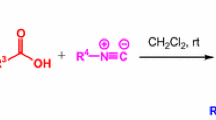
Abstract
Novel α-acyloxycarboxamides were synthesized and characterized by the Passerini three-component reaction between pyridine carbaldehydes, cyclohexyl isocyanide, and benzoic acid derivatives in water. The reactions were carried out in one pot at room temperature with a quantitative yield. The products were obtained without any need for purification.
Graphical abstract



Similar content being viewed by others
References
Sapi J, Laronze J-Y (2004) Arkivoc 7:208
Zhu J, Bienayme H (2005) Multicomponent reactions. Wiley-VCH, Weinheim
Ugi I (1962) Angew Chem Int Ed 1:8
Hazeri N, Maghsoodlou MT, Habibi-Khorassani SM, Ziyaadini M, Marandi G, Khandan-Barani K, Bijanzadeh HR (2007) Arkivoc 8:34
Dömling A, Beck B, Herdtweck E, Antuch W, Oefner C, Yehia N, Gracia-Marques A (2007) Arkivoc 17:99
Dömling A, Ugi I (2000) Angew Chem Int Ed 39:3168
Ugi I, Werner B, Dömling A (2003) Molecules 8:53
Ugi I (2001) Pure Appl Chem 73:187
Dömling A (2006) Chem Rev 106:17
Passerini M, Simone L (1921) Gazz Chim Ital 51:126
Passerini M (1921) Gazz Chim Ital 51:181
Passerini M, Ragni G (1931) Gazz Chim Ital 61:964
Hulme C, Gore V (2003) Curr Med Chem 10:51
Kazemizadeh AR, Ramazani A (2008) Arkivoc 15:159
Souldozi A, Ślepokura K, Lis T, Ramazani A (2007) Z Naturforsch 62b:835
Souldozi A, Ramazani A (2007) Tetrahedron Lett 48:1549
Souldozi A, Ramazani A, Bouslimani N, Welter R (2007) Tetrahedron Lett 48:2617
Ahmadi E, Ramazani A, Nekomanesh Haghighi M (2007) Tetrahedron Lett 48:6954
Baker RH, Stanonis D (1951) J Am Chem Soc 73:699
Acknowledgments
The authors are thankful to the Payame Noor University for financial support of this research work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Vessally, E., Ramazani, A. & Yaaghubi, E. Green synthesis and characterization of novel α-acyloxycarboxamides through three-component reaction between pyridine carbaldehydes, cyclohexyl isocyanide, and benzoic acid derivatives. Monatsh Chem 142, 1143–1147 (2011). https://doi.org/10.1007/s00706-011-0536-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0536-0