Abstract
Objectives
The objective of this study was to evaluate the cytotoxic reaction of transfected human pulp derived cells (tHPDC) and transfected bovine pulp derived cells (tBPDC) after exposure to resin cements [RelyX UnicemClicker (RX), MaxCem (MC), Panavia F 2.0 (PF), BisCem (BC), and Bistite II DC (BII)] and to compare it to the generation of reactive oxygen species (ROS).
Materials and methods
Set materials were extracted in culture medium, cell survival as a measure of cytotoxicity was determined photometrically using crystal violet after cells were exposed to the extracts for 24 h. The generation of ROS was detected by flow cytometry after cells were exposed to extract dilutions for 1 h.
Results
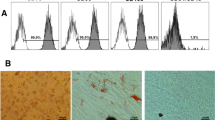
The ranking of the least to the most cytotoxic material was: RX < BII < PF < BC < MC for both cell lines, but for tHPDC, only MC and PF eluates were different from untreated controls. Generally, tBPDC were more susceptible to materials than tHPDC, but only for RX and BC was this difference statistically significant. All undiluted extracts increased ROS production in both cell lines but to a higher amount in tHPDC than in tBPDC.
Conclusions
tHPDC reacted less sensitive than tBPDC in the cytotoxicity test but with the same rank order of materials. In contrast, the cellular oxidative stress reaction was more pronounced in tHPDC than in tBPDC.
Clinical relevance
Depending on the residual dentine layer in deep cavities, biologically active resin monomers or additives released from resin cements may influence the dentine–pulp complex, for instance, its regenerative and reparative capacities.


Similar content being viewed by others
References
Burke FJ, Watts DC (1994) Fracture resistance of teeth restored with dentin-bonded crowns. Quintessence Int 25:335–340
Hikita K, Van Meerbeek B, De Munck J, Ikeda T, Van Landuyt K, Maida T et al (2007) Bonding effectiveness of adhesive luting agents to enamel and dentin. Dent Mater 23:71–80
Abo-Hamar SE, Hiller KA, Jung H, Federlin M, Friedl KH, Schmalz G (2005) Bond strength of a new universal self-adhesive resin luting cement to dentin and enamel. Clin Oral Investig 9:161–167
Schmid-Schwap M, Franz A, König F, Bristela M, Lucas T, Piehslinger E et al (2009) Cytotoxicity of four categories of dental cements. Dent Mater 25:360–368
Bakopoulou A, Mourelatos D, Tsiftsoglou AS, Giassin NP, Mioglou E, Garefis P (2009) Genotoxic and cytotoxic effects of different types of dental cement on normal cultured human lymphocytes. Mutat Res 672:103–112
Kong N, Jiang T, Zhou Z, Fu J (2009) Cytotoxicity of polymerized resin cements on human dental pulp cells in vitro. Dent Mater 25:1371–1375
Thonemann B, Schmalz G (2000) Immortalization of bovine dental papilla cells with simian virus 40 large T antigen. Arch Oral Biol 45:857–869
Thonemann B, Schmalz G (2000) Bovine dental papilla-derived cells immortalized with HPV 18 E6/E7. Eur J Oral Sci 108:432–441
Galler KM, Schweikl H, Thonemann B, D'Souza RN, Schmalz G (2006) Human pulp-derived cells immortalized with Simian Virus 40 T-antigen. Eur J Oral Sci 114:138–146
Schweikl H, Spagnuolo G, Schmalz G (2006) Genetic and cellular toxicology of dental resin monomers. J Dent Res 85:870–877
Chang HH, Guo MK, Kasten FH, Chang MC, Huang GF, Wang YL et al (2005) Stimulation of glutathione depletion, ROS production and cell cycle arrest of dental pulp cells and gingival epithelial cells by HEMA. Biomaterials 26:745–753
Schweikl H, Hartmann A, Hiller KA, Spagnuolo G, Bolay C, Brockhoff G et al (2007) Inhibition of TEGDMA and HEMA-induced genotoxicity and cell cycle arrest by N-acetylcysteine. Dent Mater 23:688–695
Eckhardt A, Müller P, Hiller KA, Krifka S, Bolay C, Spagnuolo G et al (2010) Influence of TEGDMA on the mammalian cell cycle in comparison with chemotherapeutic agents. Dent Mater 26:232–241
Galler KM, Schweikl H, Hiller KA, Cavender AC, Bolay C, D'Souza RN, et al. (2010) TEGDMA Reduces Mineralization in Dental Pulp Cells. J Dent Res in press, doi: 10.1177/0022034510384618
ISO 10993-12:2007. Biological evaluation of medical devices—part 12: sample preparation and reference materials. International Organization for Standardization, Case postale 56, CH-1211 Geneva 20, Switzerland
Gilles RJ, Didier N, Denton M (1986) Determination of cell number in monolayer cultures. Anal Biochem 159:109–113
Myhre O, Andersen JM, Aernes H, Fonnum F (2003) Evaluation of the probes 2′,7′-dichlorofluorescin diacetate, luminol, and lucigenin as indicators of reactive species formation. Biochem Pharmacol 65:1575–1582
Thonemann B, Schmalz G, Hiller KA, Schweikl H (2002) Responses of L929 mouse fibroblasts, primary and immortalized bovine dental papilla-derived cell lines to dental resin components. Dent Mater 18:318–323
McCarthy TJ, Hayes EP, Schwartz CS, Witz G (1994) The reactivity of selected acrylate esters toward glutathione and deoxyribonucleosides in vitro: structure-activity relationships. Fundam Appl Toxicol 22:543–548
Nocca G, D'Antò V, Desiderio C, Rossetti DV, Valletta R, Baquala AM et al (2010) N-acetyl cysteine directed detoxification of 2-hydroxyethyl methacrylate by adduct formation. Biomaterials 31:2508–2516
Walther UI, Siagian II, Walther SC, Reichl FX, Hickel R (2004) Antioxidative vitamins decrease cytotoxicity of HEMA and TEGDMA in cultured cell lines. Arch Oral Biol 49:125–131
Gerzina TM, Hume WR (1996) Diffusion of monomers from bonding resin-resin composite combinations through dentine in vitro. J Dent 24:125–128
Altintas SH, Usumez A (2008) Evaluation of monomer leaching from a dual cured resin cement. Biomed Mater Res B Appl Biomater 86B:523–529
Ulker HE, Sengun A (2009) Cytotoxicity evaluation of self adhesive composite resin cements by dentin barrier test on 3D pulp cells. Eur J Dent 3:120–126
Ratanasathien S, Wataha JC, Hanks CT, Dennison JB (1995) Cytotoxic interactive effects of dentin bonding components on mouse fibroblasts. J Dent Res 74:1602–1606
Geurtsen W, Lehmann F, Spahl W, Leyhausen G (1998) Cytotoxicity of 35 dental resin composite monomers/additives in permanent 3 T3 and three human primary fibroblast cultures. J Biomed Mater Res 41:474–480
de Mendonça AA, Souza PP, Costa CA (2007) Cytotoxic effects of hard-setting cements applied on the odontoblast cell line MDPC-23. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 104:102–108
de Souza Costa CA, Hebling J, Randall RC (2006) Human pulp response to resin cements used to bond inlay restorations. Dent Mater 22:954–962
Camargo SE, Camargo CH, Hiller KA, Rode SM, Schweikl H, Schmalz G (2009) Cytotoxicity and genotoxicity of pulp capping materials in two cell lines. Int Endod J 42:227–237
Camargo CH, Camargo SE, Valera MC, Hiller KA, Schmalz G, Schweikl H (2009) The induction of cytotoxicity, oxidative stress, and genotoxicity by root canal sealers in mammalian cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 108:952–960
Acknowledgements
This study was based on the work performed by H. Esra Ulker for the fulfillment of the degree of Doctor of Philosophy, Selcuk University, Turkey. This study was supported by the Department of Operative Dentistry and Periodontology, University of Regensburg, Germany; the Scientific and Technological Research Council of Turkey (TUBITAK); and Selcuk University BAP (06102038) and IADR (John Gray Fellowship—2007).
The authors gratefully acknowledge the skillful technical assistance of Carola Bolay (Department of Operative Dentistry and Periodontology, University of Regensburg) and special thanks to Dr. Mustafa Ulker (Department of Operative Dentistry, Selcuk University, Konya, Turkey) for his valuable contributions.
Conflict of interest
The authors do not have any conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ülker, H.E., Hiller, K.A., Schweikl, H. et al. Human and bovine pulp-derived cell reactions to dental resin cements. Clin Oral Invest 16, 1571–1578 (2012). https://doi.org/10.1007/s00784-011-0657-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-011-0657-1