Abstract
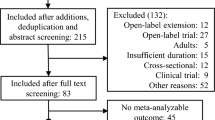
We performed an updated review of the available literature on weight gain and increase of body mass index (BMI) among children and adolescents treated with antipsychotic medications. A PubMed search was conducted specifying the following MeSH terms: (antipsychotic agents) hedged with (weight gain) or (body mass index). We selected 127 reports, including 71 intervention trials, 42 observational studies and 14 literature reviews. Second-generation antipsychotics (SGAs), in comparison with first-generation antipsychotics, are associated with a greater risk for antipsychotic-induced weight gain although this oversimplification should be clarified by distinguishing across different antipsychotic drugs. Among SGAs, olanzapine appears to cause the most significant weight gain, while ziprasidone seems to cause the least. Antipsychotic-induced BMI increase appears to remain regardless of the specific psychotropic co-treatment. Children and adolescents seem to be at a greater risk than adults for antipsychotic-induced weight gain; and the younger the child, the higher the risk. Genetic or environmental factors related to antipsychotic-induced weight gain among children and adolescents are mostly unknown, although certain genetic factors related to serotonin receptors or hormones such as leptin, adiponectin or melanocortin may be involved. Strategies to reduce this antipsychotic side effect include switching to another antipsychotic drug, lowering the dosage or initiating treatment with metformin or topiramate, as well as non-pharmacological interventions. Future research should avoid some methodological limitations such as not accounting for age- and sex-adjusted BMI (zBMI), small sample size, short period of treatment, great heterogeneity of diagnoses and confounding by indication.
Similar content being viewed by others
References
Almandil NB, Wong IC (2011) Review on the current use of antipsychotic drugs in children and adolescents. Arch Dis Child Educ Pract Ed 96:192–196
Aman MG, Arnold LE, McDougle CJ, Vitiello B, Scahill L, Davies M, McCracken JT, Tierney E, Nash PL, Posey DJ, Chuang S, Martin A, Shah B, Gonzalez NM, Swiezy NB, Ritz L, Koenig K, McGough J, Ghuman JK, Lindsay RL (2005) Acute and long-term safety and tolerability of risperidone in children with autism. J Child Adolesc Psychopharmacol 15:869–884
Aman MG, Binder C, Turgay A (2004) Risperidone effects in the presence/absence of psychostimulant medicine in children with ADHD, other disruptive behavior disorders, and subaverage IQ. J Child Adolesc Psychopharmacol 14:243–254
Arango C, Robles O, Parellada M, Fraguas D, Ruiz-Sancho A, Medina O, Zabala A, Bombin I, Moreno D (2009) Olanzapine compared to quetiapine in adolescents with a first psychotic episode. Eur Child Adolesc Psychiatry 18:418–428
Arman S, Sadramely MR, Nadi M, Koleini N (2008) A randomized, double-blind, placebo-controlled trial of metformin treatment for weight gain associated with initiation of risperidone in children and adolescents. Saudi Med J 29:1130–1134
Armenteros JL, Davies M (2006) Antipsychotics in early onset Schizophrenia: systematic review and meta-analysis. Eur Child Adolesc Psychiatry 15:141–148
Armenteros JL, Lewis JE, Davalos M (2007) Risperidone augmentation for treatment-resistant aggression in attention-deficit/hyperactivity disorder: a placebo-controlled pilot study. J Am Acad Child Adolesc Psychiatry 46:558–565
Armenteros JL, Whitaker AH, Welikson M, Stedge DJ, Gorman J (1997) Risperidone in adolescents with schizophrenia: an open pilot study. J Am Acad Child Adolesc Psychiatry 36:694–700
Arnold LE, Farmer C, Kraemer HC, Davies M, Witwer A, Chuang S, DiSilvestro R, McDougle CJ, McCracken J, Vitiello B, Aman MG, Scahill L, Posey DJ, Swiezy NB (2010) Moderators, mediators, and other predictors of risperidone response in children with autistic disorder and irritability. J Child Adolesc Psychopharmacol 20:83–93
Bachmann CJ, Gebhardt S, Lehr D, Haberhausen M, Kaiser C, Otto B, Theisen FM (2012) Subjective and biological weight-related parameters in adolescents and young adults with schizophrenia spectrum disorder under clozapine or olanzapine treatment. Z Kinder Jugendpsychiatr Psychother 40:151–158 (quiz 158–159)
Bachmann CJ, Haberhausen M, Heinzel-Gutenbrunner M, Remschmidt H, Theisen FM (2008) Large intraindividual variability of olanzapine serum concentrations in adolescent patients. Ther Drug Monit 30:108–112
Bachmann CJ, Rieger-Gies A, Heinzel-Gutenbrunner M, Hiemke C, Remschmidt H, Theisen FM (2008) Large variability of aripiprazole and dehydroaripiprazole serum concentrations in adolescent patients with schizophrenia. Ther Drug Monit 30:462–466
Biederman J, Mick E, Spencer T, Dougherty M, Aleardi M, Wozniak J (2007) A prospective open-label treatment trial of ziprasidone monotherapy in children and adolescents with bipolar disorder. Bipolar Disord 9:888–894
Biederman J, Mick E, Spencer T, Doyle R, Joshi G, Hammerness P, Kotarski M, Aleardi M, Wozniak J (2007) An open-label trial of aripiprazole monotherapy in children and adolescents with bipolar disorder. CNS Spectr 12:683–689
Björkhem-Bergman L, Asplund AB, Lindh JD (2011) Metformin for weight reduction in non-diabetic patients on antipsychotic drugs: a systematic review and meta-analysis. J Psychopharmacol 25:299–305
Bozina N, Medved V, Kuzman MR, Sain I, Sertic J (2007) Association study of olanzapine-induced weight gain and therapeutic response with SERT gene polymorphisms in female schizophrenic patients. J Psychopharmacol 21:728–734
Brown S, Birtwistle J, Roe L, Thompson C (1999) The unhealthy lifestyle of people with schizophrenia. Psychol Med 29:697–701
Buitelaar JK (2000) Open-label treatment with risperidone of 26 psychiatrically-hospitalized children and adolescents with mixed diagnoses and aggressive behavior. J Child Adolesc Psychopharmacol 10:19–26
Buitelaar JK, van der Gaag RJ, Cohen-Kettenis P, Melman CT (2001) A randomized controlled trial of risperidone in the treatment of aggression in hospitalized adolescents with subaverage cognitive abilities. J Clin Psychiatry 62:239–248
Cabanac M, Duclaux R, Spector NH (1971) Sensory feedback in regulation of body weight: is there a ponderostat? Nature 229:125–127
Calarge CA, Acion L, Kuperman S, Tansey M, Schlechte JA (2009) Weight gain and metabolic abnormalities during extended risperidone treatment in children and adolescents. J Child Adolesc Psychopharmacol 19:101–109
Calarge CA, Ellingrod VL, Zimmerman B, Acion L, Sivitz WI, Schlechte JA (2009) Leptin gene -2548G/A variants predict risperidone-associated weight gain in children and adolescents. Psychiatr Genet 19:320–327
Calarge CA, del Miller D (2011) Predictors of risperidone and 9-hydroxyrisperidone serum concentration in children and adolescents. J Child Adolesc Psychopharmacol 21:163–169
Canitano R (2005) Clinical experience with Topiramate to counteract neuroleptic induced weight gain in 10 individuals with autistic spectrum disorders. Brain Dev 27:228–232
Castro-Fornieles J, Parellada M, Soutullo CA, Baeza I, Gonzalez-Pinto A, Graell M, Paya B, Moreno D, de la Serna E, Arango C (2008) Antipsychotic treatment in child and adolescent first-episode psychosis: a longitudinal naturalistic approach. J Child Adolesc Psychopharmacol 18:327–336
Citrome L, Vreeland B (2008) Schizophrenia, obesity, and antipsychotic medications: what can we do? Postgrad Med 120:18–33
Cohen D, Bonnot O, Bodeau N, Consoli A, Laurent C (2012) Adverse effects of second-generation antipsychotics in children and adolescents: a Bayesian meta-analysis. J Clin Psychopharmacol 32:309–316
Correia CT, Almeida JP, Santos PE, Sequeira AF, Marques CE, Miguel TS, Abreu RL, Oliveira GG, Vicente AM (2010) Pharmacogenetics of risperidone therapy in autism: association analysis of eight candidate genes with drug efficacy and adverse drug reactions. Pharmacogenomics J 10:418–430
Correia FAG, Bodanese R, Silva TL, Alvares JP, Aman M, Rohde LA (2005) Comparison of risperidone and methylphenidate for reducing ADHD symptoms in children and adolescents with moderate mental retardation. J Am Acad Child Adolesc Psychiatry 44:748–755
Correll CU (2011) Addressing adverse effects of antipsychotic treatment in young patients with schizophrenia. J Clin Psychiatry 72:e01
Correll CU (2005) Metabolic side effects of second-generation antipsychotics in children and adolescents: a different story? J Clin Psychiatry 66:1331–1332
Correll CU (2007) Weight gain and metabolic effects of mood stabilizers and antipsychotics in pediatric bipolar disorder: a systematic review and pooled analysis of short-term trials. J Am Acad Child Adolesc Psychiatry 46:687–700
Correll CU, Carlson HE (2006) Endocrine and metabolic adverse effects of psychotropic medications in children and adolescents. J Am Acad Child Adolesc Psychiatry 45:771–791
Correll CU, Manu P, Olshanskiy V, Napolitano B, Kane JM, Malhotra AK (2009) Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA 302:1765–1773
Correll CU, Penzner JB, Parikh UH, Mughal T, Javed T, Carbon M, Malhotra AK (2006) Recognizing and monitoring adverse events of second-generation antipsychotics in children and adolescents. Child Adolesc Psychiatr Clin N Am 15:177–206
Correll CU, Sheridan EM, DelBello MP (2010) Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord 12:116–141
Crocq MA, Guillon MS, Bailey PE, Provost D (2007) Orally disintegrating olanzapine induces less weight gain in adolescents than standard oral tablets. Eur Psychiatry 22:453–454
Cui YH, Zheng Y, Yang YP, Liu J, Li J (2010) Effectiveness and tolerability of aripiprazole in children and adolescents with Tourette’s disorder: a pilot study in China. J Child Adolesc Psychopharmacol 20:291–298
Cheng-Shannon J, McGough JJ, Pataki C, McCracken JT (2004) Second-generation antipsychotic medications in children and adolescents. J Child Adolesc Psychopharmacol 14:372–394
De Hert M, Dobbelaere M, Sheridan EM, Cohen D, Correll CU (2011) Metabolic and endocrine adverse effects of second-generation antipsychotics in children and adolescents: a systematic review of randomized, placebo controlled trials and guidelines for clinical practice. Eur Psychiatry 26:144–158
de Hoogd S, Overbeek WA, Heerdink ER, Correll CU, de Graeff ER, Staal WG (2012) Differences in body mass index z-scores and weight status in a Dutch pediatric psychiatric population with and without use of second-generation antipsychotics. J Child Adolesc Psychopharmacol 22:166–173
Degrauw RS, Li JZ, Gilbert DL (2009) Body mass index changes and chronic neuroleptic drug treatment for Tourette syndrome. Pediatr Neurol 41:183–186
DelBello MP, Adler CM, Whitsel RM, Stanford KE, Strakowski SM (2007) A 12-week single-blind trial of quetiapine for the treatment of mood symptoms in adolescents at high risk for developing bipolar I disorder. J Clin Psychiatry 68:789–795
DelBello MP, Chang K, Welge JA, Adler CM, Rana M, Howe M, Bryan H, Vogel D, Sampang S, Delgado SV, Sorter M, Strakowski SM (2009) A double-blind, placebo-controlled pilot study of quetiapine for depressed adolescents with bipolar disorder. Bipolar Disord 11:483–493
DelBello MP, Kowatch RA, Adler CM, Stanford KE, Welge JA, Barzman DH, Nelson E, Strakowski SM (2006) A double-blind randomized pilot study comparing quetiapine and divalproex for adolescent mania. J Am Acad Child Adolesc Psychiatry 45:305–313
DelBello MP, Versavel M, Ice K, Keller D, Miceli J (2008) Tolerability of oral ziprasidone in children and adolescents with bipolar mania, schizophrenia, or schizoaffective disorder. J Child Adolesc Psychopharmacol 18:491–499
Dittmann RW, Meyer E, Freisleder FJ, Remschmidt H, Mehler-Wex C, Junghanss J, Hagenah U, Schulte-Markwort M, Poustka F, Schmidt MH, Schulz E, Mastele A, Wehmeier PM (2008) Effectiveness and tolerability of olanzapine in the treatment of adolescents with schizophrenia and related psychotic disorders: results from a large, prospective, open-label study. J Child Adolesc Psychopharmacol 18:54–69
Doey T (2012) Aripiprazole in pediatric psychosis and bipolar disorder: a clinical review. J Affect Disord 138(Suppl):S15–S21
Dori N, Green T (2011) The metabolic syndrome and antipsychotics in children and adolescents. Harefuah 150:791–796, 814, 813
Eapen V, John G (2011) Weight gain and metabolic syndrome among young patients on antipsychotic medication: what do we know and where do we go? Australas Psychiatry 19:232–235
Edlinger M, Hofer A, Rettenbacher MA, Baumgartner S, Widschwendter CG, Kemmler G, Neco NA, Fleischhacker WW (2009) Factors influencing the choice of new generation antipsychotic medication in the treatment of patients with schizophrenia. Schizophr Res 113:246–251
Fedorowicz VJ, Fombonne E (2005) Metabolic side effects of atypical antipsychotics in children: a literature review. J Psychopharmacol 19:533–550
Findling RL (2008) Atypical antipsychotic treatment of disruptive behavior disorders in children and adolescents. J Clin Psychiatry 69(Suppl 4):9–14
Findling RL, Aman MG, Eerdekens M, Derivan A, Lyons B (2004) Long-term, open-label study of risperidone in children with severe disruptive behaviors and below-average IQ. Am J Psychiatry 161:677–684
Findling RL, Cavus I, Pappadolulos E, Backinsky M, Schwartz JH, Vanderburg DG (2010) A placebo-controlled trial to evaluate the efficacy and safety of flexibly dosed oral ziprasidone in adolescent subjects with schizophrenia. Schizophr Res 117:437
Findling RL, Johnson JL, McClellan J, Frazier JA, Vitiello B, Hamer RM, Lieberman JA, Ritz L, McNamara NK, Lingler J, Hlastala S, Pierson L, Puglia M, Maloney AE, Kaufman EM, Noyes N, Sikich L (2010) Double-blind maintenance safety and effectiveness findings from the Treatment of Early-Onset Schizophrenia Spectrum (TEOSS) study. J Am Acad Child Adolesc Psychiatry 49:583–594 (quiz 632)
Findling RL, McNamara NK, Branicky LA, Schluchter MD, Lemon E, Blumer JL (2000) A double-blind pilot study of risperidone in the treatment of conduct disorder. J Am Acad Child Adolesc Psychiatry 39:509–516
Findling RL, McNamara NK, Youngstrom EA, Stansbrey RJ, Frazier TW, Lingler J, Otto BD, Demeter CA, Rowles BM, Calabrese JR (2011) An open-label study of aripiprazole in children with a bipolar disorder. J Child Adolesc Psychopharmacol 21:345–351
Findling RL, Nyilas M, Forbes RA, McQuade RD, Jin N, Iwamoto T, Ivanova S, Carson WH, Chang K (2009) Acute treatment of pediatric bipolar I disorder, manic or mixed episode, with aripiprazole: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry 70:1441–1451
Findling RL, Robb A, Nyilas M, Forbes RA, Jin N, Ivanova S, Marcus R, McQuade RD, Iwamoto T, Carson WH (2008) A multiple-center, randomized, double-blind, placebo-controlled study of oral aripiprazole for treatment of adolescents with schizophrenia. Am J Psychiatry 165:1432–1441
Fleischhaker C, Heiser P, Hennighausen K, Herpertz-Dahlmann B, Holtkamp K, Mehler-Wex C, Rauh R, Remschmidt H, Schulz E, Warnke A (2006) Clinical drug monitoring in child and adolescent psychiatry: side effects of atypical neuroleptics. J Child Adolesc Psychopharmacol 16:308–316
Fleischhaker C, Heiser P, Hennighausen K, Herpertz-Dahlmann B, Holtkamp K, Mehler-Wex C, Rauh R, Remschmidt H, Schulz E, Warnke A (2007) Weight gain associated with clozapine, olanzapine and risperidone in children and adolescents. J Neural Transm 114:273–280
Fleischhaker C, Heiser P, Hennighausen K, Herpertz-Dahlmann B, Holtkamp K, Mehler-Wex C, Rauh R, Remschmidt H, Schulz E, Warnke A (2008) Weight gain in children and adolescents during 45 weeks treatment with clozapine, olanzapine and risperidone. J Neural Transm 115:1599–1608
Fraguas D, Correll CU, Merchan-Naranjo J, Rapado-Castro M, Parellada M, Moreno C, Arango C (2011) Efficacy and safety of second-generation antipsychotics in children and adolescents with psychotic and bipolar spectrum disorders: comprehensive review of prospective head-to-head and placebo-controlled comparisons. Eur Neuropsychopharmacol 21:621–645
Fraguas D, Merchan-Naranjo J, Laita P, Parellada M, Moreno D, Ruiz-Sancho A, Cifuentes A, Giraldez M, Arango C (2008) Metabolic and hormonal side effects in children and adolescents treated with second-generation antipsychotics. J Clin Psychiatry 69:1166–1175
Frazier JA, Biederman J, Tohen M, Feldman PD, Jacobs TG, Toma V, Rater MA, Tarazi RA, Kim GS, Garfield SB, Sohma M, Gonzalez-Heydrich J, Risser RC, Nowlin ZM (2001) A prospective open-label treatment trial of olanzapine monotherapy in children and adolescents with bipolar disorder. J Child Adolesc Psychopharmacol 11:239–250
Frazier JA, Meyer MC, Biederman J, Wozniak J, Wilens TE, Spencer TJ, Kim GS, Shapiro S (1999) Risperidone treatment for juvenile bipolar disorder: a retrospective chart review. J Am Acad Child Adolesc Psychiatry 38:960–965
Frémaux T, Reymann JM, Chevreuil C, Bentue-Ferrer D (2007) Prescription of olanzapine in children and adolescent psychiatric patients. Encephale 33:188–196
Gagliano A, Germano E, Pustorino G, Impallomeni C, D’Arrigo C, Calamoneri F, Spina E (2004) Risperidone treatment of children with autistic disorder: effectiveness, tolerability, and pharmacokinetic implications. J Child Adolesc Psychopharmacol 14:39–47
Gebhardt S, Haberhausen M, Heinzel-Gutenbrunner M, Gebhardt N, Remschmidt H, Krieg JC, Hebebrand J, Theisen FM (2009) Antipsychotic-induced body weight gain: predictors and a systematic categorization of the long-term weight course. J Psychiatr Res 43:620–626
Gebhardt S, Theisen FM, Haberhausen M, Heinzel-Gutenbrunner M, Wehmeier PM, Krieg JC, Kuhnau W, Schmidtke J, Remschmidt H, Hebebrand J (2010) Body weight gain induced by atypical antipsychotics: an extension of the monozygotic twin and sib pair study. J Clin Pharm Ther 35:207–211
Geller B, Luby JL, Joshi P, Wagner KD, Emslie G, Walkup JT, Axelson DA, Bolhofner K, Robb A, Wolf DV, Riddle MA, Birmaher B, Nusrat N, Ryan ND, Vitiello B, Tillman R, Lavori P (2012) A randomized controlled trial of risperidone, lithium, or divalproex sodium for initial treatment of bipolar I disorder, manic or mixed phase, in children and adolescents. Arch Gen Psychiatry 69:515–528
Gencer O, Emiroglu FN, Miral S, Baykara B, Baykara A, Dirik E (2008) Comparison of long-term efficacy and safety of risperidone and haloperidol in children and adolescents with autistic disorder. An open label maintenance study. Eur Child Adolesc Psychiatry 17:217–225
Goeb JL, Marco S, Duhamel A, Kechid G, Bordet R, Thomas P, Delion P, Jardri R (2010) Metabolic side effects of risperidone in early onset schizophrenia. Encephale 36:242–252
Goldstein BI, Birmaher B, Axelson DA, Goldstein TR, Esposito-Smythers C, Strober MA, Hunt J, Leonard H, Gill MK, Iyengar S, Grimm C, Yang M, Ryan ND, Keller MB (2008) Preliminary findings regarding overweight and obesity in pediatric bipolar disorder. J Clin Psychiatry 69:1953–1959
Gothelf D, Falk B, Singer P, Kairi M, Phillip M, Zigel L, Poraz I, Frishman S, Constantini N, Zalsman G, Weizman A, Apter A (2002) Weight gain associated with increased food intake and low habitual activity levels in male adolescent schizophrenic inpatients treated with olanzapine. Am J Psychiatry 159:1055–1057
Green SB, Byar DP (1984) Using observational data from registries to compare treatments: the fallacy of omnimetrics. Stat Med 3:361–373
Gurpegui M, Martinez-Ortega JM, Gutierrez-Rojas L, Rivero J, Rojas C, Jurado D (2012) Overweight and obesity in patients with bipolar disorder or schizophrenia compared with a non-psychiatric sample. Prog Neuropsychopharmacol Biol Psychiatry 37:169–175
Haas M, Delbello MP, Pandina G, Kushner S, Van Hove I, Augustyns I, Quiroz J, Kusumakar V (2009) Risperidone for the treatment of acute mania in children and adolescents with bipolar disorder: a randomized, double-blind, placebo-controlled study. Bipolar Disord 11:687–700
Haas M, Eerdekens M, Kushner S, Singer J, Augustyns I, Quiroz J, Pandina G, Kusumakar V (2009) Efficacy, safety and tolerability of two dosing regimens in adolescent schizophrenia: double-blind study. Br J Psychiatry 194:158–164
Haas M, Karcher K, Pandina GJ (2008) Treating disruptive behavior disorders with risperidone: a 1-year, open-label safety study in children and adolescents. J Child Adolesc Psychopharmacol 18:337–345
Handen BL, Hardan AY (2006) Open-label, prospective trial of olanzapine in adolescents with subaverage intelligence and disruptive behavioral disorders. J Am Acad Child Adolesc Psychiatry 45:928–935
Hoekstra PJ, Troost PW, Lahuis BE, Mulder H, Mulder EJ, Franke B, Buitelaar JK, Anderson GM, Scahill L, Minderaa RB (2010) Risperidone-induced weight gain in referred children with autism spectrum disorders is associated with a common polymorphism in the 5-hydroxytryptamine 2C receptor gene. J Child Adolesc Psychopharmacol 20:473–477
Hollander E, Wasserman S, Swanson EN, Chaplin W, Schapiro ML, Zagursky K, Novotny S (2006) A double-blind placebo-controlled pilot study of olanzapine in childhood/adolescent pervasive developmental disorder. J Child Adolesc Psychopharmacol 16:541–548
Hrdlicka M, Zedkova I, Blatny M, Urbanek T (2009) Weight gain associated with atypical and typical antipsychotics during treatment of adolescent schizophrenic psychoses: a retrospective study. Neuro Endocrinol Lett 30:256–261
Jassim G, Ferno J, Theisen FM, Haberhausen M, Christoforou A, Havik B, Gebhardt S, Remschmidt H, Mehler-Wex C, Hebebrand J, Lehellard S, Steen VM (2011) Association study of energy homeostasis genes and antipsychotic-induced weight gain in patients with schizophrenia. Pharmacopsychiatry 44:15–20
Jensen JB, Kumra S, Leitten W, Oberstar J, Anjum A, White T, Wozniak J, Lee SS, Schulz SC (2008) A comparative pilot study of second-generation antipsychotics in children and adolescents with schizophrenia-spectrum disorders. J Child Adolesc Psychopharmacol 18:317–326
Jerrell JM, McIntyre RS (2008) Adverse events in children and adolescents treated with antipsychotic medications. Hum Psychopharmacol 23:283–290
Jolin EM, Weller EB, Weller RA (2007) The public health aspects of bipolar disorder in children and adolescents. Curr Psychiatry Rep 9:106–113
Joshi G, Petty C, Wozniak J, Faraone SV, Doyle R, Georgiopoulos A, Hammerness P, Walls S, Glaeser B, Brethel K, Yorks D, Biederman J (2012) A prospective open-label trial of quetiapine monotherapy in preschool and school age children with bipolar spectrum disorder. J Affect Disord 136:1143–1153
Kapetanovic S, Aaron L, Montepiedra G, Sirois PA, Oleske JM, Malee K, Pearson DA, Nichols SL, Garvie PA, Farley J, Nozyce ML, Mintz M, Williams PL (2009) The use of second-generation antipsychotics and the changes in physical growth in children and adolescents with perinatally acquired HIV. AIDS Patient Care STDS 23:939–947
Karagianis J, Grossman L, Landry J, Reed VA, de Haan L, Maguire GA, Hoffmann VP, Milev R (2009) A randomized controlled trial of the effect of sublingual orally disintegrating olanzapine versus oral olanzapine on body mass index: the PLATYPUS Study. Schizophr Res 113:41–48
Kelly DL, Conley RR, Love RC, Horn DS, Ushchak CM (1998) Weight gain in adolescents treated with risperidone and conventional antipsychotics over six months. J Child Adolesc Psychopharmacol 8:151–159
Kemner C, Willemsen-Swinkels SH, de Jonge M, Tuynman-Qua H, van Engeland H (2002) Open-label study of olanzapine in children with pervasive developmental disorder. J Clin Psychopharmacol 22:455–460
Khan RA, Mican LM, Suehs BT (2009) Effects of olanzapine and risperidone on metabolic factors in children and adolescents: a retrospective evaluation. J Psychiatr Pract 15:320–328
Klein DJ, Cottingham EM, Sorter M, Barton BA, Morrison JA (2006) A randomized, double-blind, placebo-controlled trial of metformin treatment of weight gain associated with initiation of atypical antipsychotic therapy in children and adolescents. Am J Psychiatry 163:2072–2079
Kryzhanovskaya L, Schulz SC, McDougle C, Frazier J, Dittmann R, Robertson-Plouch C, Bauer T, Xu W, Wang W, Carlson J, Tohen M (2009) Olanzapine versus placebo in adolescents with schizophrenia: a 6-week, randomized, double-blind, placebo-controlled trial. J Am Acad Child Adolesc Psychiatry 48:60–70
Kryzhanovskaya LA, Robertson-Plouch CK, Xu W, Carlson JL, Merida KM, Dittmann RW (2009) The safety of olanzapine in adolescents with schizophrenia or bipolar I disorder: a pooled analysis of 4 clinical trials. J Clin Psychiatry 70:247–258
Kryzhanovskaya LA, Xu W, Millen BA, Acharya N, Jen KY, Osuntokun O (2012) Comparison of long-term (at least 24 weeks) weight gain and metabolic changes between adolescents and adults treated with olanzapine. J Child Adolesc Psychopharmacol 22:157–165
Kumra S, Kranzler H, Gerbino-Rosen G, Kester HM, De Thomas C, Kafantaris V, Correll CU, Kane JM (2008) Clozapine and “high-dose” olanzapine in refractory early-onset schizophrenia: a 12-week randomized and double-blind comparison. Biol Psychiatry 63:524–529
Lindsay RL, Leone S, Aman MG (2004) Discontinuation of risperidone and reversibility of weight gain in children with disruptive behavior disorders. Clin Pediatr (Phila) 43:437–444
Luby J, Mrakotsky C, Stalets MM, Belden A, Heffelfinger A, Williams M, Spitznagel E (2006) Risperidone in preschool children with autistic spectrum disorders: an investigation of safety and efficacy. J Child Adolesc Psychopharmacol 16:575–587
Maayan L, Correll CU (2011) Weight gain and metabolic risks associated with antipsychotic medications in children and adolescents. J Child Adolesc Psychopharmacol 21:517–535
Malhotra AK, Correll CU, Chowdhury NI, Muller DJ, Gregersen PK, Lee AT, Tiwari AK, Kane JM, Fleischhacker WW, Kahn RS, Ophoff RA, Meltzer HY, Lencz T, Kennedy JL (2012) Association between common variants near the melanocortin 4 receptor gene and severe antipsychotic drug-induced weight gain. Arch Gen Psychiatry 69:904–912
Marcus RN, Owen R, Manos G, Mankoski R, Kamen L, McQuade RD, Carson WH, Findling RL (2011) Safety and tolerability of aripiprazole for irritability in pediatric patients with autistic disorder: a 52-week, open-label, multicenter study. J Clin Psychiatry 72:1270–1276
Martin A, Koenig K, Scahill L, Bregman J (1999) Open-label quetiapine in the treatment of children and adolescents with autistic disorder. J Child Adolesc Psychopharmacol 9:99–107
Martin A, L’Ecuyer S (2002) Triglyceride, cholesterol and weight changes among risperidone-treated youths. A retrospective study. Eur Child Adolesc Psychiatry 11:129–133
Martin A, Landau J, Leebens P, Ulizio K, Cicchetti D, Scahill L, Leckman JF (2000) Risperidone-associated weight gain in children and adolescents: a retrospective chart review. J Child Adolesc Psychopharmacol 10:259–268
Martin A, Scahill L, Anderson GM, Aman M, Arnold LE, McCracken J, McDougle CJ, Tierney E, Chuang S, Vitiello B (2004) Weight and leptin changes among risperidone-treated youths with autism: 6-month prospective data. Am J Psychiatry 161:1125–1127
Martinez-Ortega JM, Diaz-Atienza F, Gutierrez-Rojas L, Jurado D, Gurpegui M (2011) Confounding by indication of a specific antipsychotic and the increase of body mass index among children and adolescents. Eur Child Adolesc Psychiatry 20:597–598
Masi G, Cosenza A, Mucci M, Brovedani P (2001) Open trial of risperidone in 24 young children with pervasive developmental disorders. J Am Acad Child Adolesc Psychiatry 40:1206–1214
Masi G, Milone A, Canepa G, Millepiedi S, Mucci M, Muratori F (2006) Olanzapine treatment in adolescents with severe conduct disorder. Eur Psychiatry 21:51–57
Masi G, Mucci M, Millepiedi S (2002) Clozapine in adolescent inpatients with acute mania. J Child Adolesc Psychopharmacol 12:93–99
McConville B, Carrero L, Sweitzer D, Potter L, Chaney R, Foster K, Sorter M, Friedman L, Browne K (2003) Long-term safety, tolerability, and clinical efficacy of quetiapine in adolescents: an open-label extension trial. J Child Adolesc Psychopharmacol 13:75–82
McCracken JT, McGough J, Shah B, Cronin P, Hong D, Aman MG, Arnold LE, Lindsay R, Nash P, Hollway J, McDougle CJ, Posey D, Swiezy N, Kohn A, Scahill L, Martin A, Koenig K, Volkmar F, Carroll D, Lancor A, Tierney E, Ghuman J, Gonzalez NM, Grados M, Vitiello B, Ritz L, Davies M, Robinson J, McMahon D (2002) Risperidone in children with autism and serious behavioral problems. N Engl J Med 347:314–321
McCracken JT, Suddath R, Chang S, Thakur S, Piacentini J (2008) Effectiveness and tolerability of open label olanzapine in children and adolescents with Tourette syndrome. J Child Adolesc Psychopharmacol 18:501–508
McDougle CJ, Holmes JP, Bronson MR, Anderson GM, Volkmar FR, Price LH, Cohen DJ (1997) Risperidone treatment of children and adolescents with pervasive developmental disorders: a prospective open-label study. J Am Acad Child Adolesc Psychiatry 36:685–693
McIntyre RS, Jerrell JM (2008) Metabolic and cardiovascular adverse events associated with antipsychotic treatment in children and adolescents. Arch Pediatr Adolesc Med 162:929–935
Moreno C, Merchan-Naranjo J, Alvarez M, Baeza I, Alda JA, Martinez-Cantarero C, Parellada M, Sanchez B, de la Serna E, Giraldez M, Arango C (2010) Metabolic effects of second-generation antipsychotics in bipolar youth: comparison with other psychotic and nonpsychotic diagnoses. Bipolar Disord 12:172–184
Mozes T, Ebert T, Michal SE, Spivak B, Weizman A (2006) An open-label randomized comparison of olanzapine versus risperidone in the treatment of childhood-onset schizophrenia. J Child Adolesc Psychopharmacol 16:393–403
Mozes T, Greenberg Y, Spivak B, Tyano S, Weizman A, Mester R (2003) Olanzapine treatment in chronic drug-resistant childhood-onset schizophrenia: an open-label study. J Child Adolesc Psychopharmacol 13:311–317
Mukaddes NM, Abali O, Gurkan K (2004) Short-term efficacy and safety of risperidone in young children with autistic disorder (AD). World J Biol Psychiatry 5:211–214
Nagaraj R, Singhi P, Malhi P (2006) Risperidone in children with autism: randomized, placebo-controlled, double-blind study. J Child Neurol 21:450–455
Nicolson R, Awad G, Sloman L (1998) An open trial of risperidone in young autistic children. J Am Acad Child Adolesc Psychiatry 37:372–376
Owen R, Sikich L, Marcus RN, Corey-Lisle P, Manos G, McQuade RD, Carson WH, Findling RL (2009) Aripiprazole in the treatment of irritability in children and adolescents with autistic disorder. Pediatrics 124:1533–1540
Panagiotopoulos C, Ronsley R, Davidson J (2009) Increased prevalence of obesity and glucose intolerance in youth treated with second-generation antipsychotic medications. Can J Psychiatry 54:743–749
Patel NC, Hariparsad M, Matias-Akthar M, Sorter MT, Barzman DH, Morrison JA, Stanford KE, Strakowski SM, DelBello MP (2007) Body mass indexes and lipid profiles in hospitalized children and adolescents exposed to atypical antipsychotics. J Child Adolesc Psychopharmacol 17:303–311
Patel NC, Kistler JS, James EB, Crismon ML (2004) A retrospective analysis of the short-term effects of olanzapine and quetiapine on weight and body mass index in children and adolescents. Pharmacotherapy 24:824–830
Pringsheim T, Lam D, Ching H, Patten S (2011) Metabolic and neurological complications of second-generation antipsychotic use in children: a systematic review and meta-analysis of randomized controlled trials. Drug Saf 34:651–668
Pulkki-Raback L, Elovainio M, Kivimaki M, Mattsson N, Raitakari OT, Puttonen S, Marniemi J, Viikari JS, Keltikangas-Jarvinen L (2009) Depressive symptoms and the metabolic syndrome in childhood and adulthood: a prospective cohort study. Health Psychol 28:108–116
Quintana H, Wilson MS 2nd, Purnell W, Layman AK, Mercante D (2007) An open-label study of olanzapine in children and adolescents with schizophrenia. J Psychiatr Pract 13:86–96
Ratzoni G, Gothelf D, Brand-Gothelf A, Reidman J, Kikinzon L, Gal G, Phillip M, Apter A, Weizman R (2002) Weight gain associated with olanzapine and risperidone in adolescent patients: a comparative prospective study. J Am Acad Child Adolesc Psychiatry 41:337–343
Reyes M, Olah R, Csaba K, Augustyns I, Eerdekens M (2006) Long-term safety and efficacy of risperidone in children with disruptive behaviour disorders. Results of a 2-year extension study. Eur Child Adolesc Psychiatry 15:97–104
Reynolds GP, Zhang ZJ, Zhang XB (2002) Association of antipsychotic drug-induced weight gain with a 5-HT2C receptor gene polymorphism. Lancet 359:2086–2087
Robb AS, Andersson C, Bellocchio EE, Manos G, Rojas-Fernandez C, Mathew S, Marcus R, Owen R, Mankoski R (2011) Safety and tolerability of aripiprazole in the treatment of irritability associated with autistic disorder in pediatric subjects (6-17 years old): results from a pooled analysis of 2 studies. Prim Care Companion CNS Disord 13
Ross RG, Novins D, Farley GK, Adler LE (2003) A 1-year open-label trial of olanzapine in school-age children with schizophrenia. J Child Adolesc Psychopharmacol 13:301–309
Safer DJ (2004) A comparison of risperidone-induced weight gain across the age span. J Clin Psychopharmacol 24:429–436
Saklad SR, Ketchi CM, Amrung SA (2002) Gender differences in weight gain among adolescents started on atypical antipsychotics. J Child Adolesc Psychopharmacol 12:288–289
Sallee FR, Kurlan R, Goetz CG, Singer H, Scahill L, Law G, Dittman VM, Chappell PB (2000) Ziprasidone treatment of children and adolescents with Tourette’s syndrome: a pilot study. J Am Acad Child Adolesc Psychiatry 39:292–299
Schimmelmann BG, Mehler-Wex C, Lambert M, Schulze-zur-Wiesch C, Koch E, Flechtner HH, Gierow B, Maier J, Meyer E, Schulte-Markwort M (2007) A prospective 12-week study of quetiapine in adolescents with schizophrenia spectrum disorders. J Child Adolesc Psychopharmacol 17:768–778
Shaw JA, Lewis JE, Pascal S, Sharma RK, Rodriguez RA, Guillen R, Pupo-Guillen M (2001) A study of quetiapine: efficacy and tolerability in psychotic adolescents. J Child Adolesc Psychopharmacol 11:415–424
Shaw P, Sporn A, Gogtay N, Overman GP, Greenstein D, Gochman P, Tossell JW, Lenane M, Rapoport JL (2006) Childhood-onset schizophrenia: a double-blind, randomized clozapine-olanzapine comparison. Arch Gen Psychiatry 63:721–730
Shea S, Turgay A, Carroll A, Schulz M, Orlik H, Smith I, Dunbar F (2004) Risperidone in the treatment of disruptive behavioral symptoms in children with autistic and other pervasive developmental disorders. Pediatrics 114:e634–e641
Sholevar EH, Baron DA, Hardie TL (2000) Treatment of childhood-onset schizophrenia with olanzapine. J Child Adolesc Psychopharmacol 10:69–78
Sikich L, Frazier JA, McClellan J, Findling RL, Vitiello B, Ritz L, Ambler D, Puglia M, Maloney AE, Michael E, De Jong S, Slifka K, Noyes N, Hlastala S, Pierson L, McNamara NK, Delporto-Bedoya D, Anderson R, Hamer RM, Lieberman JA (2008) Double-blind comparison of first- and second-generation antipsychotics in early-onset schizophrenia and schizo-affective disorder: findings from the treatment of early-onset schizophrenia spectrum disorders (TEOSS) study. Am J Psychiatry 165:1420–1431
Sikich L, Hamer RM, Bashford RA, Sheitman BB, Lieberman JA (2004) A pilot study of risperidone, olanzapine, and haloperidol in psychotic youth: a double-blind, randomized, 8-week trial. Neuropsychopharmacology 29:133–145
Snyder R, Turgay A, Aman M, Binder C, Fisman S, Carroll A (2002) Effects of risperidone on conduct and disruptive behavior disorders in children with subaverage IQs. J Am Acad Child Adolesc Psychiatry 41:1026–1036
Swadi HS, Craig BJ, Pirwani NZ, Black VC, Buchan JC, Bobier CM (2010) A trial of quetiapine compared with risperidone in the treatment of first onset psychosis among 15- to 18-year-old adolescents. Int Clin Psychopharmacol 25:1–6
Szigethy E, Wiznitzer M, Branicky LA, Maxwell K, Findling RL (1999) Risperidone-induced hepatotoxicity in children and adolescents? A chart review study. J Child Adolesc Psychopharmacol 9:93–98
Theisen FM, Gebhardt S, Haberhausen M, Heinzel-Gutenbrunner M, Wehmeier PM, Krieg JC, Kuhnau W, Schmidtke J, Remschmidt H, Hebebrand J (2005) Clozapine-induced weight gain: a study in monozygotic twins and same-sex sib pairs. Psychiatr Genet 15:285–289
Tramontina S, Zeni CP, Ketzer CR, Pheula GF, Narvaez J, Rohde LA (2009) Aripiprazole in children and adolescents with bipolar disorder comorbid with attention-deficit/hyperactivity disorder: a pilot randomized clinical trial. J Clin Psychiatry 70:756–764
Turgay A, Binder C, Snyder R, Fisman S (2002) Long-term safety and efficacy of risperidone for the treatment of disruptive behavior disorders in children with subaverage IQs. Pediatrics 110:e34
Van Bellinghen M, De Troch C (2001) Risperidone in the treatment of behavioral disturbances in children and adolescents with borderline intellectual functioning: a double-blind, placebo-controlled pilot trial. J Child Adolesc Psychopharmacol 11:5–13
Wozniak J, Mick E, Waxmonsky J, Kotarski M, Hantsoo L, Biederman J (2009) Comparison of open-label, 8-week trials of olanzapine monotherapy and topiramate augmentation of olanzapine for the treatment of pediatric bipolar disorder. J Child Adolesc Psychopharmacol 19:539–545
Zuddas A, Di Martino A, Muglia P, Cianchetti C (2000) Long-term risperidone for pervasive developmental disorder: efficacy, tolerability, and discontinuation. J Child Adolesc Psychopharmacol 10:79–90
Conflict of interest
The authors report no biomedical financial interests or potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martínez-Ortega, J.M., Funes-Godoy, S., Díaz-Atienza, F. et al. Weight gain and increase of body mass index among children and adolescents treated with antipsychotics: a critical review. Eur Child Adolesc Psychiatry 22, 457–479 (2013). https://doi.org/10.1007/s00787-013-0399-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-013-0399-5