Abstract
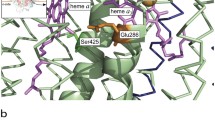
The crystal structures of the oxidized and reduced forms of cytochrome c″ from Methylophilus methylotrophus were solved from X-ray synchrotron data to atomic resolution. The overall fold of the molecule in the two redox states is very similar and is comparable to that of the oxygen-binding protein from the purple phototrophic bacterium Rhodobacter sphaeroides. However, significant modifications occur near the haem group, in particular the detachment from axial binding of His95 observed upon reduction as well as the adoption of different conformations of some protonatable residues that form a possible proton path from the haem pocket to the protein surface. These changes are associated with the previously well characterized redox-Bohr behaviour of this protein. Furthermore they provide a model for one of the presently proposed mechanisms of proton translocation in the much more complex protein cytochrome c oxidase.






Similar content being viewed by others
References
Costa HS, Santos H, Turner DL (1994) Eur J Biochem 223:783–789
Santos H, Turner DL (1988) Biochim Biophys Acta 954:277–286
Berry MJ, George SJ, Thomson AJ, Santos H, Turner DL (1990) Biochem J 270:413–417
Costa HS, Santos H, Turner DL, Xavier AV (1992) Eur J Biochem 208:427–433
Costa H, Santos H, Turner DL (1996) Eur J Biophys 25:19–24
Costa HS, Santos H, Turner DL (1993) Eur J Biochem 215:817–824
Indiani C, de Sanctis G, Neri F, Santos H, Smulevich G, Coletta M (2000) Biochemistry 39:8234–8242
Leys D, Backers K, Meyer TE, Hagen WR, Cusanovich MA, Van Beeumen JJ (2000) J Biol Chem 275:16050–16056
Papa S, Guerrieri F, Izzo G (1986) Methods Enzymol 126:331–343
Xavier AV (2004) Biochim Biophys Acta 1658:23–30
Wikstrom M (2004) Biochim Biophys Acta 1655:241–247
Enguita FJ, Rodrigues L, Archer M, Sieker L, Rodrigues A, Pohl E, Turner DL, Santos H, Carrondo MA (2003) Acta Crystallogr D Biol Crystallogr 59:580–583
Pohl E, Gonzalez A, Hermes C, van Silfhout RG (2001) J Synchrotron Rad 8:1113–1120
Collaborative Computational Project N (1994) Acta Crystallogr D Biol Crystallogr 50:760–763
Vagin A, Teplyakov A (2000) Acta Crystallogr D Biol Crystallogr 56 (Pt 12):1622–1624
Terwilliger TC (2003) Methods Enzymol 374:22–37
Cowtan KD (1996) Acta Crystallogr D Biol Crystallogr 52:43–48
Morris RJ, Perrakis A, Lamzin VS (2003) Methods Enzymol 374:229–244
McRee DE (1999) J Struct Biol 125:156–165
Sheldrick GM, Sheneider TR (1997) In: Sweet RM, Carter CW Jr (eds) Methods in enzymology. Academic, Orlando, pp 319–343
Murshudov GN, Vagin AA, Lebedev A, Wilson KS, Dodson EJ (1999) Acta Crystallogr D Biol Crystallogr 55 (Pt 1):247–255
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) J Appl Cryst 26:283–291
Fiser A, Sali A (2003) Methods Enzymol 374:461–491
DeLano WL (2002) The PyMOL molecular graphics system. Delano Scientific, San Carlos
Brennan L, Turner DL, Fareleira P, Santos H (2001) J Mol Biol 308:353–365
Frazao C, Enguita FJ, Coelho R, Sheldrick GM, Navarro JA, Hervas M, De la Rosa MA, Carrondo MA (2001) J Biol Inorg Chem 6:324–332
Allen JW, Daltrop O, Stevens JM, Ferguson SJ (2003) Philos Trans R Soc Lond B Biol Sci 358:255–266
Baker NA, Sept D, Joseph S, Holst MJ, McCammon JA (2001) Proc Natl Acad Sci USA 98:10037–10041
Fulop V, Ridout CJ, Greenwood C, Hajdu J (1995) Structure 3:1225–1233
Williams PA, Fulop V, Garman EF, Saunders NF, Ferguson SJ, Hajdu J (1997) Nature 389:406–412
Dias JM, Alves T, Bonifacio C, Pereira AS, Trincao J, Bourgeois D, Moura I, Romao MJ (2004) Structure (Camb) 12:961–973
Wikstrom M, Verkhovsky MI (2002) Biochim Biophys Acta 1555:128–132
Wikstrom M, Verkhovsky MI, Hummer G (2003) Biochim Biophys Acta 1604:61–65
Bento I, Matias PM, Baptista AM, da Costa PN, van Dongen WM, Saraiva LM, Schneider TR, Soares CM, Carrondo MA (2004) Proteins 54:135–152
Michel H (1998) Proc Natl Acad Sci USA 95:12819–12824
Wikstrom M (2000) Biochim Biophys Acta 1458:188–198
Wikstrom M (2000) Biochemistry 39:3515–3519
Xavier AV (2002) FEBS Lett 532:261–266
Acknowledgement
Access to the European Molecular Biology Laboratory–Deutsche Elektronen Synchrotron (EMBL-DESY) Hamburg Facility was supported through the European Commission program “Access to Research Infrastructure Action of the Improving Human Potential” grant HPRI-1999-CT-00017.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Enguita, F.J., Pohl, E., Turner, D.L. et al. Structural evidence for a proton transfer pathway coupled with haem reduction of cytochrome c″ from Methylophilus methylotrophus . J Biol Inorg Chem 11, 189–196 (2006). https://doi.org/10.1007/s00775-005-0065-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-005-0065-6