Abstract
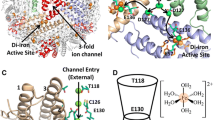
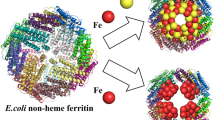
Ferritin molecules contain 24 polypeptide chains folded as four-helix bundles and arranged as a hollow shell capable of storing up to 4500 Fe(III) atoms. H chains contain ferroxidase centres which lie within the bundle, about 12 Å (1.2 nm) from the outside surface and 8 Å from the inner surface of the protein shell. Catalysis of Fe(II) oxidation precedes storage of Fe(III) as ferrihydrite, with the formation of μ-oxo-bridged Fe(III) dimers as intermediates. Factors influencing the movement of μ-oxo-bridged Fe(III) from the ferroxidase centre to the ferritin cavity are uncertain. Assistance by small chelators is one possibility. The aim of this investigation was to determine whether iron at the dinuclear centres of three ferritins (human H chain homopolymer, HuHF, the non-haem ferritin of Escherichia coli, EcFTN, and horse spleen ferritin, HoSF) is accessible to chelators. Forty-eight Fe(II) atoms/molecule were added to the apoferritins followed, 2 min later, by the addition of chelator (1,10-phenanthroline, 2,2-bipyridine, desferrioxamine or 3,4-dihydroxybenzaldehyde). Iron species were analysed by Mössbauer spectroscopy or visible absorbance. Competition between chelators and apoferritin for Fe(II) was also investigated. The main conclusions of the study are that: (1) dinuclear iron and iron in small iron-cores in HuHF and EcFTN is mobilisable by all four chelators; (2) the chelators penetrate the shell; (3) 3,4-dihydroxybenzaldehyde is the most efficient in mobilising Fe(III) but the least successful in competing for Fe(II); (4) Fe(III) is more readily released from EcFTN than from HuHF; (5) 2,2′-bipyridine aids the movement of Fe(III) from ferroxidase centre to core.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Treffry, A., Hawkins, C., Williams, J. et al. Lability of iron at the dinuclear centres of ferritin studied by competition with four chelators. JBIC 1, 49–60 (1996). https://doi.org/10.1007/s007750050022
Issue Date:
DOI: https://doi.org/10.1007/s007750050022