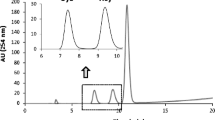
Abstract
Homocysteine (Hcy) is incorporated into protein via a reaction of the thioester Hcy-thiolactone with ε-amino group of a protein lysine residue generating N-Hcy-protein. This reaction impairs and alters protein’s function and has been implicated in atherothrombotic disease. Here, we describe new high-performance liquid chromatography assays for the determination of Hcy-thiolactone, protein N-linked Hcy, and Hcy based on an on-column derivatization with o-phthaldialdehyde and fluorescence detection. The on-column derivatization generates narrow peaks, which allows fast run times (3–5 min) and facilitates determination of N-linked Hcy directly from acid hydrolysates of plasma protein. Utility of these assays was demonstrated with human urine and plasma samples.





Similar content being viewed by others
Abbreviations
- Hcy:
-
Homocysteine
- tHcy:
-
Total Hcy, i.e., free Hcy present in a sample after reductive cleavage of disulfide bonds
- (Hcy)2 :
-
Homocystine
- N-Hcy-protein:
-
N-homocysteinylated protein
- protein N-linked Hcy:
-
Hcy bound to protein by an amide linkage to ε-amino group of lysine residue(s)
- OPA:
-
o-phthaldialdehyde
- SPE:
-
Solid phase extraction
- CMQT:
-
2-chloro-1-methylquinolinium tetrafluoroborate
- TCEP:
-
Tris(2-carboxyethyl)phosphine
- CBS:
-
Cystathionine β-synthase
- LOQ:
-
Lower limit of quantification
References
Bald E, Głowacki R (2001) 2-Chloro-1-methylquinolinium tetrafluoroborate as effective and thiol specific UV-tagging reagent for liquid chromatography. J Liq Chromatogr Rel Techn 24:1323–1339
Bald E, Głowacki R (2005) Analysis of saliva for glutathione and metabolically related thiols by liquid chromatography with ultraviolet detection. Amino Acids 28:431–433
Bald E, Chwatko G, Głowacki R, Kusmierek K (2004) Analysis of plasma thiols by high-performance liquid chromatography with ultraviolet detection. J Chromatogr A 1032:109–115
Chwatko G, Jakubowski H (2005a) Urinary excretion of homocysteine-thiolactone in humans. Clin Chem 51:408–415
Chwatko G, Jakubowski H (2005b) The determination of homocysteine-thiolactone in human plasma. Anal Biochem 337:271–277
Chwatko G, Boers GH, Strauss KA, Shih DM, Jakubowski H (2007) Mutations in methylenetetrahydrofolate reductase or cystathionine beta-synthase gene, or a high-methionine diet, increase homocysteine thiolactone levels in humans and mice. Faseb J 21:1707–1713
Daneshvar P, Yazdanpanah M, Cuthbert C, Cole DE (2003) Quantitative assay of plasma homocysteine thiolactone by gas chromatography/mass spectrometry. Rapid Commun Mass Spectrom 17:358–362
Głowacki R, Bald E (2009) Determination of N-acetylcysteine and main endogenous thiols in human plasma by HPLC with ultraviolet detection in the form of their S-quinolinium derivatives. J Liq Chromatogr Rel Techn 32:2530–2544
Głowacki R, Jakubowski H (2004) Cross-talk between Cys34 and lysine residues in human serum albumin revealed by N-homocysteinylation. J Biol Chem 279:10864–10871
Głowacki R, Borowczyk K, Bald E, Jakubowski H (2010) On-column derivatization with o-phthaldialdehyde for fast determination of homocysteine in human urine. Anal Bioanal Chem (in press). doi:10.1007/s00216-010-3456-7
Gu W, Lu J, Yang G, Dou J, Mu Y, Meng J, Pan C (2008) Plasma homocysteine thiolactone associated with risk of macrovasculopathy in Chinese patients with type 2 diabetes mellitus. Adv Ther 25:914–924
Jakubowski H (1990) Proofreading in vivo: editing of homocysteine by methionyl-tRNA synthetase in Escherichia coli. Proc Natl Acad Sci USA 87:4504–4508
Jakubowski H (1997) Metabolism of homocysteine thiolactone in human cell cultures: possible mechanism for pathological consequences of elevated homocysteine levels. J Biol Chem 272:1935–1942
Jakubowski H (1999) Protein homocysteinylation: possible mechanism underlying pathological consequences of elevated homocysteine levels. Faseb J 13:2277–2283
Jakubowski H (2000) Homocysteine thiolactone: metabolic origin and protein homocysteinylation in humans. J Nutr 130:377S–381S
Jakubowski H (2001a) Translational accuracy of aminoacyl-tRNA synthetases: implications for atherosclerosis. J Nutr 131:2983S–2987S
Jakubowski H (2001b) Protein N-homocysteinylation: implications for atherosclerosis. Biomed Pharmacother 55:443–447
Jakubowski H (2002a) Homocysteine is a protein amino acid in humans. Implications for homocysteine-linked disease. J Biol Chem 277:30425–30428
Jakubowski H (2002b) The determination of homocysteine-thiolactone in biological samples. Anal Biochem 308:112–119
Jakubowski H (2004) Molecular basis of homocysteine toxicity in humans. Cell Mol Life Sci 61:470–487
Jakubowski H (2005) Anti-N-homocysteinylated protein autoantibodies and cardiovascular disease. Clin Chem Lab Med 43:1011–1014
Jakubowski H (2006a) Pathophysiological consequences of homocysteine excess. J Nutr 136:1741S–1749S
Jakubowski H (2006b) Mechanism of the condensation of homocysteine thiolactone with aldehydes. Chem Eur J 12:8039–8043
Jakubowski H (2007) The molecular basis of homocysteine thiolactone-mediated vascular disease. Clin Chem Lab Med 45:1704–1716
Jakubowski H (2008a) The pathophysiological hypothesis of homocysteine thiolactone-mediated vascular disease. J Physiol Pharmacol 59(suppl 9):155–167
Jakubowski H (2008b) New method for the determination of protein N-linked homocysteine. Anal Biochem 380:257–261
Jakubowski H, Goldman E (1993) Synthesis of homocysteine thiolactone by methionyl-tRNA synthetase in cultured mammalian cells. FEBS Lett 317:237–240
Jakubowski H, Boers GH, Strauss KA (2008) Mutations in cystathionine {beta}-synthase or methylenetetrahydrofolate reductase gene increase N-homocysteinylated protein levels in humans. FASEB J 22:4071–4076
Jakubowski H, Perla-Kajan J, Finnell RH, Cabrera RM, Wang H, Gupta S, Kruger WD, Kraus JP, Shih DM (2009) Genetic or nutritional disorders in homocysteine or folate metabolism increase protein N-homocysteinylation in mice. FASEB J 23:1721–1727
Lentz SR (2005) Mechanisms of homocysteine-induced atherothrombosis. J Thromb Haemost 3:1646–1654
Mukai Y, Togawa T, Suzuki T, Ohata K, Tanabe S (2002) Determination of homocysteine thiolactone and homocysteine in cell cultures using high-performance liquid chromatography with fluorescence detection. J Chromatogr B Analyt Technol Biomed Life Sci 767:263–268
Perla-Kajan J, Twardowski T, Jakubowski H (2007) Mechanisms of homocysteine toxicity in humans. Amino Acids 32:561–572
Perla-Kajan J, Stanger O, Luczak M, Ziolkowska A, Malendowicz LM, Twardowski T, Lhotak S, Austin RC, Jakubowski H (2008) Immunohistochemical detection of N-homocysteinylated proteins in humans and mice. Biomed Pharmacother 62:473–479
Perna AF, Satta E, Acanfora F, Lombardi C, Ingrosso D, De Santo NG (2006) Increased plasma protein homocysteinylation in hemodialysis patients. Kidney Int 69:869–876
Uji Y, Motomiya Y, Hanyu N, Ukaji F, Okabe H (2002) Protein-bound homocystamide measured in human plasma by HPLC. Clin Chem 48:941–944
Yang X, Gao Y, Zhou J, Zhen Y, Yang Y, Wang J, Song L, Liu Y, Xu H, Chen Z, Hui R (2006) Plasma homocysteine thiolactone adducts associated with risk of coronary heart disease. Clin Chim Acta 364:230–234
Acknowledgments
This work was supported in part by grants from the American Heart Association, the University of Łódź, and the Ministry of Science and Higher Education, Poland (NN 401 230634, N401 065 32/1504, POIG.01.03.01-00-097/08).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Głowacki, R., Bald, E. & Jakubowski, H. An on-column derivatization method for the determination of homocysteine-thiolactone and protein N-linked homocysteine. Amino Acids 41, 187–194 (2011). https://doi.org/10.1007/s00726-010-0521-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0521-7