Abstract
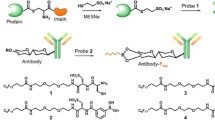
A phosphorylatable tag was designed and fused at the C-terminal end of proteins, which allowed efficient and oriented immobilization of capture proteins on glass substrates coated with a zirconium phosphonate monolayer. The concept is demonstrated using Nanofitin directed against lysozyme. This peptide tag (DSDSSSEDE) contains four serines in an acidic environment, which favored its in vitro phosphorylation by casein kinase II. The resulting phosphate cluster at the C-terminal end of the protein provided a specific, irreversible, and multipoint attachment to the zirconium surface. In a microarray format, the high surface coverage led to high fluorescence signal after incubation with Alexa Fluor 647 labeled lysozyme. The detection sensitivity of the microarray for the labeled target was below 50 pM, owing to the exceptionally low background staining, which resulted in high fluorescence signal to noise ratios. The performance of this new anchoring strategy using a zirconium phosphonate modified surface compares favorably with that of other types of microarray substrates, such as nitrocellulose-based or epoxide slides, which bind proteins in a nonoriented way.






Similar content being viewed by others
References
Zhou HH, Roy S, Schulman H, Natan MJ (2001) Trends Biotechnol 19:S34–S39
Feng YF, Ke X, Ma RS, Chen P, Hu GG, Liu FZ (2004) Clin Chem 50:416–422
Cooper MA (2002) Nat Rev Drug Discov 1:515–528
Zhu H, Bilgin M, Snyder M (2003) Annu Rev Biochem 72:783–812
Brody EN, Gold L (2000) J Biotechnol 74:5–13
Nygren PA, Skerra A (2004) J Immunol Methods 290:3–28
Porath J (1988) Trends Anal Chem 7:254–259
Porath J, Carlsson J, Olsson I, Belfrage G (1975) Nature 258:598–599
Sulkowski E (1985) Trends Biotechnol 3:1–7
Lata S, Reichel A, Brock R, Tampe R, Piehler J (2005) J Am Chem Soc 127:10205–10215
Zhu H, Bilgin M, Bangham R, Hall D, Casamayor A, Bertone P, Lan N, Jansen R, Bidlingmaier S, Houfek T, Mitchell T, Miller P, Dean RA, Gerstein M, Snyder M (2001) Science 293:2101–2105
Kroger D, Liley M, Schiweck W, Skerra A, Vogel H (1999) Biosens Bioelectron 14:155–161
Sigal GB, Bamdad C, Barberis A, Strominger J, Whitesides GM (1996) Anal Chem 68:490–497
Lata S, Piehler J (2005) Anal Chem 77:1096–1105
Cinier M, Petit M, Williams MN, Fabre RM, Pecorari F, Talham DR, Bujoli B, Tellier C (2009) Bioconjug Chem 20:2270–2277
Tinazli A, Piehler J, Beuttler M, Guckenberger R, Tampe R (2007) Nat Nanotechnol 2:220–225
Stensballe A, Andersen S, Jensen ON (2001) Proteomics 1:207–222
Posewitz MC, Tempst P (1999) Anal Chem 71:2883–2892
Zhou HJ, Ye ML, Dong J, Han GH, Jiang XN, Wu RN, Zou HF (2008) J Proteome Res 7:3957–3967
Zhou HJ, Xu SY, Ye ML, Feng S, Pan C, Jiang XG, Li X, Han GH, Fu Y, Zou H (2006) J Proteome Res 5:2431–2437
Thingholm TE, Jensen ON, Larsen MR (2009) Proteomics 9:1451–1468
Nonglaton G, Benitez IO, Guisle I, Pipelier M, Leger J, Dubreuil D, Tellier C, Talham DR, Bujoli B (2004) J Am Chem Soc 126:1497–1502
Lane SM, Monot J, Petit M, Tellier C, Bujoli B, Talham DR (2008) Langmuir 24:7394–7399
Mouratou B, Schaeffer F, Guilvout I, Tello-Manigne D, Pugsley AP, Alzari PM, Pecorari F (2007) Proc Natl Acad Sci USA 104:17983–17988
Krehenbrink M, Chami M, Guilvout I, Alzari PM, Pecorari F, Pugsley AP (2008) J Mol Biol 383:1058–1068
Yamaguchi Y, Wada T, Suzuki F, Takagi T, Hasegawa J, Handa H (1998) Nucleic Acids Res 26:3854–3861
Maoz R, Sagiv J (1984) J Colloid Interface Sci 100:465–496
Byrd H, Pike JK, Talham DR (1993) Chem Mater 5:709–715
Byrd H, Pike JK, Talham DR (1994) J Am Chem Soc 116:7903–7904
Marin O, Meggio F, Draetta G, Pinna LA (1992) FEBS Lett 301:111–114
Meggio F, Pinna LA (2003) FASEB J 17:349–368
Dobrowolska G, Meggio F, Marin O, Lozeman FJ, Li DX, Pinna LA, Krebs EG (1994) FEBS Lett 355:237–241
Meggio F, Marchiori F, Borin G, Chessa G, Pinna LA (1984) J Biol Chem 259:4576–4579
Marin O, Meggio F, Marchiori F, Borin G, Pinna LA (1986) Eur J Biochem 160:239–244
Monot J, Petit M, Lane SM, Guisle I, Leger J, Tellier C, Talham DR, Bujoli B (2008) J Am Chem Soc 130:6243–6251
Acknowledgments
This work was partially supported by grants (no. 04.04.025 and no. 2008.34.0010) from the Délégation Générale à l’Armement (DGA). M.C. was supported by a grant from the DGA. Partial support for this work was also provided by the US National Science Foundation Division of Chemistry under grant no. 0957155 (cofunded by the MPS/CHE and the Office of International Science and Engineering) (DRT). F.P. was supported by a grant from La Région des Pays de la Loire. Mass spectrometry analyses were performed within the BIBS platform located at the INRA Center of Angers-Nantes (INRA, UR1268 Biopolymères Interactions Assemblages, Nantes; http://www.angers-nantes.inra.fr/plateformes_et_plateaux_techniques/plateforme_bibs).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cinier, M., Petit, M., Pecorari, F. et al. Engineering of a phosphorylatable tag for specific protein binding on zirconium phosphonate based microarrays. J Biol Inorg Chem 17, 399–407 (2012). https://doi.org/10.1007/s00775-011-0863-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-011-0863-y