Abstract
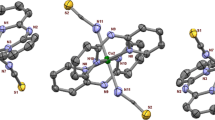
The catechol oxidase activity of three copper/bicompartmental salen derivatives has been studied. One mononuclear, [CuL] (1), one homometallic, [Cu2L(NO3)2] (2), and one heterometallic, [CuMnL(NO3)2] (3) complexes were obtained using the ligand H2L = N,N′-bis(3-methoxysalicylidene)-1,3-propanediamine through different synthetic methods (electrochemical, chemical and solid state reaction). The structural data indicate that the metal ion disposition models the active site of type-3 copper enzymes, such as catechol oxidase. In this way, their ability to act as functional models of the enzyme has been spectrophotometrically determined by monitorization of the oxidation of 3,5-di-tert-butylcatechol (3,5-DTBC) to 3,5-di-tert-butyl-o-benzoquinone (3,5-DTBQ). All the complexes show significant catalytic activity with ratio constants (kobs) lying in the range (223–294) × 10–4 min−1. A thorough kinetic study was carried out for complexes 2 and 3, since they show structural similarities with the catechol oxidase enzyme. The greatest catalytic activity was found for the homonuclear dicopper compound (2) with a turnover value (kcat) of (3.89 ± 0.05) × 106 h−1, which it is the higher reported to date, comparable to the enzyme itself (8.25 × 106 h−1).
Graphic abstract












Similar content being viewed by others
References
Sureshbabu P, Junaid QM, Upadhyay C et al (2019) Di and tetranuclear Cu(II) complexes with simple 2-aminoethylpyridine: magnetic properties, phosphodiester hydrolysis, DNA binding/cleavage, cytotoxicity and catecholase activity. Polyhedron 164:202–218. https://doi.org/10.1016/j.poly.2019.02.015
Garcia-Bosch I, Karlin KD (2014) Copper peroxide bioinorganic chemistry: from metalloenzymes to bioinspired synthetic systems. In: Rappoport Z (ed) PATAI’S chemistry of functional groups. Wiley, Chichester, 1–52. https://doi.org/10.1002/9780470682531.pat0863
Neves A, Rossi LM, Bortoluzzi AJ et al (2002) Catecholase activity of a series of dicopper(II) complexes with variable Cu−OH(phenol) moieties. Inorg Chem 41:1788–1794. https://doi.org/10.1021/ic010708u
Sathya V, Murali M (2018) Functional mimics of type-2 and type-3 copper oxidases: self-assembled molecular association in mononuclear copper(II) complex enhances the catalytic activity. Inorg Chem Commun 92:55–59. https://doi.org/10.1016/j.inoche.2018.04.003
Banu KS, Chattopadhyay T, Banerjee A et al (2009) Mono- and dinuclear manganese(III) complexes showing efficient catechol oxidase activity: syntheses, characterization and spectroscopic studies. Dalton Trans. https://doi.org/10.1039/b902498k
Klabunde T, Eicken C, Sacchettini JC, Krebs B (1998) Crystal structure of a plant catechol oxidase containing a dicopper center. Nat Struct Biol 5:1084–1090. https://doi.org/10.1038/4193
Dey SK, Mukherjee A (2016) Catechol oxidase and phenoxazinone synthase: biomimetic functional models and mechanistic studies. Coord Chem Rev 310:80–115. https://doi.org/10.1016/j.ccr.2015.11.002
Abu-Dief AM, Mohamed IMA (2015) A review on versatile applications of transition metal complexes incorporating Schiff bases. Beni Suef Univ J Basic Appl Sci 4:119–133. https://doi.org/10.1016/j.bjbas.2015.05.004
Madalan AM, Ene CD (2018) Supramolecular rectangles and ladders constructed from Ni(II), Cu(II) and Zn(II) mononuclear complexes with bicompartmental ligands and 4-aminopyridine as tectons. Inorg Chim Acta 475:184–192. https://doi.org/10.1016/j.ica.2017.06.058
Cozzi PG (2004) Metal–Salen Schiff base complexes in catalysis: practical aspects. Chem Soc Rev 33:410–421. https://doi.org/10.1039/B307853C
Finelli A, Hérault N, Crochet A, Fromm KM (2018) Threading Salen-type Cu- and Ni-complexes into one-dimensional coordination polymers: solution versus solid state and the size effect of the alkali metal ion. Cryst Growth Des 18:1215–1226. https://doi.org/10.1021/acs.cgd.7b01769
Eicken C, Zippel F, Büldt-Karentzopoulos K, Krebs B (1998) Biochemical and spectroscopic characterization of catechol oxidase from sweet potatoes (Ipomoea batatas) containing a type-3 dicopper center 1. FEBS Lett 436:293–299. https://doi.org/10.1016/S0014-5793(98)01113-2
Rompel A, Fischer H, Meiwes D et al (1999) Substrate specificity of catechol oxidase from Lycopus europaeus and characterization of the bioproducts of enzymic caffeic acid oxidation 1. FEBS Lett 445:103–110. https://doi.org/10.1016/S0014-5793(99)00106-4
Thakurta S, Chakraborty J, Rosair G et al (2009) The interplay of O–H⋯O hydrogen bonding in the generation of three new supramolecular complexes of CuII, NiII and CoIII: Syntheses, characterization and structural aspects. Inorg Chim Acta 362:2828–2836. https://doi.org/10.1016/j.ica.2009.01.002
Gomes L, Sousa C, Freire C, de Castro B (2000) Diaqua{6,6′-dimethoxy-2,2′-[propane-1,3-diylbis(nitrilomethylidyne-N)]diphenolato-O, O′}nickel(II). Acta Crystallogr Sect C Cryst Struct Commun 56:1201–1203. https://doi.org/10.1107/S010827010001009X
Thakurta S, Butcher RJ, Gómez-García CJ et al (2010) Synthesis, structural aspects and magnetic properties of an unusual 2D thiocyanato-bridged cobalt(II)–Schiff base network. Inorg Chim Acta 363:3981–3986. https://doi.org/10.1016/j.ica.2010.07.069
Basak T, Ghosh K, Gómez-García CJ, Chattopadhyay S (2018) Synthesis, structure and magnetic characterization of a dinuclear and two mononuclear iron(III) complexes with N, O-donor Schiff base ligands. Polyhedron 146:42–54. https://doi.org/10.1016/j.poly.2017.12.040
Banerjee S, Saha A (2016) A new end-on (μ-1,1) azido bridged [Zn2(L)2(Na)N3] n 1D chain derived from a trinuclear zinc complex: syntheses, crystal structures, photoluminescence properties and DFT study. J Coord Chem 69:3092–3106. https://doi.org/10.1080/00958972.2016.1226502
Banerjee S, Ghorai P, Sarkar P et al (2020) A rare flattened tetrahedral Mn(II) salen type complex: synthesis, crystal structure, biomimetic catalysis and DFT study. Inorg Chim Acta 499:119176. https://doi.org/10.1016/j.ica.2019.119176
Biswas D, Chakrabarty PP, Saha S et al (2013) Ligand mediated structural diversity and role of different weak interactions in molecular self-assembly of a series of copper(II)–sodium(I) Schiff-base heterometallic complexes. Inorg Chim Acta 408:172–180. https://doi.org/10.1016/j.ica.2013.09.011
Chiboub Fellah FZ, Costes J-P, Dahan F et al (2007) Varying the metal/metal ratio in related Cu–Ca complexes. Polyhedron 26:4209–4215. https://doi.org/10.1016/j.poly.2007.05.019
Branzea DG, Madalan AM, Ciattini S et al (2010) New heterometallic coordination polymers constructed from 3d–3d′ binuclear nodes. New J Chem 34:2479. https://doi.org/10.1039/c0nj00238k
Cai X, Ning H (2015) Crystal structure of di-μ-acetato-diacetatobis(μ-6,6′-dimethoxy-2,2′-{[(propane-1,3-diylbis(azanylylidene)]bis(methanylylidene)}diphenolato)tetrazinc. Acta Crystallogr Sect E Crystallogr Commun 71:m217–m218. https://doi.org/10.1107/S2056989015020551
Wang J-H, Yan P-F, Li G-M et al (2010) N,N′-Bis(2-hydroxy-3-methoxybenzylidene)-1,3-diaminopropane dimeric 4f and 3d–4f heterodinuclear complexes: syntheses, crystal structures and magnetic properties. Inorg Chim Acta 363:3706–3713. https://doi.org/10.1016/j.ica.2010.05.030
Lee JH, Im SY, Lee SW (2018) Pd–Ln and Pt–Ln complexes of a bi-compartmental ligand: [MLn(L)(NO3)3] (M = Pd, Pt; Ln= Eu, Tb; H2L= N,N′-bis(3-methoxysalicylidenimino-1,3-diaminopropane)). Inorg Chim Acta 474:89–95. https://doi.org/10.1016/j.ica.2018.01.020
Pasatoiu TD, Ghirri A, Madalan AM et al (2014) Octanuclear [NiII4LnIII4] complexes. Synthesis, crystal structures and magnetocaloric properties. Dalton Trans 43:9136–9142. https://doi.org/10.1039/C4DT00515E
Wang H (2009) Aqua{6,6′-dimethoxy-2,2′-[propane-1,3-diylbis(nitrilomethylidyne)]diphenolato}copper(II). Acta Crystallogr Sect E Struct Rep Online 65:m1490–m1490. https://doi.org/10.1107/S1600536809042755
Roy S, Basak T, Khan S et al (2017) A combined experimental and theoretical study on the formation of a cyclic tetrameric water cluster and a similar type of cyclic cluster in copper(II) Schiff base complexes. Chem Sel 2:9336–9343. https://doi.org/10.1002/slct.201701266
Cucos A, Ursu A, Madalan AM et al (2011) Co-crystallization of coordination compounds through second-coordination sphere interactions. CrystEngComm 13:3756. https://doi.org/10.1039/c1ce05112a
Routaray A, Nath N, Maharana T, Kumar AS (2015) Synthesis and immortal ROP of l-lactide using copper complex. J Macromol Sci Part A 52:444–453. https://doi.org/10.1080/10601325.2015.1029370
Sheldrick GM (2015) SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr Sect A Found Adv 71:3–8. https://doi.org/10.1107/S2053273314026370
Dolomanov OV, Bourhis LJ, Gildea RJ et al (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Crystallogr 42:339–341. https://doi.org/10.1107/S0021889808042726
Routaray A, Nath N, Maharana T et al (2016) Salicylaldimine Copper(II) complex catalyst: pioneer for ring opening polymerization of lactide. J Chem Sci 128:883–891. https://doi.org/10.1007/s12039-016-1091-3
Nakamoto K (2008) Infrared and Raman spectra of inorganic and coordination compounds. Wiley, Hoboken, pp 1–273. https://doi.org/10.1002/9780470405888.ch1
Maurya RC, Patel P, Rajput S (2003) Synthesis and characterization of N-(o-Vanillinidene)-p-anisidine and N,N′-bis(o-Vanillinidene)ethylenediamine and their metal complexes. Synth React Inorg Met Chem 33:817–836. https://doi.org/10.1081/SIM-120021648
Banu KS, Mukherjee M, Guha A et al (2012) Dinuclear copper(II) complexes: solvent dependent catecholase activity. Polyhedron 45:245–254. https://doi.org/10.1016/j.poly.2012.06.087
Chattopadhyay T, Mukherjee M, Mondal A et al (2010) A unique nickel system having versatile catalytic activity of biological significance. Inorg Chem 49:3121–3129. https://doi.org/10.1021/ic901546t
Banu KS, Chattopadhyay T, Banerjee A et al (2008) Catechol oxidase activity of a series of new dinuclear copper(II) complexes with 3,5-DTBC and TCC as substrates: syntheses, X-ray crystal structures, spectroscopic characterization of the adducts and kinetic studies. Inorg Chem 47:7083–7093. https://doi.org/10.1021/ic701332w
Banu KS, Chattopadhyay T, Banerjee A et al (2009) Catechol oxidase activity of dinuclear copper(II) complexes of Robson type macrocyclic ligands: syntheses, X-ray crystal structure, spectroscopic characterization of the adducts and kinetic studies. J Mol Catal A Chem 310:34–41. https://doi.org/10.1016/j.molcata.2009.05.016
Adak P, Mondal A, Chattopadhyay SK (2020) Manganese(II) complex of an oxygen–nitrogen donor Schiff base ligand showing efficient catechol oxidase activity: synthesis, spectroscopic and kinetic study. New J Chem 44:3748–3754. https://doi.org/10.1039/C9NJ04591K
Solomon EI, Sundaram UM, Machonkin TE (1996) Multicopper oxidases and oxygenases. Chem Rev 96:2563–2606. https://doi.org/10.1021/cr950046o
Güell M, Siegbahn PEM (2007) Theoretical study of the catalytic mechanism of catechol oxidase. JBIC J Biol Inorg Chem 12:1251–1264. https://doi.org/10.1007/s00775-007-0293-z
Kaizer J, Baráth G, Csonka R et al (2008) Catechol oxidase and phenoxazinone synthase activity of a manganese(II) isoindoline complex. J Inorg Biochem 102:773–780. https://doi.org/10.1016/j.jinorgbio.2007.11.014
Blay G, Fernández I, Pedro JR et al (2006) Chemistry and reactivity of dinuclear manganese oxamate complexes: aerobic catechol oxidation catalyzed by high-valent bis(oxo)-bridged dimanganese(IV) complexes with a homologous series of binucleating 4,5-disubstituted-o-phenylenedioxamate ligands. J Mol Catal A Chem 250:20–26. https://doi.org/10.1016/j.molcata.2006.01.021
Hitomi Y, Ando A, Matsui H et al (2005) Aerobic catechol oxidation catalyzed by a Bis(μ-oxo)dimanganese(III, III) complex via a manganese(II)−semiquinonate complex. Inorg Chem 44:3473–3478. https://doi.org/10.1021/ic050109d
Mukherjee S, Weyhermüller T, Bothe E et al (2004) Dinuclear and mononuclear manganese(IV)–radical complexes and their catalytic catecholase activity. Dalton Trans. https://doi.org/10.1039/B410842F
Doctrow SR, Huffman K, Marcus CB et al (2002) Salen−manganese complexes as catalytic scavengers of hydrogen peroxide and cytoprotective agents: structure−activity relationship studies. J Med Chem 45:4549–4558. https://doi.org/10.1021/jm020207y
Prasad RV, Thakkar NV (1994) Study of cobalt complexes as catalysts in the decomposition of hydrogen peroxide. J Mol Catal 92:9–20. https://doi.org/10.1016/0304-5102(94)00063-8
Acknowledgements
The authors gratefully acknowledge the Spanish Ministry of Economy and Competitiveness (MINECO/FEDER) for financial support (Project CTQ2015-63858-P) and Comunidad de Madrid (Project S2017/BMD-3770-CM). One of us (Aarón Terán) acknowledge Comunidad de Madrid (Project S2017/BMD-3770-CM) for a predoctoral Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. The authors declare no competing financial interest.
Additional information
Dedicated to Professor José Antonio Campo Santillana, in memoriam.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Terán, A., Jaafar, A., Sánchez-Peláez, A.E. et al. Design and catalytic studies of structural and functional models of the catechol oxidase enzyme. J Biol Inorg Chem 25, 671–683 (2020). https://doi.org/10.1007/s00775-020-01791-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-020-01791-2