Abstract
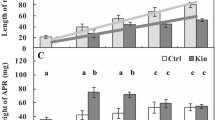
Programmed cell death (PCD) is a crucial process in plant development. In this paper, proteolytically related aspects of kinetin-induced PCD in cortex cells of Vicia faba ssp. minor seedlings were examined using morphological, fluorometric, spectrophotometric, and fluorescence microscopic analyses. Cell viability estimation after 46 μM kinetin treatment of seedling roots showed that the number of dying cortex cells increased with treatment duration, reaching maximum after 72 h. Weight of the apical root segments increased with time and was about 2.5-fold greater after 96 h, while the protein content remained unchanged, compared to the control. The total and cysteine-dependent proteolytic activities fluctuated during 1–96-h treatment, which was not accompanied by the changes in the protein amount, indicating that the absolute protein amounts decreased during kinetin-induced PCD. N-ethylmaleimide (NEM), phenylmethylsulfonyl fluoride (PMSF), and Z-Leu-Leu-Nva-H (MG115), the respective cysteine, serine, and proteasome inhibitors, suppressed kinetin-induced PCD. PMSF significantly decreased serine-dependent proteolytic activities without changing the amount of proteins, unlike NEM and MG115. More pronounced effect of PMSF over NEM indicated that in the root apical segments, the most important proteolytic activity during kinetin-induced PCD was that of serine proteases, while that of cysteine proteases may be important for protein degradation in the last phase of the process. Both NEM and PMSF inhibited apoptotic-like structure formation during kinetin-induced PCD. The level of caspase-3-like activity of β1 proteasome subunit increased after kinetin treatment. Addition of proteasome inhibitor MG-115 reduced the number of dying cells, suggesting that proteasomes might play an important role during kinetin-induced PCD.








Similar content being viewed by others
Abbreviations
- Ctrl:
-
control
- Kin:
-
Kinetin
- MG115:
-
Z-Leu-Leu-Nva-H
- NEM:
-
N-ethylmaleimide
- PCD:
-
Programmed cell death
- PMSF:
-
Phenylmethylsulfonyl fluoride
References
Aleksandrushkina NI, Vanyushin BF (2009) Endonucleases and their involvement in plant apoptosis. Russ J Plant Physiol 56:291–305
Barciszewski J, Massino F, Clark BFC (2007) Kinetin—a multiactive molecule. Inter J Biol Macromol 40:182–192
Beynon R, Bond JS (1999) Proteolytic enzymes, IInd edn. Oxford University press, Oxford
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Byczkowska A, Kunikowska A, Kaźmierczak A (2013) Determination of ACC-induced cell-programmed death in roots of Vicia faba ssp. minor seedlings by acridine orange and ethidium bromide staining. Protoplasma 250:121–128
Cacas JL (2010) Devil inside: does plant programmed cell death involve the endomembrane system? Plant Cell Environ 33:1453–1473
Cai YM, Yu J, Gallois P (2014) Endoplasmic reticulum stress-induced PCD and caspase-like activities involved. Front Plant Sci 5:41–46
Chichkova NV, Shaw J, Galiullina RA, Drury GE, Tuzhikov AI, Kim SH, Kalkum M, Hong TB, Gorshkova EN, Torrance L, Vartapetian AB, Taliansky M (2010) Phytaspase, a relocalisable cell death promoting plant protease with caspase specificity. The EMBO J 29:1149–1161
Coffeen WC, Wolpert TJ (2004) Purification and characterization of serine proteases that exhibit caspase-like activity and are associated with programmed cell death in Avena sativa. Plant Cell 16:857–873
Collazo C, Chacon O, Boras O (2006) Programmed cell death in plants resembles apoptosis of animals. Biotecnol Aplic 23:1–10
Conradt B, Xue D (2005) Programmed cell death. In: Wormbook. The C. elegans Research Community, Wormbook. doi:10.1895/wormbook.1.32.1
Dhyèvre A, Foltête AS, Aran D, Muller S, Cotelle S (2014) Effects of soil pH on the Vicia-micronucleus genotoxicity assay. Mutation Res 774:17–21
Domínguez F, Cejudo FJ (2012) A comparison between nuclear dismantling during plant and animal programmed cell death. Plant Sci 197:114–121
Doniak M, Barciszewska MZ, Kaźmierczak J, Kaźmierczak A (2014) The crucial elements of the ‘last step’ of programmed cell death induced by kinetin in root cortex of V. faba ssp. minor seedlings. Plant Cell Rep 33:2063–2067
Doniak M, Byczkowska A, Kaźmierczak A (2016) Kinetin-induced programmed cell death of cortex cells is mediated by ethylene and calcium ions in roots of Vicia faba ssp. minor. Plant Growth Regul 78:335–343
Doniak M, Kaźmierczak A, Byczkowska A, Glińska S (2017) Reactive oxygen species and sugars may be the messengers in kinetin-induced death of root cortex cells of V. faba ssp. minor seedlings. Biol Plant. doi:10.1007/s10535-016-0654-y
Drew MC, He CJ, Morgan PW (2000) Programmed cell death and aerenchyma formation in roots. Trends Plant Sci 5:123–127
Fernández MB, Daleo GR, Guevara MG (2012) DEVDase activity is induced in potato leaves during Phytophthora infestans infection. Plant Physiol Biochem 61:197–203
Fritz H, Rrautshold I, Werle E (1974) Protease inhibitors. In: Bergmeyer HU (ed) Methods enzymatic analysis. T. 2. Acad. Press, New York
Fukuda H (2016) Signaling, transcriptional regulation, and asynchronous pattern formation governing plant xylem development. Proc Jpn Acad Ser B 92. doi:10.2183/pjab.92.98
Galluzzi L, Bravo-San Pedro JM, Vitale I, Aaronson SA, Abrams JM et al (2015) Essential versus accessory aspects of cell death: recommendations of the NCCD 2015. Cell Death Differ 22:58–73
Galluzzi L, López-Soto A, Kumar S, Kroemer G (2016) Caspases connect cell-death signaling to organismal homeostasis. Immunity 44:221–231
Ge ZQ, Yang S, Cheng JS, Yuan YJ (2005) Signal role for activation of caspase-3-like protease and burst of superoxide anions during Ce4+-induced apoptosis of cultured Taxus cuspidata cells. Biometals 18:221–232
Gunawardena AH (2008) Programmed cell death and tissue remodelling in plants. J Exp Bot 59:445–451
Han JJ, Lin W, Oda Y, Cui KM, Fukuda H, He X (2012) The proteasome is responsible for caspase-3-like activity during xylem development. Plant J 72:129–141
Hatsugai N, Iwasaki S, Tamura K, Kondo M, Fuji K, Ogasawara K, Nishimura M, Hara-Nishimura I (2009) A novel membrane fusion-mediated plant immunity against bacterial pathogens. Genes Dev 23:2496–2506
Iqbal N, Trivellini A, Masood A, Ferrante A, Khan NA (2013) Current understanding on ethylene signaling in plants: the influence of nutrient availability. Plant Physiol Biochem 73:128–138
Kacprzyk J, Daly CT, McCabe PF (2011) The botanical dance of death: programmed cell death in plants. Advances Bot Res 60:169–271
Kunikowska A, Byczkowska A, Kaźmierczak A (2013) Kinetin induces cell death in root cortex cells of Vicia faba ssp. minor seedlings. Protoplasma 250:851–861
Kuranaga E (2012) Beyond apoptosis: caspase regulatory mechanisms and functions in vivo. Genes Cells 17:83–97
Kurepa J, Smalle JA (2008) Structure, function and regulation of plant proteasomes. Biochimie 90:324–335
Moravec R, O’Brien MA, Daily WJ, Scurria MA, Bernad L, Riss TL (2009) Cell-based bioluminescent assays for all three proteasome activities in a homogeneous format. Anal Biochem 387:294–302
Pesquet E, Tuominen H (2011) Ethylene stimulates tracheary element differentiation in Zinnia elegans cell cultures. New Phytol 190:138–149
Poór P, Kovács J, Szopkó D, Tari I (2012) Ethylene signaling in salt stress- and salicylic acid-induced programmed cell death in tomato suspension cells. Protoplasma 250:273–284
Reape TJ, McCabe PF (2008) Apoptotic-like programmed cell death in plants. New Phytol 180:13–26
Reape TJ, McCabe PF (2010) Apoptotic-like regulation of programmed cell death in plants. Apoptosis 15:249–256
Sakamoto W, Takami T (2014) Nucleases in higher plants and their possible involvement in DNA degradation during leaf senescence. J Exp Bot 65:3835–3843
Schaller A, Stintzi A, Graff L (2012) Subtilases—versatile tools for protein turnover, plant development, and interactions with the environment. Physiol Plant 145:52–66
Secades P, Guijarro J (1999) Purification and characterization of an extracellular protease from the fish pathogen Yersinia ruckeri and effect of culture conditions on production. Appl Environ Microbiol 65:3969–3975
Shimizu N (2011) Molecular mechanisms of the origin of micronuclei from extrachromosomal elements. Mutagenesis 26:119–123
Twumasi P, Iakimova ET, Qian T, van Ieperen W, Schel JHN, Emons AMC, van Kooten O, Woltering EJ (2010) Caspase inhibitors affect the kinetics and dimensions of tracheary elements in xylogenic Zinnia (Zinnia elegans) cell cultures. BMC Plant Biol 10:162
Vacca RA, Valenti D, Bobba A, de Pinto MC, Merafina RS, De Garab L, Passarella S, Marra E (2007) Proteasome function is required for activation of programmed cell death in heat shocked tobacco Bright-Yellow 2 cells. FEBS Lett 581:917–922
Van Doorn WG (2005) Plant programmed cell death and the point of no return. Trends Plant Sci 10:478–483
Vartapetian B, Tuzhikov AI, Chichkova NV, Taliansky M, Wolpert TJ (2011) A plant alternative to animal caspases: subtilisin-like proteases. Cell Death Differ 18:1289–1297
Vercammen D, van de Cotte B, De Jaeger G, Eeckhout D, Casteels P, Vandepoele K, Vandenberghe I, Van Beeumen J, Inze D, Van Breusegem F (2004) Type II metacaspases Atmc4 and Atmc9 of Arabidopsis thaliana cleave substrates after arginine and lysine. J Biol Chem 279:45329–45336
Wang H, Li J, Bostock RM, Gilchrista DG (1996) Apoptosis: a functional paradigm for programmed plant cell death induced by a host-selective phytotoxin and invoked during development. Plant Cell 8:375–391
Williams JA, Hou Y, Ni HM, Ding WX (2013) Role of intracellular calcium in proteasome inhibitor-induced endoplasmic reticulum stress, autophagy, and cell death. Pharm Res 30:2279–2289
Ye Y, Li Z, Xing D (2013) Nitric oxide promotes MPK6-mediated caspase-3-like activation in cadmium-induced Arabidopsis thaliana programmed cell death. Plant Cell Environ 36:1–15
Acknowledgments
We thank Ms. M. Fronczak for the help in preparing the manuscript in English. This work was supported by a grant from the University of Łódź, no. 506/1141 and no. B1551000000988.02.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Pavel Dráber
Electronic supplementary material
ESM 1
(RTF 16 kb)
Fig. S1
Simple exemplification of existence signalling, killing and execution phase of Kin-induced of death of cortex cells of the root apical segments of V. faba ssp. minor seedlings in a triangular form. The scheme shows that kinetin induced death signal via cytokinin receptors, then the proteolytic machinery is activated activating the nuclei and cell wall degradation. (GIF 36 kb)
Rights and permissions
About this article
Cite this article
Kaźmierczak, A., Doniak, M. & Kunikowska, A. Proteolytic activities in cortex of apical parts of Vicia faba ssp. minor seedling roots during kinetin-induced programmed cell death. Protoplasma 254, 2273–2285 (2017). https://doi.org/10.1007/s00709-017-1119-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-017-1119-7