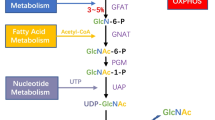
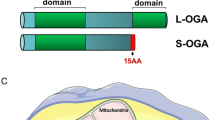
Abstract
O-GlcNAc transferase (OGT) and O-GlcNAcase (OGA) catalyze the dynamic cycling of intracellular, post-translational O-GlcNAc modification on thousands of Ser/Thr residues of cytosolic, nuclear, and mitochondrial signaling proteins. The identification of O-GlcNAc modified substrates has revealed a functionally diverse set of proteins, and the extent of O-GlcNAcylation fluctuates in response to nutrients and cellular stress. As a result, OGT and OGA are implicated in widespread, nutrient-responsive regulation of numerous signaling pathways and transcriptional programs. These enzymes are required for normal embryonic development and are dysregulated in metabolic and age-related disease states. While a recent surge of interest in the field has contributed to understanding the functional impacts of protein O-GlcNAcylation, little is known about the upstream mechanisms which modulate OGT and OGA substrate targeting. This review focuses on elements of enzyme structure among splice variants, post-translational modification, localization, and regulatory protein interactions which drive the specificity of OGT and OGA toward different subsets of the cellular proteome. Ongoing efforts in this rapidly advancing field are aimed at revealing mechanisms of OGT and OGA regulation to harness the potential therapeutic benefit of manipulating these enzymes’ activities.



Similar content being viewed by others
References
Akimoto Y, Kreppel LK, Hirano H, Hart GW (1999) Localization of the O-linked N-acetylglucosamine transferase in rat pancreas. Diabetes 48:2407–2413
Olsen JV et al (2010) Quantitative phosphoproteomics reveals widespread full phosphorylation site occupancy during mitosis. Sci Signal 3:ra3. doi:10.1126/scisignal.2000475
Alfaro JF et al (2012) Tandem mass spectrometry identifies many mouse brain O-GlcNAcylated proteins including EGF domain-specific O-GlcNAc transferase targets. Proc Natl Acad Sci USA 109:7280–7285. doi:10.1073/pnas.1200425109
Andres-Bergos J, Tardio L, Larranaga-Vera A, Gomez R, Herrero-Beaumont G, Largo R (2012) The increase in o-linked N-acetylglucosamine protein modification stimulates chondrogenic differentiation both in vitro and in vivo. J Biol Chem 287:33615–33628. doi:10.1074/jbc.M112.354241
Anthonisen EH, Berven L, Holm S, Nygard M, Nebb HI, Gronning-Wang LM (2010) Nuclear receptor liver X receptor is O-GlcNAc-modified in response to glucose. J Biol Chem 285:1607–1615. doi:10.1074/jbc.M109.082685
Basu R et al (2006) Effects of age and sex on postprandial glucose metabolism: differences in glucose turnover, insulin secretion, insulin action, and hepatic insulin extraction. Diabetes 55:2001–2014. doi:10.2337/db05-1692
Boehmelt G et al (2000) Decreased UDP-GlcNAc levels abrogate proliferation control in EMeg32-deficient cells. The EMBO journal 19:5092–5104. doi:10.1093/emboj/19.19.5092
Boss GR, Seegmiller JE (1981) Age-related physiological changes and their clinical significance. West J med 135:434–440
Braidman I et al (1974) Characterisation of human N-acetyl-beta-hexosaminidase C. FEBS Lett 41:181–184
Bullen JW, Balsbaugh JL, Chanda D, Shabanowitz J, Hunt DF, Neumann D, Hart GW (2014) Cross-talk between two essential nutrient-sensitive enzymes: O-GlcNAc transferase (OGT) and AMP-ACTIVATED protein kinase (AMPK). J Biol Chem 289:10592–10606. doi:10.1074/jbc.M113.523068
Butkinaree C, Cheung WD, Park S, Park K, Barber M, Hart GW (2008) Characterization of beta-N-acetylglucosaminidase cleavage by caspase-3 during apoptosis. J Biol Chem 283:23557–23566. doi:10.1074/jbc.M804116200
Butkinaree C, Park K, Hart GW (2010) O-linked beta-N-acetylglucosamine (O-GlcNAc): extensive crosstalk with phosphorylation to regulate signaling and transcription in response to nutrients and stress. Biochim Biophys Acta 1800:96–106. doi:10.1016/j.bbagen.2009.07.018
Capotosti F et al (2011) O-GlcNAc transferase catalyzes site-specific proteolysis of HCF-1. Cell 144:376–388. doi:10.1016/j.cell.2010.12.030
Chen Q, Chen Y, Bian C, Fujiki R, Yu X (2013) TET2 promotes histone O-GlcNAcylation during gene transcription. Nature 493:561–564. doi:10.1038/nature11742
Cheung WD, Hart GW (2008) AMP-activated protein kinase and p38 MAPK activate O-GlcNAcylation of neuronal proteins during glucose deprivation. J Biol Chem 283:13009–13020. doi:10.1074/jbc.M801222200
Cheung WD, Sakabe K, Housley MP, Dias WB, Hart GW (2008) O-linked beta-N-acetylglucosaminyltransferase substrate specificity is regulated by myosin phosphatase targeting and other interacting proteins. J Biol Chem 283:33935–33941. doi:10.1074/jbc.M806199200
Comer FI, Hart GW (2001) Reciprocity between O-GlcNAc and O-phosphate on the carboxyl terminal domain of RNA polymerase II. Biochemistry 40:7845–7852
Comtesse N, Maldener E, Meese E (2001) Identification of a nuclear variant of MGEA5, a cytoplasmic hyaluronidase and a beta-N-acetylglucosaminidase. Biochem Biophys Res Commun 283:634–640. doi:10.1006/bbrc.2001.4815
Dehennaut V et al (2007) O-linked N-acetylglucosaminyltransferase inhibition prevents G2/M transition in Xenopus laevis oocytes. J Biol Chem 282:12527–12536. doi:10.1074/jbc.M700444200
Dehennaut V et al (2008) Identification of structural and functional O-linked N-acetylglucosamine-bearing proteins in Xenopus laevis oocyte. MCP 7:2229–2245. doi:10.1074/mcp.M700494-MCP200
Dennis RJ et al (2006) Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity. Nat Struct Mol Biol 13:365–371. doi:10.1038/nsmb1079
Dentin R, Hedrick S, Xie J, Yates J, 3rd, Montminy M (2008) Hepatic glucose sensing via the CREB coactivator CRTC2. Science (New York, NY) 319:1402–1405. doi:10.1126/science.1151363
Deplus R et al (2013) TET2 and TET3 regulate GlcNAcylation and H3K4 methylation through OGT and SET1/COMPASS. EMBO J 32:645–655. doi:10.1038/emboj.2012.357
Dong DL, Hart GW (1994) Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol. J Biol Chem 269:19321–19330
Drougat L et al (2012) Characterization of O-GlcNAc cycling and proteomic identification of differentially O-GlcNAcylated proteins during G1/S transition. Biochim Biophys Acta 1820:1839–1848. doi:10.1016/j.bbagen.2012.08.024
Feng Z, Hui Y, Ling L, Xiaoyan L, Yuqiu W, Peng W, Lianwen Z (2013) FBXW10 is negatively regulated in transcription and expression level by protein O-GlcNAcylation. Biochem Biophys Res Commun 438:427–432. doi:10.1016/j.bbrc.2013.07.091
Fletcher BS, Dragstedt C, Notterpek L, Nolan GP (2002) Functional cloning of SPIN-2, a nuclear anti-apoptotic protein with roles in cell cycle progression. Leukemia 16:1507–1518. doi:10.1038/sj.leu.2402557
Fujiki R et al (2009) GlcNAcylation of a histone methyltransferase in retinoic-acid-induced granulopoiesis. Nature 459:455–459. doi:10.1038/nature07954
Fulop N et al (2008) Aging leads to increased levels of protein O-linked N-acetylglucosamine in heart, aorta, brain and skeletal muscle in Brown-Norway rats. Biogerontology 9:139–151. doi:10.1007/s10522-007-9123-5
Gambetta MC, Oktaba K, Muller J (2009) Essential role of the glycosyltransferase sxc/Ogt in polycomb repression. Science (New York, NY) 325:93–96. doi:10.1126/science.1169727
Gao Y (2010) Mechanism, structure, and inhibition of O-GlcNAc processing enzymes. Curr Signal Transduct Ther 5:74–91
Gao Y, Wells L, Comer FI, Parker GJ, Hart GW (2001) Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain. J Biol Chem 276:9838–9845. doi:10.1074/jbc.M010420200
Gawlowski T et al (2012) Modulation of dynamin-related protein 1 (DRP1) function by increased O-linked-beta-N-acetylglucosamine modification (O-GlcNAc) in cardiac myocytes. J Biol Chem 287:30024–30034. doi:10.1074/jbc.M112.390682
Guinez C et al (2011) O-GlcNAcylation increases ChREBP protein content and transcriptional activity in the liver. Diabetes 60:1399–1413. doi:10.2337/db10-0452
Haltiwanger RS, Blomberg MA, Hart GW (1992) Glycosylation of nuclear and cytoplasmic proteins. Purification and characterization of a uridine diphospho-N-acetylglucosamine:polypeptide beta-N-acetylglucosaminyltransferase. J Biol Chem 267:9005–9013
Hanover JA, Yu S, Lubas WB, Shin SH, Ragano-Caracciola M, Kochran J, Love DC (2003) Mitochondrial and nucleocytoplasmic isoforms of O-linked GlcNAc transferase encoded by a single mammalian gene. Arch Biochem Biophys 409:287–297
Hanover JA, Krause MW, Love DC (2010) The Hexosamine Signaling Pathway: O-GlcNAc cycling in feast or famine. Biochim Biophys Acta 1800:80. doi:10.1016/j.bbagen.2009.07.017
Hanover JA, Krause MW, Love DC (2012) Bittersweet memories: linking metabolism to epigenetics through O-GlcNAcylation. Nat Rev Mol Cell Biol 13:312–321. doi:10.1038/nrm3334
Hart GW, Slawson C, Ramirez-Correa G, Lagerlof O (2011) Cross talk between O-GlcNAcylation and phosphorylation: roles in signaling, transcription, and chronic disease. Annu Rev Biochem 80:825–858. doi:10.1146/annurev-biochem-060608-102511
Hayakawa K et al (2013) Epigenetic switching by the metabolism-sensing factors in the generation of orexin neurons from mouse embryonic stem cells. J Biol Chem 288:17099–17110. doi:10.1074/jbc.M113.455899
Heckel D, Comtesse N, Brass N, Blin N, Zang KD, Meese E (1998) Novel immunogenic antigen homologous to hyaluronidase in meningioma. Hum Mol Genet 7:1859–1872
Holt GD, Hart GW (1986) The subcellular distribution of terminal N-acetylglucosamine moieties. Localization of a novel protein-saccharide linkage, O-linked GlcNAc. J Biol Chem 261:8049–8057
Housley MP, Udeshi ND, Rodgers JT, Shabanowitz J, Puigserver P, Hunt DF, Hart GW (2009) A PGC-1alpha-O-GlcNAc transferase complex regulates FoxO transcription factor activity in response to glucose. J Biol Chem 284:5148–5157. doi:10.1074/jbc.M808890200
Howerton CL, Morgan CP, Fischer DB, Bale TL (2013) O-GlcNAc transferase (OGT) as a placental biomarker of maternal stress and reprogramming of CNS gene transcription in development. Proc Natl Acad Sci USA 110:5169–5174. doi:10.1073/pnas.1300065110
Hu Y et al (2009) Increased enzymatic O-GlcNAcylation of mitochondrial proteins impairs mitochondrial function in cardiac myocytes exposed to high glucose. J Biol Chem 284:547–555. doi:10.1074/jbc.M808518200
Iyer SP, Hart GW (2003) Roles of the tetratricopeptide repeat domain in O-GlcNAc transferase targeting and protein substrate specificity. J Biol Chem 278:24608–24616. doi:10.1074/jbc.M300036200
Iyer SP, Akimoto Y, Hart GW (2003) Identification and cloning of a novel family of coiled-coil domain proteins that interact with O-GlcNAc transferase. J Biol Chem 278:5399–5409. doi:10.1074/jbc.M209384200
Ji S, Park SY, Roth J, Kim HS, Cho JW (2012) O-GlcNAc modification of PPARgamma reduces its transcriptional activity. Biochem Biophys Res Commun 417:1158–1163. doi:10.1016/j.bbrc.2011.12.086
Jinek M, Rehwinkel J, Lazarus BD, Izaurralde E, Hanover JA, Conti E (2004) The superhelical TPR-repeat domain of O-linked GlcNAc transferase exhibits structural similarities to importin alpha. Nat Struct Mol Biol 11:1001–1007. doi:10.1038/nsmb833
Jones SP et al (2008) Cardioprotection by N-acetylglucosamine linkage to cellular proteins. Circulation 117:1172–1182. doi:10.1161/circulationaha.107.730515
Kaasik K et al (2013) Glucose sensor O-GlcNAcylation coordinates with phosphorylation to regulate circadian clock. Cell Metab 17:291–302. doi:10.1016/j.cmet.2012.12.017
Kazemi Z, Chang H, Haserodt S, McKen C, Zachara NE (2010) O-linked beta-N-acetylglucosamine (O-GlcNAc) regulates stress-induced heat shock protein expression in a GSK-3beta-dependent manner. J Biol Chem 285:39096–39107. doi:10.1074/jbc.M110.131102
Keembiyehetty CN, Krzeslak A, Love DC, Hanover JA (2011) A lipid-droplet-targeted O-GlcNAcase isoform is a key regulator of the proteasome. J Cell Sci 124:2851–2860. doi:10.1242/jcs.083287
Kelly WG, Dahmus ME, Hart GW (1993) RNA polymerase II is a glycoprotein. Modification of the COOH-terminal domain by O-GlcNAc. J Biol Chem 268:10416–10424
Khidekel N et al (2007) Probing the dynamics of O-GlcNAc glycosylation in the brain using quantitative proteomics. Nat Chem Biol 3:339–348. doi:10.1038/nchembio881
Kreppel LK, Hart GW (1999) Regulation of a cytosolic and nuclear O-GlcNAc transferase. Role of the tetratricopeptide repeats. J Biol Chem 274:32015–32022
Kreppel LK, Blomberg MA, Hart GW (1997) Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats. J Biol Chem 272:9308–9315
Lazarus BD, Love DC, Hanover JA (2006) Recombinant O-GlcNAc transferase isoforms: identification of O-GlcNAcase, yes tyrosine kinase, and tau as isoform-specific substrates. Glycobiology 16:415–421. doi:10.1093/glycob/cwj078
Lazarus MB, Nam Y, Jiang J, Sliz P, Walker S (2011) Structure of human O-GlcNAc transferase and its complex with a peptide substrate. Nature 469:564–567. doi:10.1038/nature09638
Lazarus MB et al (2013) HCF-1 is cleaved in the active site of O-GlcNAc transferase. Science (New York, NY) 342:1235–1239. doi:10.1126/science.1243990
Lechner M, Lirk P, Rieder J (2005) Inducible nitric oxide synthase (iNOS) in tumor biology: the two sides of the same coin. Semin Cancer Biol 15:277–289. doi:10.1016/j.semcancer.2005.04.004
Lefebvre T, Baert F, Bodart JF, Flament S, Michalski JC, Vilain JP (2004) Modulation of O-GlcNAc glycosylation during Xenopus oocyte maturation. J Cell Biochem 93:999–1010. doi:10.1002/jcb.20242
Lehman DM et al (2005) A single nucleotide polymorphism in MGEA5 encoding O-GlcNAc-selective N-acetyl-beta-D glucosaminidase is associated with type 2 diabetes in Mexican Americans. Diabetes 54:1214–1221
Li MD et al (2012) O-GlcNAc transferase is involved in glucocorticoid receptor-mediated transrepression. J Biol Chem 287:12904–12912. doi:10.1074/jbc.M111.303792
Licastro F, Candore G, Lio D, Porcellini E, Colonna-Romano G, Franceschi C, Caruso C (2005) Innate immunity and inflammation in ageing: a key for understanding age-related diseases. Immunity and ageing: I and A 2:8. doi:10.1186/1742-4933-2-8
Liu X, Li L, Wang Y, Yan H, Ma X, Wang PG, Zhang L (2014) A peptide panel investigation reveals the acceptor specificity of O-GlcNAc transferase. FASEB J. doi:10.1096/fj.13-246850
Lopez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153:1194–1217. doi:10.1016/j.cell.2013.05.039
Love DC, Hanover JA (2005) The hexosamine signaling pathway: deciphering the “O-GlcNAc code”. Science’s STKE : signal transduction knowledge environment 2005:re13. doi:10.1126/stke.3122005re13
Love DC, Kochan J, Cathey RL, Shin SH, Hanover JA (2003) Mitochondrial and nucleocytoplasmic targeting of O-linked GlcNAc transferase. J Cell Sci 116:647–654
Love DC et al (2010a) Dynamic O-GlcNAc cycling at promoters of Caenorhabditis elegans genes regulating longevity, stress, and immunity. Proc Natl Acad Sci USA 107:7413–7418. doi:10.1073/pnas.0911857107
Love DC, Krause MW, Hanover JA (2010b) O-GlcNAc cycling: emerging roles in development and epigenetics. Semin Cell Dev Biol 21:646–654. doi:10.1016/j.semcdb.2010.05.001
Lubas WA, Hanover JA (2000) Functional expression of O-linked GlcNAc transferase. Domain structure and substrate specificity. J Biol Chem 275:10983–10988
Lubas WA, Frank DW, Krause M, Hanover JA (1997) O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats. J Biol Chem 272:9316–9324
Ma J, Hart GW (2013) Protein O-GlcNAcylation in diabetes and diabetic complications. Expert Rev proteomics 10:365–380. doi:10.1586/14789450.2013.820536
Ma J, Hart GW (2014) O-GlcNAc profiling: from proteins to proteomes. Clin Proteomics 11:8. doi:10.1186/1559-0275-11-8
Ma Z, Vosseller K (2013) O-GlcNAc in cancer biology. Amino acids 45:719–733. doi:10.1007/s00726-013-1543-8
Makino A, Suarez J, Gawlowski T, Han W, Wang H, Scott BT, Dillmann WH (2011) Regulation of mitochondrial morphology and function by O-GlcNAcylation in neonatal cardiac myocytes. Am J Physiol Regul Integr Comp Physiol 300:R1296–R1302. doi:10.1152/ajpregu.00437.2010
Marshall S, Bacote V, Traxinger RR (1991) Discovery of a metabolic pathway mediating glucose-induced desensitization of the glucose transport system. Role of hexosamine biosynthesis in the induction of insulin resistance. J Biol Chem 266:4706–4712
Marz P et al (2006) Ataxin-10 interacts with O-linked beta-N-acetylglucosamine transferase in the brain. J Biol Chem 281:20263–20270. doi:10.1074/jbc.M601563200
Mazars R et al (2010) The THAP-zinc finger protein THAP1 associates with coactivator HCF-1 and O-GlcNAc transferase: a link between DYT6 and DYT3 dystonias. J Biol Chem 285:13364–13371. doi:10.1074/jbc.M109.072579
Myers SA, Panning B, Burlingame AL (2011) Polycomb repressive complex 2 is necessary for the normal site-specific O-GlcNAc distribution in mouse embryonic stem cells. Proc Natl Acad Sci USA 108:9490–9495. doi:10.1073/pnas.1019289108
Nagel AK, Schilling M, Comte-Walters S, Berkaw MN, Ball LE (2013) Identification of O-linked N-acetylglucosamine (O-GlcNAc)-modified osteoblast proteins by electron transfer dissociation tandem mass spectrometry reveals proteins critical for bone formation. MCP 12:945–955. doi:10.1074/mcp.M112.026633
Ngoh GA, Watson LJ, Facundo HT, Dillmann W, Jones SP (2008) Non-canonical glycosyltransferase modulates post-hypoxic cardiac myocyte death and mitochondrial permeability transition. J Mol Cell Cardiol 45:313–325. doi:10.1016/j.yjmcc.2008.04.009
Nolte D, Muller U (2002) Human O-GlcNAc transferase (OGT): genomic structure, analysis of splice variants, fine mapping in Xq13.1. Mamm Gen: Off J Intern Mamm Gen Soc 13:62–64
O’Donnell N, Zachara NE, Hart GW, Marth JD (2004) Ogt-dependent X-chromosome-linked protein glycosylation is a requisite modification in somatic cell function and embryo viability. Mol Cell Biol 24:1680–1690
Ogawa M, Mizofuchi H, Kobayashi Y, Tsuzuki G, Yamamoto M, Wada S, Kamemura K (2012) Terminal differentiation program of skeletal myogenesis is negatively regulated by O-GlcNAc glycosylation. Biochim Biophys Acta 1820:24–32. doi:10.1016/j.bbagen.2011.10.011
Ortiz-Meoz RF, Merbl Y, Kirschner MW, Walker S (2014) Microarray discovery of new OGT substrates: the medulloblastoma oncogene OTX2 is O-GlcNAcylated. J Am Chem Soc 136:4845–4848. doi:10.1021/ja500451w
Rao FV, Dorfmueller HC, Villa F, Allwood M, Eggleston IM, van Aalten DM (2006) Structural insights into the mechanism and inhibition of eukaryotic O-GlcNAc hydrolysis. EMBO J 25:1569–1578. doi:10.1038/sj.emboj.7601026
Rao FV, Schuttelkopf AW, Dorfmueller HC, Ferenbach AT, Navratilova I, van Aalten DM (2013) Structure of a bacterial putative acetyltransferase defines the fold of the human O-GlcNAcase C-terminal domain. Open biology 3:130021. doi:10.1098/rsob.130021
Ryu IH, Do SI (2011) Denitrosylation of S-nitrosylated OGT is triggered in LPS-stimulated innate immune response. Biochem Biophys Res Commun 408:52–57. doi:10.1016/j.bbrc.2011.03.115
Sakabe K, Wang Z, Hart GW (2010) Beta-N-acetylglucosamine (O-GlcNAc) is part of the histone code. Proc Natl Acad Sci USA 107:19915–19920. doi:10.1073/pnas.1009023107
Sakaidani Y et al (2011) O-linked-N-acetylglucosamine on extracellular protein domains mediates epithelial cell-matrix interactions. Nature Commun 2:583. doi:10.1038/ncomms1591
Sakaidani Y et al (2012) O-linked-N-acetylglucosamine modification of mammalian Notch receptors by an atypical O-GlcNAc transferase Eogt1. Biochem Biophys Res Commun 419:14–19. doi:10.1016/j.bbrc.2012.01.098
Shafi R et al (2000) The O-GlcNAc transferase gene resides on the X chromosome and is essential for embryonic stem cell viability and mouse ontogeny. Proc Natl Acad Sci USA 97:5735–5739. doi:10.1073/pnas.100471497
Shin SH, Love DC, Hanover JA (2011) Elevated O-GlcNAc-dependent signaling through inducible mOGT expression selectively triggers apoptosis. Amino Acids 40:885–893. doi:10.1007/s00726-010-0719-8
Sinclair DA et al (2009) Drosophila O-GlcNAc transferase (OGT) is encoded by the Polycomb group (PcG) gene, super sex combs (sxc). Proc Natl Acad Sci USA 106:13427–13432. doi:10.1073/pnas.0904638106
Singh JP, Zhang K, Wu J, Yang X (2014) O-GlcNAc signaling in cancer metabolism and epigenetics. Cancer Lett. doi:10.1016/j.canlet.2014.04.014
Slawson C, Zachara NE, Vosseller K, Cheung WD, Lane MD, Hart GW (2005) Perturbations in O-linked beta-N-acetylglucosamine protein modification cause severe defects in mitotic progression and cytokinesis. J Biol Chem 280:32944–32956. doi:10.1074/jbc.M503396200
Slawson C, Lakshmanan T, Knapp S, Hart GW (2008) A mitotic GlcNAcylation/phosphorylation signaling complex alters the posttranslational state of the cytoskeletal protein vimentin. Mol Biol Cell 19:4130–4140. doi:10.1091/mbc.E07-11-1146
Sohn KC, Do SI (2005) Transcriptional regulation and O-GlcNAcylation activity of zebrafish OGT during embryogenesis. Biochem Biophys Res Commun 337:256–263. doi:10.1016/j.bbrc.2005.09.049
Solary E, Bernard OA, Tefferi A, Fuks F, Vainchenker W (2014) The Ten-Eleven Translocation-2 (TET2) gene in hematopoiesis and hematopoietic diseases. Leukemia 28:485–496. doi:10.1038/leu.2013.337
Steentoft C et al (2013) Precision mapping of the human O-GalNAc glycoproteome through SimpleCell technology. EMBO J 32:1478–1488. doi:10.1038/emboj.2013.79
Tai HC, Khidekel N, Ficarro SB, Peters EC, Hsieh-Wilson LC (2004) Parallel identification of O-GlcNAc-modified proteins from cell lysates. J Am Chem Soc 126:10500–10501. doi:10.1021/ja047872b
Tan EP et al (2014) Altering O-linked beta-N-Acetylglucosamine cycling disrupts mitochondrial function. J Biol Chem. doi:10.1074/jbc.M113.525790
Toleman C, Paterson AJ, Whisenhunt TR, Kudlow JE (2004) Characterization of the histone acetyltransferase (HAT) domain of a bifunctional protein with activable O-GlcNAcase and HAT activities. J Biol Chem 279:53665–53673. doi:10.1074/jbc.M410406200
Tonks NK (2006) Protein tyrosine phosphatases: from genes, to function, to disease. Nat Rev Mol Cell Biol 7:833–846. doi:10.1038/nrm2039
Trinidad JC, Barkan DT, Gulledge BF, Thalhammer A, Sali A, Schoepfer R, Burlingame AL (2012) Global identification and characterization of both O-GlcNAcylation and phosphorylation at the murine synapse. MCP 11:215–229. doi:10.1074/mcp.O112.018366
Vaidyanathan K, Durning S, Wells L (2014) Functional O-GlcNAc modifications: implications in molecular regulation and pathophysiology. Crit Rev Biochem Mol Biol 49:140–163. doi:10.3109/10409238.2014.884535
Vella P et al (2013) Tet proteins connect the O-linked N-acetylglucosamine transferase Ogt to chromatin in embryonic stem cells. Mol Cell 49:645–656. doi:10.1016/j.molcel.2012.12.019
Vosseller K et al (2006) O-linked N-acetylglucosamine proteomics of postsynaptic density preparations using lectin weak affinity chromatography and mass spectrometry. MCP 5:923–934. doi:10.1074/mcp.T500040-MCP200
Wang Z, Pandey A, Hart GW (2007) Dynamic interplay between O-linked N-acetylglucosaminylation and glycogen synthase kinase-3-dependent phosphorylation. MCP 6:1365–1379. doi:10.1074/mcp.M600453-MCP200
Wang Z, Udeshi ND, O’Malley M, Shabanowitz J, Hunt DF, Hart GW (2010a) Enrichment and site mapping of O-linked N-acetylglucosamine by a combination of chemical/enzymatic tagging, photochemical cleavage, and electron transfer dissociation mass spectrometry. MCP 9:153–160. doi:10.1074/mcp.M900268-MCP200
Wang Z et al (2010b) Extensive crosstalk between O-GlcNAcylation and phosphorylation regulates cytokinesis. Sci Signal 3: ra2. doi:10.1126/scisignal.2000526
Webster DM et al (2009) O-GlcNAc modifications regulate cell survival and epiboly during zebrafish development. BMC Dev Biol 9:28. doi:10.1186/1471-213x-9-28
Wells L, Gao Y, Mahoney JA, Vosseller K, Chen C, Rosen A, Hart GW (2002) Dynamic O-glycosylation of nuclear and cytosolic proteins: further characterization of the nucleocytoplasmic beta-N-acetylglucosaminidase, O-GlcNAcase. J Biol Chem 277:1755–1761
Whelan SA, Lane MD, Hart GW (2008) Regulation of the O-linked beta-N-acetylglucosamine transferase by insulin signaling. J Biol Chem 283:21411–21417. doi:10.1074/jbc.M800677200
Whisenhunt TR, Yang X, Bowe DB, Paterson AJ, Van Tine BA, Kudlow JE (2006) Disrupting the enzyme complex regulating O-GlcNAcylation blocks signaling and development. Glycobiology 16:551–563. doi:10.1093/glycob/cwj096
Yang X, Zhang F, Kudlow JE (2002) Recruitment of O-GlcNAc transferase to promoters by corepressor mSin3A: coupling protein O-GlcNAcylation to transcriptional repression. Cell 110:69–80
Yang X et al (2008) Phosphoinositide signalling links O-GlcNAc transferase to insulin resistance. Nature 451:964–969. doi:10.1038/nature06668
Yang YR et al (2012) O-GlcNAcase is essential for embryonic development and maintenance of genomic stability. Aging Cell 11:439–448. doi:10.1111/j.1474-9726.2012.00801.x
Yi W et al (2012) Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science (New York, NY) 337:975–980. doi:10.1126/science.1222278
Yuzwa SA, Vocadlo DJ (2014) O-GlcNAc and neurodegeneration: biochemical mechanisms and potential roles in Alzheimer’s disease and beyond. Chem Soc Rev. doi:10.1039/c4cs00038b
Zachara NE, Hart GW (2004) O-GlcNAc a sensor of cellular state: the role of nucleocytoplasmic glycosylation in modulating cellular function in response to nutrition and stress. Biochim Biophys Acta 1673:13–28. doi:10.1016/j.bbagen.2004.03.016
Zachara NE, O’Donnell N, Cheung WD, Mercer JJ, Marth JD, Hart GW (2004) Dynamic O-GlcNAc modification of nucleocytoplasmic proteins in response to stress. A survival response of mammalian cells. J Biol Chem 279:30133–30142. doi:10.1074/jbc.M403773200
Zeidan Q, Hart GW (2010) The intersections between O-GlcNAcylation and phosphorylation: implications for multiple signaling pathways. J Cell Sci 123:13–22. doi:10.1242/jcs.053678
Conflict of interest
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagel, A.K., Ball, L.E. O-GlcNAc transferase and O-GlcNAcase: achieving target substrate specificity. Amino Acids 46, 2305–2316 (2014). https://doi.org/10.1007/s00726-014-1827-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-014-1827-7