Abstract
Objectives
The aim of this study was to investigate whether variation in pH of sodium hypochlorite (NaOCl) increased its antibacterial and dissolution ability on polymicrobial biofilms formed in situ.
Materials and methods
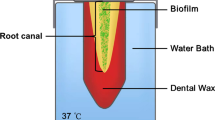
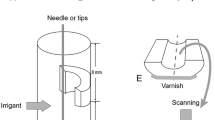
Fifty-six dentin blocks (eight/group) were intraorally infected for 48 h and incubated in BHI for 48 h to standardize the biofilm growth. The specimens were irrigated with 1 and 2.5 % NaOCl with pH levels of 5, 7, and 12 for 20 min. The control group was irrigated with distilled water. The cell viability and the bacterial volume were measured at the pre- and post-irrigation procedures. Five random areas of each sample were chosen and analyzed with confocal laser scanning microscopy (CLSM). Statistical analysis was performed using the non-parametric Kruskal-Wallis and Dunn’s tests (p < 0.05).
Results
All the experimental solutions were able to decrease the biomass (p < 0.05) except for the 1 % NaOCl-pH 5 group. The antibacterial ability of the NaOCl was dependent on the concentration and acidification of the solution.
Conclusions
The acidification of NaOCl improves its antibacterial ability, but the dissolution effect of the irrigant is decreased.
Clinical relevance
Bacteria and their products are the main factors in development of apical periodontitis. The pH reduction in the NaOCl could enhance the reduction or elimination of the root canal bacterial colonies in comparison with the unaltered solution.


Similar content being viewed by others
References
Costerton JW, Stewart PS, Greenberg EP (1999) Bacterial biofilms: a common cause of persistent infections. Science 284:1318–1322
Mah TF, O'Toole GA (2001) Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol 9:34–39
Siqueira JF Jr, Paiva SS, Rocas IN (2007) Reduction in the cultivable bacterial populations in infected root canals by a chlorhexidine-based antimicrobial protocol. J Endod 33:541–547
Clegg MS, Vertucci FJ, Walker C, Belanger M, Britto LR (2006) The effect of exposure to irrigant solutions on apical dentin biofilms in vitro. J Endod 32:434–437
Del Carpio-Perochena AE, Bramante CM, Duarte MA, Cavenago BC, Villas-Boas MH, Graeff MS, Bernardineli N, de Andrade FB, Ordinola-Zapata R (2011) Biofilm dissolution and cleaning ability of different irrigant solutions on intraorally infected dentin. J Endod 37:1134–1138
Siqueira JF Jr, Machado AG, Silveira RM, Lopes HP, de Uzeda M (1997) Evaluation of the effectiveness of sodium hypochlorite used with three irrigation methods in the elimination of Enterococcus faecalis from the root canal, in vitro. Int Endod J 30:279–282
Estrela C, Estrela CR, Barbin EL, Spano JC, Marchesan MA, Pecora JD (2002) Mechanism of action of sodium hypochlorite. Braz Dent J 13:113–117
Sirtes G, Waltimo T, Schaetzle M, Zehnder M (2005) The effects of temperature on sodium hypochlorite short-term stability, pulp dissolution capacity, and antimicrobial efficacy. J Endod 31:669–671
Christensen CE, McNeal SF, Eleazer P (2008) Effect of lowering the pH of sodium hypochlorite on dissolving tissue in vitro. J Endod 34:449–452
Camps J, Pommel L, Aubut V et al (2009) Shelf life, dissolving action, and antibacterial activity of a neutralized 2.5% sodium hypochlorite solution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 108:e66–73
Aubut V, Pommel L, Verhille B, Orsiere T, Garcia S, About I, Camps J (2010) Biological properties of a neutralized 2.5% sodium hypochlorite solution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 109:e120–125
Mercade M, Duran-Sindreu F, Kuttler S, Roig M, Durany N (2009) Antimicrobial efficacy of 4.2% sodium hypochlorite adjusted to pH 12, 7.5, and 6.5 in infected human root canals. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 107:295–298
Hugo WB, Russell AD (2007) Pharmaceutical microbiology. Blackwell Science, Massachusetts
Bloomfield SF, Miles GA (1979) Antibacterial properties of sodium dichloroisocyanurate and sodium-hypochlorite formulations. J Appl Bacteriol 46:65–73
Guerreiro-Tanomaru JM, Morgental RD, Flumignan DL, Gasparini F, Oliveira JE, Tanomaru-Filho M (2011) Evaluation of pH, available chlorine content, and antibacterial activity of endodontic irrigants and their combinations against Enterococcus faecalis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 112:132–135
Chavez de Paz LE (2009) Image analysis software based on color segmentation for characterization of viability and physiological activity of biofilms. Appl Environ Microbiol 75:1734–1739
Heydorn A, Nielsen AT, Hentzer M, Sternberg C, Givskov M, Ersboll BK, Molin S (2000) Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 146:2395–2407
Moller S, Korber DR, Wolfaardt GM, Molin S, Caldwell DE (1997) Impact of nutrient composition on a degradative biofilm community. Appl Environ Microbiol 63:2432–2438
Nielsen AT, Tolker-Nielsen T, Barken KB, Molin S (2000) Role of commensal relationships on the spatial structure of a surface-attached microbial consortium. Environ Microbiol 2:59–68
Yang X, Beyenal H, Harkin G, Lewandowski Z (2000) Quantifying biofilm structure using image analysis. J Microbiol Methods 39:109–119
Xavier JB, Schnell A, Wuertz S, Palmer R, White DC, Almeida JS (2001) Objective threshold selection procedure (OTS) for segmentation of scanning laser confocal microscope images. J Microbiol Methods 47:169–180
Ordinola-Zapata R, Bramante CM, Brandao Garcia R, Bombarda de Andrade F, Bernardineli N, Gomes de Moraes I, Duarte MA (2012) The antimicrobial effect of new and conventional endodontic irrigants on intra-orally infected dentin. Acta Odontol Scand 71:424–431
Chavez de Paz LE, Bergenholtz G, Svensater G (2010) The effects of antimicrobials on endodontic biofilm bacteria. J Endod 36:70–77
Andersen M, Lund A, Andreasen JO, Andreasen FM (1992) In vitro solubility of human pulp tissue in calcium hydroxide and sodium hypochlorite. Endod Dent Traumatol 8:104–108
Oliveira DP, Barbizam JV, Trope M, Teixeira FB (2007) In vitro antibacterial efficacy of endodontic irrigants against Enterococcus faecalis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 103:702–706
Chavez de Paz LE (2007) Redefining the persistent infection in root canals: possible role of biofilm communities. J Endod 33:652–662
Elias S, Banin E (2012) Multi-species biofilms: living with friendly neighbors. FEMS Microbiol Rev 36:990–1004
Gomes BP, Ferraz CC, Vianna ME, Berber VB, Teixeira FB, Souza-Filho FJ (2001) In vitro antimicrobial activity of several concentrations of sodium hypochlorite and chlorhexidine gluconate in the elimination of Enterococcus faecalis. Int Endod J 34:424–428
Bystrom A, Sundqvist G (1985) The antibacterial action of sodium hypochlorite and EDTA in 60 cases of endodontic therapy. Int Endod J 18:35–40
Radcliffe CE, Potouridou L, Qureshi R, Habahbeh N, Qualtrough A, Worthington H, Drucker DB (2004) Antimicrobial activity of varying concentrations of sodium hypochlorite on the endodontic microorganisms Actinomyces israelii, A. naeslundii, Candida albicans and Enterococcus faecalis. Int Endod J 37:438–446
Haapasalo M, Shen Y, Qian W, Gao Y (2010) Irrigation in endodontics. Dent Clin North Am 54:291–312
Zehnder M, Schmidlin P, Sener B, Waltimo T (2005) Chelation in root canal therapy reconsidered. J Endod 31:817–820
Baumgartner JC, Ibay AC (1987) The chemical reactions of irrigants used for root canal debridement. J Endod 13:47–51
Hand RE, Smith ML, Harrison JW (1978) Analysis of the effect of dilution on the necrotic tissue dissolution property of sodium hypochlorite. J Endod 4:60–64
Moorer WR, Wesselink PR (1982) Factors promoting the tissue dissolving capability of sodium hypochlorite. Int Endod J 15:187–196
Harrison JW, Hand RE (1981) The effect of dilution and organic matter on the anti-bacterial property of 5.25% sodium hypochlorite. J Endod 7:128–132
Zehnder M, Kosicki D, Luder H, Sener B, Waltimo T (2002) Tissue-dissolving capacity and antibacterial effect of buffered and unbuffered hypochlorite solutions. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 94:756–762
Acknowledgments
This study was supported by the São Paulo Research Foundation (FAPESP 2011/08184-8) and the National Council for Scientific and Technological Development (CNPq 135419/2011-5).
Conflict of interest
The authors deny any conflicts of interest related to this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
del Carpio-Perochena, A., Bramante, C.M., de Andrade, F.B. et al. Antibacterial and dissolution ability of sodium hypochlorite in different pHs on multi-species biofilms. Clin Oral Invest 19, 2067–2073 (2015). https://doi.org/10.1007/s00784-015-1431-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1431-6