen Psychiatry 62(7):734–741. doi:10.1001/archpsyc.62.7.734
Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P, Williamson D, Ryan N (1997) Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry 36(7):980–988
First MB, Spitzer RL, Gibbon M, Williams JB (2012) Structured clinical interview for DSM-IV® axis I disorders (SCID-I), clinician version, administration booklet. American Psychiatric Press, Washington, DC
de Bie HM, Boersma M, Wattjes MP, Adriaanse S, Vermeulen RJ, Oostrom KJ, Huisman J, Veltman DJ, Delemarre-Van de Waal HA (2010) Preparing children with a mock scanner training protocol results in high quality structural and functional MRI scans. Eur J Pediatr 169(9):1079–1085
Nora M R, Michelle L, Roman B, Joanna A C, Maria C, Monica V, Patrice L S, Nadine G (2009) Making MR imaging child’s play-pediatric neuroimaging protocol, guidelines and procedure. J Vis Exp (29):1309
Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ, Frackowiak RS (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. NeuroImage 14:21–36
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. Neuroimage 11(6):805–821
Wilke M, Holland SK, Altaye M, Gaser C (2008) Template-O-Matic: a toolbox for creating customized pediatric templates. Neuroimage 41(3):903–913
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, Mazoyer B, Joliot M (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15(1):273–289
Maldjian JA, Laurienti PJ, Burdette JH (2004) Precentral gyrus discrepancy in electronic versions of the Talairach atlas. Neuroimage 21(1):450–455
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH (2003) An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage 19(3):1233–1239
Nakao T, Radua J, Rubia K, Mataix-Cols D (2011) Gray matter volume abnormalities in ADHD: voxel-based meta-analysis exploring the effects of age and stimulant medication. Perspectives 168(11):1154–1163
Carmona S, Vilarroya O, Bielsa A, Tremols V, Soliva J, Rovira M, Tomas J, Raheb C, Gispert J, Batlle S (2005) Global and regional gray matter reductions in ADHD: a voxel-based morphometric study. Neuroscience Letters 389(2):88–93
Bora E, Fornito A, Pantelis C, Yücel M (2012) Gray matter abnormalities in major depressive disorder: a meta-analysis of voxel based morphometry studies. J Affect Disord 138(1):9–18
Vasic N, Walter H, Höse A, Wolf RC (2008) Gray matter reduction associated with psychopathology and cognitive dysfunction in unipolar depression: a voxel-based morphometry study. J Affect Disord 109(1):107–116
Radua J, van den Heuvel OA, Surguladze S, Mataix-Cols D (2010) Meta-analytical comparison of voxel-based morphometry studies in obsessive-compulsive disorder vs other anxiety disorders. Arch Gen Psychiatry 67(7):701–711
Uchida RR, Del-Ben CM, Busatto GF, Duran FL, Guimarães FS, Crippa JA, Araújo D, Santos AC, Graeff FG (2008) Regional gray matter abnormalities in panic disorder: a voxel-based morphometry study. Psychiatry Res: Neuroimaging 163(1):21–29
Ratnanather JT, Cebron S, Ceyhan E, Postell E, Pisano DV, Poynton CB, Crocker B, Honeycutt NA, Mahon PB, Barta PE (2014) Morphometric differences in planum temporale in schizophrenia and bipolar disorder revealed by statistical analysis of labeled cortical depth maps. Front Psychiatry 5(94):1–7
Price CJ (2000) The anatomy of language: contributions from functional neuroimaging. J Anat 197(3):335–359
Price JL, Drevets WC (2010) Neurocircuitry of mood disorders. Neuropsychopharmacol: Off Publ Am Coll Neuropsychopharmacol 35(1):192–216. doi:10.1038/npp.2009.104
Griffiths TD, Warren JD (2002) The planum temporale as a computational hub. Trends Neurosci 25(7):348–353
Olson IR, Plotzker A, Ezzyat Y (2007) The enigmatic temporal pole: a review of findings on social and emotional processing. Brain 130(Pt 7):1718–1731. doi:10.1093/brain/awm052
Olsavsky AK, Brotman MA, Rutenberg JG, Muhrer EJ, Deveney CM, Fromm SJ, Towbin K, Pine DS, Leibenluft E (2012) Amygdala hyperactivation during face emotion processing in unaffected youth at risk for bipolar disorder. J Am Acad Child Adolesc Psychiatry 51(3):294–303. doi:10.1016/j.jaac.2011.12.008
Ladouceur CD, Diwadkar VA, White R, Bass J, Birmaher B, Axelson DA, Phillips ML (2013) Fronto-limbic function in unaffected offspring at familial risk for bipolar disorder during an emotional working memory paradigm. Dev Cognit Neurosci 5:185–196. doi:10.1016/j.dcn.2013.03.004
Kim P, Jenkins SE, Connolly ME, Deveney CM, Fromm SJ, Brotman MA, Nelson EE, Pine DS, Leibenluft E (2012) Neural correlates of cognitive flexibility in children at risk for bipolar disorder. J Psychiatr Res 46(1):22–30
Thermenos HW, Goldstein JM, Milanovic SM, Whitfield-Gabrieli S, Makris N, LaViolette P, Koch JK, Faraone SV, Tsuang MT, Buka SL (2010) An fMRI study of working memory in persons with bipolar disorder or at genetic risk for bipolar disorder. Am J Med Genet Part B: Neuropsychiatr Genet 153(1):120–131
Roberts G, Green MJ, Breakspear M, McCormack C, Frankland A, Wright A, Levy F, Lenroot R, Chan HN, Mitchell PB (2013) Reduced inferior frontal gyrus activation during response inhibition to emotional stimuli in youth at high risk of bipolar disorder. Biol Psychiatry 74(1):55–61
Whalley HC, Sussmann JE, Romaniuk L, Stewart T (2013) Prediction of depression in individuals at high familial risk of mood disorders using functional magnetic resonance imaging. PLoS One 8(3):e57357–e57365
Park H-J, Friston K (2013) Structural and functional brain networks: from connections to cognition. Science 342(6158):1238411. doi:10.1126/science.1238411
Rykhlevskaia E, Gratton G, Fabiani M (2008) Combining structural and functional neuroimaging data for studying brain connectivity: a review. Psychophysiology 45(2):173–187. doi:10.1111/j.1469-8986.2007.00621.x
Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC, Nugent TF, Herman DH, Clasen LS, Toga AW (2004) Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci USA 101(21):8174–8179
Paus T, Keshavan M, Giedd JN (2008) Why do many psychiatric disorders emerge during adolescence? Nat Rev Neurosci 9(12):947–957
Sowell ER, Peterson BS, Thompson PM, Welcome SE, Henkenius AL, Toga AW (2003) Mapping cortical change across the human life span. Nat Neurosci 6(3):309–315
Huttenlocher PR (1990) Morphometric study of human cerebral cortex development. Neuropsychologia 28(6):517–527
Lemaitre H, Goldman AL, Sambataro F, Verchinski BA, Meyer-Lindenberg A, Weinberger DR, Mattay VS (2012) Normal age-related brain morphometric changes: nonuniformity across cortical thickness, surface area and gray matter volume? Neurobiol Aging 33(3):617.e611–617.e619
Gogtay N, Thompson PM (2010) Mapping gray matter development: implications for typical development and vulnerability to psychopathology. Brain Cognit 72(1):6–15
Acknowledgments
The authors would like to graciously acknowledge the Brain & Behaviour Research Foundation (formerly known as NARSAD) for their financial contribution to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
RBS received consulting and speaker support from BMS, Jansen. The remaining authors do not have any associations that might pose a conflict of interest in relation to this manuscript.
Rights and permissions
About this article
Cite this article
Hanford, L.C., Hall, G.B., Minuzzi, L. et al. Gray matter volumes in symptomatic and asymptomatic offspring of parents diagnosed with bipolar disorder. Eur Child Adolesc Psychiatry 25, 959–967 (2016). https://doi.org/10.1007/s00787-015-0809-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-015-0809-y