Abstract
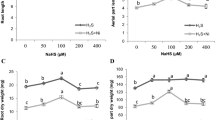
Impact of various nitrate concentrations (14.12 mM, 3.53 mM, no nitrate) or ammonium presence (14.12 mM) on physiological and metabolic changes in Hypericum perforatum after 14 days of cultivation was monitored. Nitrate deficiency suppressed growth of shoots but stimulated root growth while ammonium suppressed root growth: concomitant changes of ascorbic acid and glutathione supported these growth changes, e.g., unaltered level in roots under nitrate deficiency but depleted in ammonium treatment. Soluble proteins and water content were more suppressed by nitrate deficiency but total ROS, nitric oxide formation, and antioxidative enzyme activities (APX and SOD) indicate higher sensitivity of plants to ammonium. Though both extreme treatments (NO3− deficiency or ammonium) stimulated accumulation of total soluble phenols and affected PAL activity (in comparison with full or 1/4× nitrate dose), major phenols (chlorogenic acid and three flavonoids) were elevated mainly by NO3– deficiency. At the level of specific metabolites, NO3– deficiency had stimulatory impact on pseudohypericin (but not hypericin) content while hyperforin decreased. Expression of earlier putative gene of hypericin biosynthesis (hyp-1) showed rather partial correlation with pseudohypericin amount. Data indicate that depletion of NO3– is useful to obtain Hypericum plants with higher amount of health-positive secondary metabolites.





Similar content being viewed by others
References
Adavi SB, Sathee L (2019) Elevated CO2-induced production of nitric oxide differentially modulates nitrate assimilation and root growth of wheat seedlings in a nitrate dose-dependent manner. Protoplasma 256:147–159
Babula P, Klejdus B, Kováčik J, Hedbavny J, Hlavna M (2015) Lanthanum rather than cadmium induces oxidative stress and metabolite changes in Hypericum perforatum. J Hazard Mater 286:334–342
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Briskin DP, Leroy A, Gawienowski MC (2000) Influence of nitrogen on the production of hypericins by St. John’s worth. Plant Physiol Biochem 38:413–420
Cui X-H, Murthy HN, Wo C-H, Paek K-Y (2010) Adventitious root suspension cultures of Hypericum perforatum: effect of nitrogen source on production of biomass and secondary metabolites. In Vitro Cell Dev Biol Plant 46:437–444
Dakora FD, Phillips DA (2002) Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant Soil 245:35–47
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7:1085–1097
Dresler S, Kováčik J, Strzemski M, Sowa I, Wójciak-Kosior M (2018) Methodological aspects of biologically active compounds quantification in the genus Hypericum. J Pharm Biomed Anal 155:82–90
Ganie AH, Ahmad A, Yousuf PY, Pandey R, Ahmad S, Aref IM (2017) Nitrogen-regulated changes in total amino acid profile of maize genotypes having contrasting response to nitrogen deficit. Protoplasma 254:2143–2153
Jozefczak M, Bohler S, Schat H, Horemans N, Guisez Y, Remans T, Vangronsveld J, Cuypers A (2015) Both the concentration and redox state of glutathione and ascorbate influence the sensitivity of Arabidopsis to cadmium. Ann Bot 116:601–612
Juszczuk IM, Wiktorowska A, Malusá E, Rychter AM (2004) Changes in the concentration of phenolic compounds and exudation induced by phosphate deficiency in bean plants (Phaseolus vulgaris L.). Plant Soil 267:41–49
Košuth J, Smelcerovic A, Borsch T, Zuehlke S, Karppinen K, Spiteller M, Hohtola A, Čellárová E (2011) The hyp-1 gene is not a limiting factor for hypericin biosynthesis in the genus Hypericum. Funct Plant Biol 38:35–43
Kováčik J, Bačkor M (2007) Changes of phenolic metabolism and oxidative status in nitrogen-deficient Matricaria chamomilla plants. Plant Soil 297:255–265
Kováčik J, Klejdus B (2014) Induction of phenolic metabolites and physiological changes in chamomile plants in relation to nitrogen nutrition. Food Chem 142:334–341
Kováčik J, Klejdus B, Babula P, Jarošová M (2014) Variation of antioxidants and secondary metabolites in nitrogen-deficient barley plants. J Plant Physiol 171:260–268
Kováčik J, Babula P, Hedbavny J (2017) Comparison of vascular and non-vascular aquatic plant as indicators of cadmium toxicity. Chemosphere 180:86–92
Kováčik J, Dresler S, Micalizzi G, Babula P, Hladký J, Mondello L (2019) Nitric oxide affects cadmium-induced changes in the lichen Ramalina farinacea. Nitric Oxide 83:11–18
Lin Y-L, Chao Y-Y, Huang W-D, Kao CH (2011) Effect of nitrogen deficiency on antioxidant status and Cd toxicity in rice seedlings. Plant Growth Regul 64:263–273
Liszkay A, van der Zalm E, Schopfer P (2004) Production of reactive oxygen intermediates (O2.−, H2O2 and .OH) by maize roots and their role in wall loosening and elongation growth. Plant Physiol 136:3114–3123
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Medina-Pérez V, López-Laredo AR, Sepúlveda-Jiménez G, Zamilpa A, Trejo-Tapia G (2015) Nitrogen deficiency stimulates biosynthesis of bioactive phenylethanoid glycosides in the medicinal plant Castilleja tenuiflora Benth. Acta Physiol Plant 37:93
Rice-Evans CA, Miller NJ, Paganga G (1996) Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med 20:933–956
Shin R, Berg RH, Schachtman DP (2005) Reactive oxygen species and root hairs in Arabidopsis root response to nitrogen, phosphorus and potassium deficiency. Plant Cell Physiol 46:1350–1357
Sliwiak J, Dauter Z, Jaskolski M (2016) Crystal structure of hyp-1, a Hypericum perforatum PR-10 protein, in complex with melatonin. Front Plant Sci 7:668. https://doi.org/10.3389/fpls.2016.00668
Wang JW, Zheng LP, Wu JY, Tan RX (2006) Involvement of nitric oxide in oxidative burst, phenylalanine ammonia-lyase activation and taxol production induced by low-energy ultrasound in Taxus yunnanensis cell suspension cultures. Nitric Oxide 15:351–358
Zhang X, Yu HJ, Zhang XM, Yang XY, Zhao WC, Li Q, Jiang WJ (2016) Effect of nitrogen deficiency on ascorbic acid biosynthesis and recycling pathway in cucumber seedlings. Plant Physiol Biochem 108:222–230
Funding
The work was supported by Slovak grant agency VEGA (project no. 1/0041/18).
Role of the funding sources
Sponsor had no involvement in the present study.
Author information
Authors and Affiliations
Contributions
Plant cultivation and spectrophotometry (JK and VP), experimental design and manuscript preparation (JK), chromatography and other methods (SD), fluorescence microscopy (PB).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Handling Editor: Peter Nick
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kováčik, J., Dresler, S., Peterková, V. et al. Nitrogen nutrition modulates oxidative stress and metabolite production in Hypericum perforatum. Protoplasma 257, 439–447 (2020). https://doi.org/10.1007/s00709-019-01448-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-019-01448-1