Abstract.
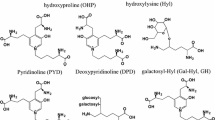
The measurement of hydroxylysylpyridinoline (PYD) and lysylpyridinoline (DPD), the degradation products of type I collagen, by manual HPLC assay posed practical difficulties. The present study was undertaken to evaluate the first commercially available HPLC kit, which provides a convenient substitute for cumbersome classical HPLC methods. The HPLC procedure is based on an improved sample clean-up chromatography, convenient ready-to-use HPLC reagents, and quicker isocratic elution of PYR and DPD on reverse-phase analytical column. The analytical parameters assessed include sensitivity, within- and between-assay variation, method comparison, recoveries, and interference. Clinical evaluation included discriminatory power of PYD and DPD and response to treatment of osteoporosis patients with Alendronate. DPD and PYD concentrations showed linear (r 2 > 0.99) response between 10–400 pmol/ml and 75–4000 pmol/ml, respectively. The average within-assay imprecision, over a range of clinically relevant cross-links concentrations, was CV < 7% (n = 30). The total imprecision (n = 35 days), by ANOVA, for PYD and DPD was CV < 7.5% and CV < 10%, respectively. Average spike recovery was 95.4% ± 6.5%. Comparison with the historical HPLC method exhibited a close correlation (r values between 0.87 and 0.91, P < 0.0001). Creatinine-corrected DPD in postmenopausal (Z score = 2.4, P < 0.05, n = 17) and osteoporotic (Z score = 3.0, P < 0.01, n = 29) women were 44% and 64% higher, respectively, compared to premenopausal samples (n = 15). Similarly, PYD concentration was 26% and 54% higher in postmenopausal and postmenopausal osteoporotic women, respectively. There was a 47% (P < 0.001) decrease in DPD concentration (n = 16), and a 30% decrease in PYD concentration after 90 days of treatment of osteoporotic patients with Alendronate. DPD concentration correlated with N-telopeptide with an r value of 0.69 (n = 67, P < 0.0001). The reported kit method is substantially simpler and precise than the manual method. DPD concentrations determined by the current method reaffirm the clinical value in identifying increased bone resorption in pathological conditions and monitoring response to antiresorptive therapy.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: June 28, 2002 / Accepted: November 6, 2002
Offprint requests to: D.J. Baylink
About this article
Cite this article
Srivastava, A., Libanati, C., Oberrauch, W. et al. Analytical and clinical evaluation of the Bio-Rad HPLC kit for measurement of type I collagen cross links. J Bone Miner Metab 21, 134–144 (2003). https://doi.org/10.1007/s007740300022
Issue Date:
DOI: https://doi.org/10.1007/s007740300022