Summary.
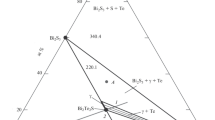
The partial and integral enthalpies of mixing of liquid Bi–Sn–Zn alloys were determined at 500°C by a drop calorimetric technique using a Calvet-type microcalorimeter. The ternary interaction parameters in the Bi–Sn–Zn system were fitted using the Redlich-Kister-Muggianu model for substitutional solutions, and isoenthalpy curves of the integral molar enthalpy of mixing at 500°C were constructed. Furthermore, a DSC technique was used to determine the liquidus temperatures in three sections (3, 5, and 7 at.% Zn) as well as the invariant reaction temperature of the ternary eutectic L ⇄ (Bi) + (Sn) + (Zn). The ternary eutectic reaction was found at 135°C.
Similar content being viewed by others
References
EP Wood KL Nimmo (1994) J Electron Mater 23 709 Occurrence Handle1:CAS:528:DyaK2cXls1Wqt7g%3D
MJ Realff M Raymond J Ammons (2004) Mater Today 7 40 Occurrence Handle1:CAS:528:DC%2BD2cXpsVajtLY%3D
K Nimmo (2003) Second European Lead-free Soldering Technology Roadmap. SOLDERTEC at Tin Technology Ltd Uxbridge, UK 12
I Shohji C Gagg WJ Plumbridge (2004) J Electron Mater 33 923 Occurrence Handle1:CAS:528:DC%2BD2cXmtlylsrs%3D
Flandorfer H, Sabbar A, Luef C, Ipser H (2005) Publication in preparation
N Asryan A Mikula (2004) Z Metallkd 95 132 Occurrence Handle1:CAS:528:DC%2BD2cXjvFyktL4%3D
FE Wittig F Huber (1958) Z Phys Chem (Frankfurt) 18 330 Occurrence Handle1:CAS:528:DyaF3MXis1Wjtw%3D%3D
A Yazawa T Kawashima K Itagaki (1968) J Jpn Inst Met 32 1281 Occurrence Handle1:CAS:528:DyaF1MXotFKmtA%3D%3D
RL Sharkey MJ Pool (1972) Met Trans 3 1773 Occurrence Handle1:CAS:528:DyaE38Xks1Kqsbo%3D
H Seltz FJ Dunkerley (1942) J Am Chem Soc 64 1392 Occurrence Handle10.1021/ja01258a044 Occurrence Handle1:CAS:528:DyaH38XivVKrsg%3D%3D
M Kawakami (1930) Sci Repts Tohoku Imp Univ 19 521 Occurrence Handle1:CAS:528:DyaA3MXhsFOksw%3D%3D
FE Wittig E Müller W Schilling (1958) Z Elektrochem 62 529 Occurrence Handle1:CAS:528:DyaG1cXhtVais7o%3D
DV Malakhov (2000) Calphad 24 1 Occurrence Handle10.1016/S0364-5916(00)00011-0 Occurrence Handle1:CAS:528:DC%2BD3cXivFSgsrw%3D
OJ Kleppa (1955) J Phys Chem 59 354 Occurrence Handle1:CAS:528:DyaG2MXlvFWmtg%3D%3D
W Oelson (1957) Z Metallkd 48 1
B Dobovisek B Straus (1960) Rudarsko Met Zbornik 3 273
M Genot R Hagege (1960) Compt Rend, Acad Sci Fr 25 2901
E Schürmann H Träger (1961) Arch Eisenhüttenwes 32 397
K Itagaki A Yazawa (1975) J Jpn Inst Met 39 880 Occurrence Handle1:CAS:528:DyaE28XnvVSrsQ%3D%3D
Z Moser K Rzyman S Randzio (1987) Bull Pol Acad Sci, Tech Sci 35 461 Occurrence Handle1:CAS:528:DyaL1cXhtlyrsb4%3D
W Ptak (1960) Arch Hutnictwa 5 169
K Sano K Okajima S Tatsuo (1953) Mem Fac Eng, Nagoya Univ 5 299 Occurrence Handle1:CAS:528:DyaG28XjtVyktA%3D%3D
M Fiorani V Valenti (1955) Gazz Chim Ital 85 607 Occurrence Handle1:CAS:528:DyaG2MXns1ektQ%3D%3D
E Scheil ED Müller (1962) Z Metallkd 53 389 Occurrence Handle1:CAS:528:DyaF38XksVWrurs%3D
B-J Lee (1996) Calphad 20 471 Occurrence Handle10.1016/S0364-5916(97)00009-6 Occurrence Handle1:CAS:528:DyaK2sXhsVWltLg%3D
DV Malakhov XJ Liu I Ohnuma K Ishida (2000) J Phase Equil 21 514 Occurrence Handle1:CAS:528:DC%2BD3MXhtFWmsg%3D%3D
N Moelans KC Hari Kumar P Wollants (2003) J Alloys Comp 360 98 Occurrence Handle10.1016/S0925-8388(03)00325-6 Occurrence Handle1:CAS:528:DC%2BD3sXnt1CksLg%3D
SD Muzaffar (1923) J Chem Soc 123 2341 Occurrence Handle1:CAS:528:DyaB2cXhsFKl
E Jänecke (1937) Z Metallkd 29 367
AD Pelton CW Bale M Rigaud (1977) Z Metallkd 68 135 Occurrence Handle1:CAS:528:DyaE2sXht1yhs7k%3D
JV Gluck RD Pehlke (1967) Trans AIME 239 36 Occurrence Handle1:CAS:528:DyaF2sXlvFGhtg%3D%3D
L Oleari M Fiorani (1959) Ric Sci 29 2219 Occurrence Handle1:CAS:528:DyaF3MXlslY%3D
W Ptak Z Moser (1966) Arch Hutnictwa 11 289 Occurrence Handle1:CAS:528:DyaF2sXkvFCmsb8%3D
CW Bale AD Pelton M Rigaud (1977) Z Metallkd 68 69 Occurrence Handle1:CAS:528:DyaE2sXps12qsg%3D%3D
RL Louvet JV Gluck RD Pehlke (1968) Trans AIME 242 2369 Occurrence Handle1:CAS:528:DyaF1MXltFaqsQ%3D%3D
VNS Mathur ML Kapoor (1985) Z Metallkd 76 16 Occurrence Handle1:CAS:528:DyaL2MXhtVWnsLw%3D
Massalski TB, Murray JL, Bennett LH, Baker H (eds) (1990) Binary Alloy Phase Diagrams, ASM, Materials Park, OH
Ansara I, Dupin N (1998) In: COST 507 Thermochemical database for light metal alloys, vol 2. European Commission DG XII, Luxembourg, p 1
Saunders N, Miodownik AP (1998) In: CALPHAD (Calculation of Phase Diagrams): A Comprehensive Guide, Pergamon Press, Oxford, UK
H Flandorfer F Gehringer E Hayer (2002) Thermochim Acta 382 77 Occurrence Handle10.1016/S0040-6031(01)00739-0 Occurrence Handle1:CAS:528:DC%2BD38XmsV2rsQ%3D%3D
AT Dinsdale (1991) CALPHAD 15 317 Occurrence Handle10.1016/0364-5916(91)90030-N Occurrence Handle1:CAS:528:DyaK38XmvVOgtQ%3D%3D
C Luef H Flandorfer H Ipser (2004) Z Metallkd 95 151 Occurrence Handle1:CAS:528:DC%2BD2cXjvFyktLo%3D
C Luef H Flandorfer H Ipser (2004) Thermochim Acta 417 47 Occurrence Handle10.1016/j.tca.2004.01.019 Occurrence Handle1:CAS:528:DC%2BD2cXksFynt78%3D
C Luef A Paul H Flandorfer A Kodentsov H Ipser (2005) J Alloys Comp 391 67 Occurrence Handle10.1016/j.jallcom.2004.08.056 Occurrence Handle1:CAS:528:DC%2BD2MXitVajsLY%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luef, C., Paul, A., Vizdal, J. et al. Thermodynamic Properties and Melting Behavior of Bi–Sn–Zn Alloys. Monatsh. Chem. 137, 381–395 (2006). https://doi.org/10.1007/s00706-005-0457-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-005-0457-x