Abstract
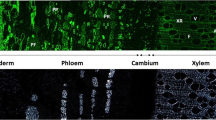
Under the effect of disturbances, like unbalanced stem, but also during normal development, poplar trees can develop a specific secondary xylem, called “tension wood” (TW), which is easily identifiable by the presence of a gelatinous layer in the secondary cell walls (SCW) of the xylem fibers. Since TW formation was mainly performed on 2-year-old poplar models, an in vitro poplar that produces gelatinous fibers (G-fibers) while offering the same experimental advantages as herbaceous plants has been developed. Using specific cell wall staining techniques, wood structural features and lignin/cellulose distribution were both detailed in cross-sections obtained from the curved stem part of in vitro poplars. A supposed delay in the SCW lignification process in the G-fibers, along with the presence of a G-layer, could be observed in the juvenile plants. Moreover, in this G-layer, the immunolabeling of various polymers carried out in the SCW of TW has allowed detecting crystalline cellulose, arabinogalactans proteins, and rhamnogalacturonans I; however, homogalacturonans, xylans, and xyloglucans could not be found. Interestingly, extensins were detected in this typical adaptative or stress-induced structure. These observations were corroborated by a quantitation of the immunorecognized polymer distribution using gold particle labeling. In conclusion, the in vitro poplar model seems highly convenient for TW studies focusing on the implementation of wall polymers that provide the cell wall with greater plasticity in adapting to the environment.








Similar content being viewed by others
References
Abedini R, Clair B, Pourtahmasi K, Laurans F, Arnould O (2015) Cell wall thickening in developing tension wood of artificially bent poplar trees. IAWA J 36:44–57. https://doi.org/10.1163/22941932-00000084
Abot A (2010) Caractérisation des fibres longues de chanvre (Cannabis sativa) en vue de leurs utilisations dans des matériaux composites. PhD dissertation, University of Poitiers - France
Alméras T, Clair B (2016) Critical review on the mechanisms of maturation stress generation in trees. J R Soc Interface 13(122):20160550 https://doi.org/10.1098/rsif.2016.0550
Altaner C, Hapca AI, Knox JP, Jarvis MC (2007) Detection of β-1-4-galactan in compression wood of Sitka spruce [Picea sitchensis (Bong.) Carrière] by immunofluorescence. Holzforschung 61(3):311–316. https://doi.org/10.1515/HF.2007.049
Andersson-Gunnerås S, Mellerowicz EJ, Love J, Segerman B, Ohmiya Y, Coutinho PM, Nilsson P, Henrissat B, Moritz T, Sundberg B (2006) Biosynthesis of cellulose-enriched tension wood in Populus: global analysis of transcripts and metabolites identifies biochemical and developmental regulators in secondary wall biosynthesis. Plant J 45:144–165. https://doi.org/10.1111/j.1365-313X.2005.02584.x
Araki N, Fujita M, Saiki H, Harada H (1983) Transition of fiber wall structure from normal wood to tension wood in certain species having gelatinous fibers of S1+G and S1+S2+S3+G types. J Japan Wood Res Soc 29:491–499
Arend M (2008) Immunolocalization of (1,4)-β-galactan in tension wood fibers of poplar. Tree Physiol 28:1263–1267
Baillères H, Chanson B, Fournier M, Tollier MT, Monties B (1995) Wood structure, chemical composition and growth strains in Eucalyptus clones. Ann Sci 52:157–172. https://doi.org/10.1051/forest:19950206
Baillères H, Castan M, Monties B, Pollet B, Lapierre C (1997) Lignin structure in Buxus sempervirens reaction wood. Phytochem 44:35–39. https://doi.org/10.1016/S0031-9422(96)00499-2
Barros J, Serk H, Granlund I, Pesquet E (2015) The cell biology of lignification in higher plants. Ann Bot 115(7):1053–1074. https://doi.org/10.1093/aob/mcv046
Bowling AJ, Vaughn KC (2008) Structural and immunocytochemical characterization of the adhesive tendril of Virginia creeper (Parthenocissus quinquefolia [L.] Planch.). Protoplasma 232:153–163. https://doi.org/10.1007/s00709-008-0287-x
Cai M, Huang H, Ni F, Tong Z, Lin E, Zhu M (2018) RNA-Seq analysis of differential gene expression in Betula luminifera xylem during the early stages of tension wood formation. PeerJ 6:e5427. https://doi.org/10.7717/peerj.5427
Chaffey N, Cholewa E, Regan S, Sundberg B (2002) Secondary xylem development in Arabidopsis: a model for wood formation. Physiol Plant 114:594–600. https://doi.org/10.1034/j.1399-3054.2002.1140413.x
Chang SS, Clair B, Ruelle J, Beauchêne J, Di Renzo F, Quignard F, Zhao GJ, Yamamoto H, Gril J (2009) Mesoporosity as a new parameter in understanding of tension stress generation in trees. J Exp Bot 60:3023–3030. https://doi.org/10.1093/jxb/erp133
Chang SS, Quignard F, Alméras T, Clair B (2015) Mesoporosity changes from cambium to mature tension wood: a new step toward the understanding of maturation stress generation in trees. New Phytol 205:1277–1287. https://doi.org/10.1111/nph.13126
Clair B, Thibaut B, Sugiyama J (2005) On the detachment of the gelatinous layer in tension wood fiber. J Wood Sci 51:218–221. https://doi.org/10.1007/s10086-004-0648-9
Clair B, Gril J, Di Renzo F, Yamamoto H, Quignard F (2008) Characterization of a gel in the cell wall to elucidate the paradoxical shrinkage of tension wood. Biomacromolecules 9:494–498. https://doi.org/10.1021/bm700987q
Clair B, Alméras T, Pilate G, Jullien D, Sugiyama J, Riekel C (2011) Maturation stress generation in poplar tension wood studied by synchrotron radiation microdiffraction. Plant Physiol 155:562–570. https://doi.org/10.1104/pp.110.167270
Clair B, Déjardin A, Pilate G, Alméras T (2018) Is the G-layer a tertiary cell wall? Front Plant Sci 9:623. https://doi.org/10.3389/fpls.2018.00623
Clair B, Ghislain B, Prunier J, Lehnebach R, Beauchêne J, Alméras T (2019) Mechanical contribution of secondary phloem to postural control in trees: the bark side of the force. New Phytol 221:209–217. https://doi.org/10.1111/nph.15375
Decou R (2009) Contribution à l'étude du rôle des xylanes et des xyloglucanes fucosylés au cours de la formation du bois : application à la formation du bois de tension chez le peuplier. PhD dissertation, University of Limoges - France
Decou R, Lhernould S, Laurans F, Sulpice E, Leplé JC, Déjardin A, Pilate G, Costa G (2009) Cloning and expression analysis of a wood-associated xylosidase gene (PtaBXL1) in poplar tension wood. Phytochem 70:163–172. https://doi.org/10.1016/j.phytochem.2008.12.019
Déjardin A, Leplé JC, Lesage-Descauses MC, Costa G, Pilate G (2004) Expressed sequence tags from poplar wood tissues - a comparative analysis from multiple libraries. Plant Biol 6:55–64. https://doi.org/10.1055/s-2003-44744
Fleurat-Lessard P, Bouché-Pillon S, Leloup C, Bonnemain JL (1997) Distribution and activity of the plasma membrane H+-ATPase in Mimosa pudica L. in relation to ionic fluxes and leaf movements. Plant Physiol 113:747–754
Fleurat-Lessard P, Bourbouloux A, Thibault F, Ménard E, Béré E, Valtaud C, Roblin G (2013) Differential occurrence of suberized sheaths in canes of grapevines suffering from black dead arm, esca or Eutypa dieback. Trees-Struct Funct 27(4):1087–1100. https://doi.org/10.1007/s00468-013-0859-z
Gorshkova T, Mokshina N, Chernova T, Ibragimova N, Salnikov V, Mikshina P, Tryfona T, Banasiak A, Immerzeel P, Dupree P, Mellerowicz EJ (2015) Aspen tension wood fibers contain β-(1→4)-galactans and acidic arabinogalactans retained by cellulose microfibrils in gelatinous walls. Plant Physiol 169:2048–2063. https://doi.org/10.1104/pp.15.00690
Gorzsás A, Stenlund H, Persson P, Trygg J, Sundberg B (2011) Cell-specific chemotyping and multivariate imaging by combined FT-IR microspectroscopy and orthogonal projections to latent structures (OPLS) analysis reveals the chemical landscape of secondary xylem. Plant J 66:903–914. https://doi.org/10.1111/j.1365-313X.2011.04542.x
Guedes FTP, Laurans F, Quemener B. Assor C, Lainé-Prade V, Boizot N, Vigouroux J, Lesage-Descauses MC, Leplé JC, Déjardin A, Pilate G (2017) Non-cellulosic polysaccharide distribution during G-layer formation in poplar tension wood fibers: abundance of rhamnogalacturonan I and arabinogalactan proteins but no evidence of xyloglucan. Planta 246(5):857–878. https://doi.org/10.1007/s00425-017-2737-1
Han KH (2001) Molecular biology of secondary growth. J Plant Biotech 3(2):45–57
His I, Andème-Onzighi C, Morvan C, Driouich A (2001) Microscopic studies on mature flax fibers embedded in LR white: immunogold localization of cell wall matrix polysaccharides. J Histochem Cytochem 49(12):1525–1535. https://doi.org/10.1177/002215540104901206
Hrmova M, Farkas V, Lahnstein J, Fincher GB (2007) A barley xyloglucan xyloglucosyl transferase covalently links xyloglucan, cellulosic substrates, and (1,3;1,4)-β-D-glucans. J Biol Chem 282:12951–12962. https://doi.org/10.1074/jbc.M611487200
Joseleau JP, Imai T, Kuroda K, Ruel K (2004) Detection in situ and characterization of lignin in the G-layer of tension wood fibres of Populus deltoides. Planta 219(2):338–345. https://doi.org/10.1007/s00425-004-1226-5
Jourez B (1997) Le bois de tension 2. Evaluation quantitative, formation et rôle dans l'arbre. Biotech Agronomy Soc Environ 1:167–177
Kaku T, Serada S, Baba KI, Tanaka F, Hayashi T (2009) Proteomic analysis of the G-layer in poplar tension wood. J Wood Sci 55(4):250–257. https://doi.org/10.1007/s10086-009-1032-6
Kumar M, Campbell L, Turner S (2016) Secondary cell walls: biosynthesis and manipulation. J Exp Bot 67(2):515–531. https://doi.org/10.1093/jxb/erv533
Lafarguette F, Leplé JC, Déjardin A, Laurans F, Costa G, Lesage-Descauses MC, Pilate G (2004) Poplar genes encoding fasciclin-like arabinogalactan proteins are highly expressed in tension wood. New Phytol 164:107–121. https://doi.org/10.1111/j.1469-8137.2004.01175.x
Lee KJ, Sakata Y, Mau SL, Pettolino F, Bacic A, Quatrano RS, Knight CD, Knox JP (2005) Arabinogalactan proteins are required for apical cell extension in the moss Physcomitrella patens. Plant Cell 17(11):3051–3065. https://doi.org/10.1105/tpc.105.034413
Li L, Cheng XF, Leshkevich J, Umezawa T, Harding SA, Chiang VL (2001) The last step of syringyl monolignol biosynthesis in angiosperms is regulated by a novel gene encoding sinapyl alcohol dehydrogenase. Plant Cell 13(7):1567–1586. https://doi.org/10.1105/TPC.010111
Li S, Zhang Y, Ding C, Gao X, Wang R, Mo W, lv F, Wang S, Liu L, Tang Z, Tian H, Zhang J, Zhang B, Huang Q, Lu M, Wuyun TN, Hu Z, Xia Y, Su X (2019) Proline-rich protein gene PdPRP regulates secondary wall formation in poplar. Plant Physiol 233:58–72. https://doi.org/10.1016/j.jplph.2018.12.007
Marcus SE, Verhertbruggen Y, Hervé C, Ordaz-Ortiz JJ, Farkas V, Pedersen H, Willats WGT, Knox JP (2008) Pectic homogalacturonan masks abundant sets of xyloglucan epitopes in plant cell walls. BMC Plant Biol 8(1):60. https://doi.org/10.1186/1471-2229-8-60
Mellerowicz EJ, Gorshkova TA (2012) Tensional stress generation in gelatinous fibres: a review and possible mechanism based on cell wall structure and composition. J Exp Bot 63:551–565. https://doi.org/10.1093/jxb/err339
Mellerowicz EJ, Baucher M, Sundberg B, Boerjan W (2001) Unravelling cell wall formation in the woody dicot stem. Plant Mol Biol 47:239–274. https://doi.org/10.1023/A:1010699919325
Mellerowicz EJ, Immerzeel P, Hayashi T (2008) Xyloglucan: the molecular muscle of trees. Ann Bot 102:659–665. https://doi.org/10.1093/aob/mcn170
Meloche C, Knox J, Vaughn K (2007) A cortical band of gelatinous fibers causes the coiling of redvine tendrils: a model based upon cytochemical and immunocytochemical studies. Planta 225:485–498. https://doi.org/10.1007/s00425-006-0363-4
Mitra PP, Loqué D (2014) Histochemical staining of Arabidopsis thaliana secondary cell wall elements. JoVE 87:51381. https://doi.org/10.3791/51381
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nishikubo N, Awano T, Banasiak A, Bourquin V, Ibatullin F, Funada R, Brumer H, Teeri TT, Hayashi T, Sundberg B, Mellerowicz EJ (2007) Xyloglucan endo-transglycosylase (XET) functions in gelatinous layers of tension wood fibers in poplar—a glimpse into the mechanism of the balancing act of trees. Plant Cell Physiol 48(6):843–855. https://doi.org/10.1093/pcp/pcm055
Onaka F (1949) Studies on compression and tension wood. Wood Res 1:1–88
Pattathil S, Avci U, Baldwin D, Swennes AG, McGill JA, Popper Z, Bootten T, Albert A, Davis RH, Chennareddy C, Dong R, O'Shea B, Rossi R, Leoff C, Freshour G, Narra R, O'Neil M, York WS, Hahn MG (2010) A Comprehensive Toolkit of Plant Cell Wall Glycan-Directed Monoclonal Antibodies. Plant Physiol 153(2):514-525
Pilate G, Déjardin A, Laurans F, Leplé JC (2004a) Tension wood as a model for functional genomics of wood formation. New Phytol 164:63–72. https://doi.org/10.1111/j.1469-8137.2004.01176.x
Pilate G, Chabbert B, Cathala B, Yoshinaga A, Leplé JC, Laurans F, Lapierre C, Ruel K (2004b) Lignification and tension wood. C R Biologies 327:889–901. https://doi.org/10.1016/j.crvi.2004.07.006
Prodhan AKMA, Ohtani J, Funada R, Abe H, Fukazawa K (1995) Ultrastructural investigation of tension wood fibre in Fraxinus mandshurica Rupr. var. japonica Maxim. Ann Bot 75:311–317. https://doi.org/10.1006/anbo.1995.1026
Qiu D, Wilson IW, Gan S, Washusen R, Moran GF, Southerton SG (2008) Gene expression in Eucalyptus branch wood with marked variation in cellulose microfibril orientation and lacking G-layers. New Phytol 179:94–103. https://doi.org/10.1111/j.1469-8137.2008.02439.x
Roussel JR, Clair B (2015) Evidence of the late lignification of the G-layer in Simarouba tension wood, to assist understanding how non-G-layer species produce tensile stress. Tree Physiol 35:1366–1377. https://doi.org/10.1093/treephys/tpv082
Ruelle J, Clair B, Beauchêne J, Prevost MF, Fournier M (2006) Tension wood and opposite wood in 21 tropical rain forest species 2. Comparison of some anatomical and ultrastructural criteria. IAWA J 27:341–376 https://hal.archives-ouvertes.fr/hal-00194915
Serk H, Gorzsás A, Tuominen H, Pesquet E (2015) Cooperative lignification of xylem tracheary elements. Plant Signal Behav 10(4):1–5. https://doi.org/10.1080/15592324.2014.1003753
Sundell D, Street NR, Kumar M, Mellerowicz EJ, Kucukoglu M, Johnsson C, Kumar V, Mannapperuma C, Delhomme N, Nilsson O, Tuominen H, Pesquet E, Fischer U, Niittylä T, Sundberg B, Hvidsten TR (2017) AspWood: high-spatial-resolution transcriptome profiles reveal uncharacterized modularity of wood formation in Populus tremula. Plant Cell 29:1585–1604. https://doi.org/10.1105/tpc.17.00153
Tolivia D, Tolivia J (1987) Fasga: a new polychromatic method for simultaneous and differential staining of plant tissues. J Microsc 148:113–117. https://doi.org/10.1111/j.1365-2818.1987.tb02859.x
Van Acker R, Dejardin A, Desmet S, Hoengenaert L, Vanholme R, Morreel K, Laurans F, Kim H, Santoro N, Foster C, Goeminne G, Légée F, Lapierre C, Pilate G, Ralph J, Boerjan W (2017) Different routes for conifer- and sinapaldehyde and higher saccharification upon deficiency in the dehydrogenase CAD1. Plant Physiol 175(3):1018–1039. https://doi.org/10.1104/pp.17.0083
Wang Y, Gril J, Sugiyama J (2009) Variation in xylem formation of Viburnum odoratissimum var. awabuki: growth strain and related anatomical features of branches exhibiting unusual eccentric growth. Tree Physiol 29(5):707–713. https://doi.org/10.1093/treephys/tpp007
Wardrop AB, Dadswell HE (1955) The nature of reaction wood IV. Variations in cell wall organization of tension wood fibres. Aust J Bot 3:177–189
Weng J, Chapple C (2010) The origin and evolution of lignin biosynthesis. New Phytol 187:273–285. https://doi.org/10.1111/j.1469-8137.2010.03327.x
Yokoyama R, Rose JKC, Nishitani K (2004) A surprising diversity and abundance of xyloglucan endotransglucosylase/hydrolases in rice: classification and expression analysis. Plant Physiol 134:1088–1099. https://doi.org/10.1104/pp.103.035261
Yoshida M, Ohta H, Yamamoto H, Okuyama T (2002) Tensile growth stress and lignin distribution in the cell walls of yellow poplar, Liriodendron tulipifera Linn. Trees-Struct Funct 16:457–464. https://doi.org/10.1007/s00468-002-0186-2
Yoshizawa N, Watanabe N, Yokota S, Idei T (1993) Distribution of guaiacyl and syringyl lignins in normal and compression wood of Buxus microphylla var. insularis Nakai. IAWA J 14:139–151. https://doi.org/10.1163/22941932-90001307
Yoshizawa N, Inami A, Miyake S, Ishiguri F, Yokota S (2000) Anatomy and lignin distribution of reaction wood in two Magnolia species. Wood Sci Technol 34:183–196. https://doi.org/10.1007/s002260000046
Acknowledgments
The authors would like to express their gratitude to the National Institute of Research and Agronomy of Orléans (INRA, France) for providing the poplar clone, to S. Lhernould and G. Costa for their scientific and technical advice, to F. Lalloué for access to the cryostat of the “Homéostasie Cellulaire et Pathologies” Laboratory at Limoges University (France), to P. Carles from the Electron Microscopy Department at the University of Limoges (France), and lastly to A. Cantereau for her contribution with the Confocal Microscopy, Image UP, University of Poitiers (France).
Funding
This research was supported by the Limousin Regional Council on behalf of R. Decou and by the PEIRENE EA7500 Laboratory (ex-LCSN - UPRES EA 1069).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human or animal subjects.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Anne-Catherine Schmit
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Decou, R., Labrousse, P., Béré, E. et al. Structural features in tension wood and distribution of wall polymers in the G-layer of in vitro grown poplars. Protoplasma 257, 13–29 (2020). https://doi.org/10.1007/s00709-019-01416-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-019-01416-9