Abstract
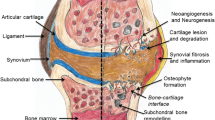
Rotator cuff tears are common musculoskeletal injuries that can cause significant pain and disability. While the clinical results of rotator cuff repair can be good, failure of tendon healing remains a significant problem. Molecular mechanisms underlying structural failure following surgical repair remain unclear. Histologically, enhanced inflammation, disorganization of the collagen fibers, calcification, apoptosis and tissue necrosis affect the normal healing process. Mesenchymal stem cells (MSCs) have the ability to provide improved healing following rotator cuff repair via the release of mediators from secreted 30–100 nm extracellular vesicles called exosomes. They carry regulatory proteins, mRNA and miRNA and have the ability to increase collagen synthesis and angiogenesis through increased expression of mRNA and release of proangiogenic factors and regulatory proteins that play a major role in proper tissue remodeling and preventing extracellular matrix degradation. Various studies have shown the effect of exosomes on improving outcome of cutaneous wound healing, scar tissue formation, degenerative bone disease and Duchenne Muscular Dystrophy. In this article, we critically reviewed the potential role of exosomes in tendon regeneration and propose the novel use of exosomes alone or seeded onto biomaterial matrices to stimulate secretion of favorable cellular factors in accelerating the healing response following rotator cuff repair.





Similar content being viewed by others
References
Zhao S, Su W, Shah V et al (2017) Biomaterials based strategies for rotator cuff repair. Colloids Surf B 157:407–416. https://doi.org/10.1016/j.colsurfb.2017.06.004
Gold JE, Hallman DM, Hellström F et al (2017) Systematic review of quantitative imaging biomarkers for neck and shoulder musculoskeletal disorders. BMC Musculoskelet Disorders 18:395. https://doi.org/10.1186/s12891-017-1694-y
Jain NB, Higgins LD, Losina E, Collins J, Blazar PE, Katz JN (2014) Epidemiology of musculoskeletal upper extremity ambulatory surgery in the United States. BMC Musculoskelet Disorders 15:4. https://doi.org/10.1186/1471-2474-15-4
Longo UG, Berton A, Khan WS, Maffulli N, Denaro V (2011) Histopathology of rotator cuff tears. Sports Med Arthrosc Rev 19:227–236. https://doi.org/10.1097/JSA.0b013e318213bccb
Rashid MS, Cooper C, Cook J et al (2017) Increasing age and tear size reduce rotator cuff repair healing rate at 1 year. Acta Orthop 88:606–611. https://doi.org/10.1080/17453674.2017.1370844
Patel S, Caldwell J‐M, Doty SB et al (2017) Integrating soft and hard tissues via interface tissue engineering. J Orthop Res. https://doi.org/10.1002/jor.23810
Ruyssen-Witrand A, Jamard B, Cantagrel A et al (2017) Relationships between ultrasound enthesitis, disease activity and axial radiographic structural changes in patients with early spondyloarthritis: data from DESIR cohort. RMD Open 3:e000482. https://doi.org/10.1136/rmdopen-2017-000482
Hideaki Takahashi, Hiroyuki Tamaki, Mineo Oyama, Noriaki Yamamoto, Hideaki Onishi (2017) Time-dependent changes in the structure of calcified fibrocartilage in the rat achilles tendon-bone interface with sciatic denervation. Anat Rec 300:2166–2174. https://doi.org/10.1002/ar.23684
Hexter AT, Pendegrass C, Haddad F, Blunn G (2017) Demineralized bone matrix to augment tendon-bone healing: a systematic review. Orthop J Sports Med. https://doi.org/10.1177/2325967117734517
Nakagawa H, Morihara T, Fujiwara H et al (2017) Effect of footprint preparation on tendon-to-bone healing a histologic and biomechanical study in a rat rotator cuff repair model. Arthrosc J Arthroscopic Relat Surg 33:1482–1492. https://doi.org/10.1016/j.arthro.2017.03.031
Hernigou P, Flouzat Lachaniette CH, Delambre J et al (2014) Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop 38:1811–1818. https://doi.org/10.1007/s00264-014-2391-1
Zitnay JL, Reese SP, Tran G, Farhang N, Bowles RD, Weiss JA (2018) Fabrication of dense anisotropic collagen scaffolds using biaxial compression. Acta Biomater 65:76–87. https://doi.org/10.1016/j.actbio.2017.11.017
Fellows CR, Matta C, Zakany R, Khan IM, Mobasheri A (2016) Adipose, bone marrow and synovial joint-derived mesenchymal stem cells for cartilage repair. Front Genet. https://doi.org/10.3389/fgene.2016.00213
Mazzocca AD, McCarthy MBR, Chowaniec DM, Cote MP, Arciero RA, Drissi H (2010) Rapid isolation of human stem cells (connective tissue progenitor cells) from the proximal humerus during arthroscopic rotator cuff surgery. Am J Sports Med 38:1438–1447. https://doi.org/10.1177/0363546509360924
Honda H, Gotoh M, Kanazawa T et al (2017) Hyaluronic acid accelerates tendon-to-bone healing after rotator cuff repair. Am J Sports Med 45:3322–3330. https://doi.org/10.1177/0363546517720199
Sevivas N, Teixeira FG, Portugal R et al (2018) Mesenchymal stem cell secretome improves tendon cell viability in vitro and tendon-bone healing in vivo when a tissue engineering strategy is used in a rat model of chronic massive rotator cuff tear. Am J Sports Med 46:449–459. https://doi.org/10.1177/0363546517735850
Kim YS, Sung CH, Chung SH, Kwak SJ, Koh YG (2017) Does an injection of adipose-derived mesenchymal stem cells loaded in fibrin glue influence rotator cuff repair outcomes? A clinical and magnetic resonance imaging study. Am J Sports Med 45:2010–2018. https://doi.org/10.1177/0363546517702863
Bi Y, Ehirchiou D, Kilts TM et al (2007) Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med 13:1219–1227. https://doi.org/10.1038/nm1630
Zhang J, Pan T, Liu Y, Wang JH-C (2010) Mouse treadmill running enhances tendons by expanding the pool of tendon stem cells (TSCs) and TSC-related cellular production of collagen. J Orthop Res 28:1178–1183. https://doi.org/10.1002/jor.21123
Crescitelli R, Lässer C, Szabó TG et al (2013) Distinct RNA profiles in subpopulations of extracellular vesicles: apoptotic bodies, microvesicles and exosomes. J Extracell Vesicles 2:20677. https://doi.org/10.3402/jev.v2i0.20677
Zonneveld MI, Brisson AR, van Herwijnen MJC et al (2014) Recovery of extracellular vesicles from human breast milk is influenced by sample collection and vesicle isolation procedures. J Extracell Vesicles 3:24215. https://doi.org/10.3402/jev.v3.24215
Kida Y, Morihara T, Matsuda K-I et al (2013) Bone marrow–derived cells from the footprint infiltrate into the repaired rotator cuff. J Shoulder Elbow Surg 22:197–205. https://doi.org/10.1016/j.jse.2012.02.007
Gasperi RD, Hamidi S, Harlow LM, Ksiezak-Reding H, Bauman WA, Cardozo CP (2017) Denervation-related alterations and biological activity of miRNAs contained in exosomes released by skeletal muscle fibers. Sci Rep 7:12888. https://doi.org/10.1038/s41598-017-13105-9
Munoz JL, Bliss SA, Greco SJ, Ramkissoon SH, Ligon KL, Rameshwar P (2013) Delivery of functional anti-mir-9 by mesenchymal stem cell–derived exosomes to glioblastoma multiforme cells conferred chemosensitivity. Mol Ther Nucleic Acids 2:e126. https://doi.org/10.1038/mtna.2013.60
Than UTT, Guanzon D, Leavesley D, Parker T (2017) Association of extracellular membrane vesicles with cutaneous wound healing. Int J Mol Sci 18:956. https://doi.org/10.3390/ijms18050956
Fouda MB, Thankam FG, Dilisio MF, Agrawal DK (2017) Alterations in tendon microenvironment in response to mechanical load: potential molecular targets for treatment strategies. Am J Transl Res. 9:4341–4360
Hu L, Wang J, Zhou X et al (2016) Exosomes derived from human adipose mesenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. https://doi.org/10.1038/srep32993
Zhang J, Guan J, Niu X et al (2015) Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J Transl Med 13:1–2. https://doi.org/10.1186/s12967-015-0417-0
Choi EW, Seo MK, Woo EY, Kim SH, Park EJ, Kim S (2017) Exosomes from human adipose-derived stem cells promote proliferation and migration of skin fibroblasts. Exp Dermatol. https://doi.org/10.1111/exd.13451
El-Tookhy OS, Shamaa AA, Shehab GG, Abdallah AN, Azzam OM (2017) Histological evaluation of experimentally induced critical size defect skin wounds using exosomal solution of mesenchymal stem cells derived microvesicles. Int J Stem Cells 10:144–153. https://doi.org/10.15283/ijsc17043
Hocking AM (2012) Mesenchymal stem cell therapy for cutaneous wounds. Adv Wound Care (New Rochelle) 1:166–171. https://doi.org/10.1089/wound.2011.0294
Wang L, Hu L, Zhou X et al (2017) Exosomes secreted by human adipose mesenchymal stem cells promote scarless cutaneous repair by regulating extracellular matrix remodelling. Sci Rep 7:13321. https://doi.org/10.1038/s41598-017-12919-x
Komaki M, Numata Y, Morioka C et al (2017) Exosomes of human placenta-derived mesenchymal stem cells stimulate angiogenesis. Stem Cell Res Ther. https://doi.org/10.1186/s13287-017-0660-9
Phinney DG, Pittenger MF (2017) MSC-derived exosomes for cell-free therapy. Stem Cells 35:851–858
Xue M, Jackson CJ (2015) Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. Adv Wound Care (New Rochelle). 4:119–136. https://doi.org/10.1089/wound.2013.0485
Laghezza Masci V, Taddei AR, Gambellini G, Giorgi F, Fausto AM (2016) Microvesicles shed from fibroblasts act as metalloproteinase carriers in a 3-D collagen matrix. J Circ Biomark. https://doi.org/10.1177/1849454416663660
Nakamura K, Jinnin M, Harada M et al (2016) Altered expression of CD63 and exosomes in scleroderma dermal fibroblasts. J Dermatol Sci 84:30–39. https://doi.org/10.1016/j.jdermsci.2016.06.013
Qi X, Zhang J, Yuan H et al (2016) Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells repair critical-sized bone defects through enhanced angiogenesis and osteogenesis in osteoporotic rats. Int J Biol Sci 12:836–849. https://doi.org/10.7150/ijbs.14809
Wang Y, Yu D, Liu Z et al (2017) Exosomes from embryonic mesenchymal stem cells alleviate osteoarthritis through balancing synthesis and degradation of cartilage extracellular matrix. Stem Cell Res Ther 8:189. https://doi.org/10.1186/s13287-017-0632-0
Tao S-C, Yuan T, Zhang Y-L, Yin W-J, Guo S-C, Zhang C-Q (2017) Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics 7:180–195. https://doi.org/10.7150/thno.17133
Bier A, Berenstein P, Kronfeld N et al (2018) Placenta-derived mesenchymal stromal cells and their exosomes exert therapeutic effects in Duchenne Muscular Dystrophy. Biomaterials 174:67–78. https://doi.org/10.1016/j.biomaterials.2018.04.055
McNally E (2012) Chapter 81—novel targets and approaches to treating skeletal muscle disease. In: Hill JA, Olson EN (eds) Muscle. Academic Press, Boston/Waltham, pp 1095–1103. https://doi.org/10.1016/b978-0-12-381510-1.00081-8
Feng Y, Huang W, Wani M, Yu X, Ashraf M (2014) Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS One 9:e88685. https://doi.org/10.1371/journal.pone.0088685
Ferguson SW, Wang J, Lee CJ et al (2018) The microRNA regulatory landscape of MSC-derived exosomes: a systems view. Sci Rep. https://doi.org/10.1038/s41598-018-19581-x
Xu T, Xu M, Bai J et al (2019) Tenocyte-derived exosomes induce the tenogenic differentiation of mesenchymal stem cells through TGF-β. Cytotechnology 71:57–65. https://doi.org/10.1007/s10616-018-0264-y
Tokunaga T, Shukunami C, Okamoto N et al (2015) FGF-2 stimulates the growth of tenogenic progenitor cells to facilitate the generation of tenomodulin-positive tenocytes in a rat rotator cuff healing model. Am J Sports Med 43:2411–2422. https://doi.org/10.1177/0363546515597488
Tokunaga T, Karasugi T, Arimura H et al (2017) Enhancement of rotator cuff tendon–bone healing with fibroblast growth factor 2 impregnated in gelatin hydrogel sheets in a rabbit model. J Shoulder Elbow Surg 26:1708–1717. https://doi.org/10.1016/j.jse.2017.03.020
Cui H, He Y, Chen S, Zhang D, Yu Y, Fan C (2018) Macrophage-derived miRNA-containing exosomes induce peritendinous fibrosis after tendon injury through the miR-21-5p/Smad7 pathway. Mol Ther Nucleic Acids 14:114–130. https://doi.org/10.1016/j.omtn.2018.11.006
Gaffey AC, Chen MH, Venkataraman CM et al (2015) Injectable shear-thinning hydrogels used to deliver endothelial progenitor cells, enhance cell engraftment, and improve ischemic myocardium. J Thorac Cardiovasc Surg 150:1268–1277. https://doi.org/10.1016/j.jtcvs.2015.07.035
Agrawal DK, Siddique A (2018) Rejuvenation of “broken heart” with bioengineered gel. J Thorac Cardiovasc Surg. https://doi.org/10.1016/j.jtcvs.2018.08.076
Nam HY, Pingguan-Murphy B, Abbas AA, Merican AM, Kamarul T (2015) The proliferation and tenogenic differentiation potential of bone marrow-derived mesenchymal stromal cell are influenced by specific uniaxial cyclic tensile loading conditions. Biomech Model Mechanobiol 14:649–663. https://doi.org/10.1007/s10237-014-0628-y
Miyamoto H, Aoki M, Hidaka E, Fujimiya M, Uchiyama E (2017) Measurement of strain and tensile force of the Supraspinatus tendon under conditions that simulates low angle isometric elevation of the gleno-humeral joint: influence of adduction torque and joint positioning. Clin Biomech 50:92–98. https://doi.org/10.1016/j.clinbiomech.2017.10.014
Nam HY, Raghavendran HRB, Pingguan-Murphy B, Abbas AA, Merican AM, Kamarul T (2017) Fate of tenogenic differentiation potential of human bone marrow stromal cells by uniaxial stretching affected by stretch-activated calcium channel agonist gadolinium. PLoS One 12:e0178117. https://doi.org/10.1371/journal.pone.0178117
Samanta S, Rajasingh S, Drosos N, Zhou Z, Dawn B, Rajasingh J (2018) Exosomes: new molecular targets of diseases. Acta Pharmacol Sin 39:501–513. https://doi.org/10.1038/aps.2017.162
Tracy SA, Ahmed A, Tigges JC et al (2019) A comparison of clinically relevant sources of mesenchymal stem cell-derived exosomes: bone marrow and amniotic fluid. J Pediatr Surg 54:86–90. https://doi.org/10.1016/j.jpedsurg.2018.10.020
Acknowledgements
This work was supported primarily by the State of Nebraska LB506 Grant to DKA and LB692 Grant to MFD by Creighton University. The research work of DKA is also supported by Grants R01HL120659 and R01HL144125 from the National Institutes of Health (NIH). The content of this original research article is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or the State of Nebraska.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have read the journal’s policy on disclosure of potential conflicts of interest. The authors have no other relevant affiliations or financial or non-financial involvement with any organization or entity with financial or non-financial interest or conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. No writing assistance was utilized in the production of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Connor, D.E., Paulus, J.A., Dabestani, P.J. et al. Therapeutic potential of exosomes in rotator cuff tendon healing. J Bone Miner Metab 37, 759–767 (2019). https://doi.org/10.1007/s00774-019-01013-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-019-01013-z