Abstract
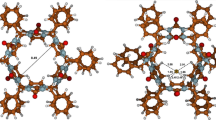
Quantum mechanical density functional theory (DFT) calculations were used to derive the most probable structure of the bambus[6]uril·Cs+I− electroneutral complex species. In this complex, the considered anion I−, included in the macrocyclic cavity, is bound by 12 weak hydrogen bonds between methine hydrogen atoms on the convex face of glycoluril units and the I− anion, whereas the cesium cation Cs+ interacts only with two carbonyl oxygen atoms of the parent macrocyclic receptor. The interaction energy of the resulting complex, involving the Boys–Bernardi counterpoise corrections of the basis set superposition error, was found to be −472.4 kJ/mol.
Graphical abstract




Similar content being viewed by others
References
Lagona J, Mukhopadhyay P, Chakrabarti S, Isaacs L (2005) Angew Chem Int Ed 44:4844
Lee JW, Samal S, Selvapalam N, Kim HJ, Kim K (2003) Acc Chem Res 36:621
Liu S, Ruspic C, Mukhopadhyay P, Chakrabarti S, Zavalij PY, Isaacs L (2005) J Am Chem Soc 127:15959
Freeman WA, Mock WL, Shih NY (1981) J Am Chem Soc 103:7367
Mock WL, Shih NY (1983) J Org Chem 48:3618
Mock WL, Shih NY (1986) J Org Chem 51:4440
Mock WL, Shih NY (1988) J Am Chem Soc 110:4706
Mock WL, Shih NY (1989) J Am Chem Soc 111:2697
Isobe H, Tomita N, Lee JW, Kim HJ, Kim K, Nakamura E (2000) Angew Chem Int Ed 39:4257
Isobe H, Sota S, Lee JW, Kim HJ, Kim K, Nakamura E (2005) Chem Commun 12:1549
Tan Y, Choi S, Lee JW, Ko YH, Kim K (2002) Macromolecules 35:7161
Márquez C, Hudgins RR, Nau WM (2004) J Am Chem Soc 126:5806
Buschmann HJ, Mutihac L, Mutihac RC, Schollmeyer E (2005) Thermochim Acta 430:79
Buschmann HJ, Schollmeyer E, Mutihac L (2003) Thermochim Acta 399:203
Miyahara Y, Goto K, Oka M, Inazu T (2004) Angew Chem Int Ed 43:5019
Li Y, Li L, Zhu Y, Meng X, Wu A (2009) Cryst Growth Des 9:4255
Buschmann HJ, Zielesny A, Schollmeyer E (2006) J Incl Phenom Macrocycl Chem 54:181
Buschmann HJ, Cleve E, Schollmeyer E (2005) Inorg Chem Commun 8:125
Svec J, Necas M, Sindelar V (2010) Angew Chem Int Ed 49:2378
Toman P, Makrlík E, Vaňura P (2011) Monatsh Chem 142:881
Kříž J, Dybal J, Makrlík E (2006) Biopolymers 82:536
Kříž J, Dybal J, Makrlík E, Vaňura P, Lang J (2007) Supramol Chem 19:419
Kříž J, Dybal J, Makrlík E, Vaňura P (2008) Supramol Chem 20:387
Kříž J, Dybal J, Makrlík E, Budka J, Vaňura P (2008) Supramol Chem 20:487
Kříž J, Dybal J, Makrlík E, Budka J (2008) J Phys Chem A 112:10236
Kříž J, Dybal J, Makrlík E, Budka J, Vaňura P (2009) J Phys Chem A 113:5896
Kříž J, Toman P, Makrlík E, Budka J, Shukla R, Rathore R (2010) J Phys Chem A 114:5327
Kříž J, Dybal J, Makrlík E, Vaňura P, Moyer BA (2011) J Phys Chem B 115:7578
Boys SF, Bernardi F (1970) Mol Phys 19:553
van Duijneveldt FB, van Duijneveldt-van de Rijdt JGCM, van Lenthe JH (1994) Chem Rev 94:1873
Hobza P, Šponer J (1999) Chem Rev 99:3247
Barsky D, Colvin ME (2000) J Phys Chem A 104:8570
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Becke AD (1993) J Chem Phys 98:5648
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, Revision C. 02, Gaussian, Wallingford
Acknowledgments
This work was supported by the Grant Agency of Faculty of Environmental Sciences, Czech University of Life Sciences, Prague, project no. 42900/1312/3114 “Environmental Aspects of Sustainable Development of Society”, by the Czech Ministry of Education, Youth, and Sports (project MSM 6046137307), and by the Czech Science Foundation (project P 205/10/2280). The computer time at the MetaCentrum (project LM 2010005), as well as at the Institute of Physics (computer Luna/Apollo), Academy of Sciences of the Czech Republic, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Toman, P., Makrlík, E. & Vaňura, P. Bambus[6]uril as a ditopic ion-pair molecular receptor for Cs+I− . Monatsh Chem 143, 1365–1368 (2012). https://doi.org/10.1007/s00706-012-0806-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-012-0806-5