Abstract
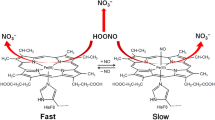
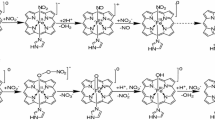
The mechanism for the reaction between nitric oxide (NO) and O2 bound to the heme iron of myoglobin (Mb), including the following isomerization to nitrate, has been investigated using hybrid density functional theory (B3LYP). Myoglobin working as a NO scavenger could be of importance, since NO reversibly inhibits the terminal enzyme in the respiration chain, cytochrome c oxidase. The concentration of NO in the cell will thus affect the respiration and thereby the synthesis of ATP. The calculations show that the reaction between NO and the heme-bound O2 gives a peroxynitrite intermediate whose O–O bond undergoes a homolytic cleavage, forming a NO2 radical and myoglobin in the oxo-ferryl state. The NO2 radical then recombines with the oxo-ferryl, forming heme-bound nitrate. Nine different models have been used in the present study to examine the effect on the reaction both by the presence and the protonation state of the distal His64, and by the surroundings of the proximal His93. The barriers going from the oxy-Mb and nitric oxide reactant to the peroxynitrite intermediate and further to the oxo-ferryl and NO2 radical are around 10 and 7 kcal/mol, respectively. Forming the product, nitrate bound to the heme iron has a barrier of less than ~7 kcal/mol. The overall reaction going from a free nitric oxide and oxy-Mb to the heme bound nitrate is exergonic by more than 30 kcal/mol.










Similar content being viewed by others
References
Olson JS, Phillips GN Jr (1997) J Inorg Biochem 2:544–552
Miller L, Patel M, Chance MR (1996) J Inorg Biochem 118:4511–4517
Brunori M (2001) Trends Biochem Sci 26:21–23
Brunori M (2001) Trends Biochem Sci 26:209–210
Bredt DS, Snyder SH (1994) Annu Rev Biochem 63:175–195
Doyle MP, Hoekstra JW (1981) J Inorg Biochem 14:351–358
Eich RF, Li T, Lemon DD, Doherty DH, Curry SR, Aitken JF, Mathews AJ, Johnson KA, Smith RD, Phillips GN Jr, Olson JS (1996) Biochemistry 35:6976–6983
Wade RS, Castro CE (1996) Chem Res Toxicol 9:1382–1390
Livingston DJ, McLachlan SJ, La Mar GN, Brown WD (1985) J Biol Chem 260:15699–15707
Herold S, Exner M, Nauser T (2001) Biochemistry 40:3385–3395
Herold S, Shivashankar K, Mehl M (2002) Biochemistry 41:13460–13472
Maurus R, Bogumil R, Nguyen NT, Mauk G, Brayer G (1998) Nitric Oxide Biol Chem 332:67–74
Cleeter MWJ, Cooper JM, Darley-Usmar VM, Moncada S, Schapira AHV (1994) FEBS Lett 345:50–54
Brown GC, Cooper CE (1994) FEBS Lett 356:295–298
Brown GC (1995) FEBS Lett 369:136–139
Sarti P, Lendaro E, Ippoliti R, Bellelli A, Benedetti PA, Brunori M (1999) FASEB J 13:191–197
Radi R, Beckman JS, Bush KM, Freeman BA (1991) J Biol Chem 266:4244–4250
Ischiropoulos H (1998) Arch Biochem Biophys 356:1–11
Radi R, Beckman JS, Bush KM, Freeman BA (1991) Arch Biochem Biophys 288:481–487
Denicola A, Freeman BA, Trujillo M, Radi R (1996) Arch Biochem Biophys 333:49–58
King PA, Anderson VE, Edwards JO, Gustafson G, Plumb RC, Suggs JW (1992) J Am Chem Soc 114:5430–5432
Radi R (1996) Chem Res Toxicol 9:828–835
Herold S, Matsui T, Watanabe Y (2001) J Am Chem Soc 123:4085–4086
Kissner R, Nauser T, Bugnon P, Lye PG, Koppenol WH (1997) Chem Res Toxicol 10:1285–1292
Bartberger MD, Olson LP, Houk KN (1998) Chem Res Toxicol 11:710–711
Merényi G, Lind J, Goldstein S, Czapski G (1998) Chem Res Toxicol 11:712–713
Lymar SV, Hurst JK (1998) Chem Res Toxicol 11:714–715
Koppenol WH (1998) Chem Res Toxicol 11:716–717
Squadrito GL, Pryor WA (1998) Chem Res Toxicol 11:718–719
Radi R (1998) Chem Res Toxicol 11:720–721
Lee J, Hunt JA, Groves JT (1998) J Am Chem Soc 120:7493–7501
Crow JP (1999) Arch Biochem Biophys 371:41–52
Balavoine GGA, Geletii YV, Bejan D (1997) Nitric Oxide Biol Chem 1:507–521
Potter WT, Tucker MP, Houtchens RA, Caughey WW (1987) Biochemistry 26:4699–4707
George P, Hanania GIH, Irvine DH, Abu-Issa I (1964) J Chem Soc 5689–5694
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA Jr, Stratmann RE, Burant JC, Dapprich S, Millan JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Head-Gordon M, Replogle ES, Pople JA (1998) Gaussian98. Gaussian. Pittsburgh, Pa., USA
Schrodinger (2000) Jaguar 4.2. Schrodinger, Portland, Ore., USA
Tannor DJ, Marten B, Murphy R, Friesner RA, Sitkoff D, Nicholls A, Ringnalda M, Goddard WA III (1994) J Am Chem Soc 116:11875–11882
Blomberg MRA, Siegbahn PEM, Babcock GT (1998) J Am Chem Soc 120:8812–8824
Curtiis LA, Raghavachari K, Redfern RC, Pople JA (2000) J Chem Phys 112:7374–7383
Siegbahn PEM, Blomberg MRA (1999) Annu Rev Phys Chem 50:221–249
Siegbahn PEM, Blomberg MRA (2000) Chem Rev 100:421–437
Blomberg MRA, Siegbahn PEM (2001) J Phys Chem B 105:9375–9386
Blomberg M, Blomberg MRA, Siegbahn PEM (2003) J Phys Chem B 107:3297–3308
Tsai HH, Hamilton TP, Tsai JHM, van der Woerd M, Harrison JG, Jablonsky MJ, Beckman JS, Koppenol WH (1996) J Phys Chem 100:15087–15095
Wanat A, Gdula-Argasinska J, Rutkowska-Zbik D, Witko M, Stochel G, van Eldik R (2002) J Biol Inorg Chem 7:165–176
Lin J, Merryweather J, Vitello LB, Erman JE (1999) Arch Biochem Biophys 362:148–158
Merryweather J, Summers F, Vitello LB, Erman JE (1998) Arch Biochem Biophys 358:359–368
Acknowledgements
We gratefully acknowledge the National Supercomputer Center (Sweden) for generous grants of computer time at the SGI3800. We also thank Dr Susanna Herold for useful discussions and suggestions.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blomberg, L.M., Blomberg, M.R.A. & Siegbahn, P.E.M. A theoretical study of myoglobin working as a nitric oxide scavenger. J Biol Inorg Chem 9, 923–935 (2004). https://doi.org/10.1007/s00775-004-0585-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-004-0585-5