Summary.
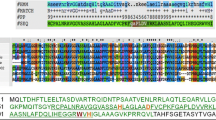
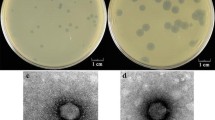
The genes encoding the host cell wall-lytic proteins were searched in the genome DNA of phage PL-1 active against Lactobacillus casei ATCC 27092 by comparing the amino acid sequences with those of others using a computer software of the DDBJ data base. The gene regions found were cloned into E. coli by inserting PCR-amplified DNA fragments into the EcoRI site of pUC19, and the nucleotide sequences were determined. One of the ORFs (hol) consisted of 270 bp encoding 90 amino acids. The hol product (holin) possessed a putative secretion signal, two putative transmembrane helices, and a highly charged C-terminus. Another ORF (lys) consisted of 1050 bp encoding an N-acetylmuramoyl-L-alanine amidase of 350 amino acids. The gene lys was expressed in E. coli using pCALn expression vector, and the purified gene product hydrolysed the amide linkage in the peptidoglycans of L. casei. The amino acid sequence of PL-1 amidase showed a high homology to those of Lactococcus lactis phage rlt and Listeria monocytogenes phage A511. It was suggested that the N-terminal region was involved in enzyme activity and the C-terminal region in binding the enzyme to the cell wall substrate, respectively.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received January 10, 2000 Accepted April 13, 2000
Rights and permissions
About this article
Cite this article
Kashige, N., Nakashima, Y., Miake, F. et al. Cloning, sequence analysis, and expression of Lactobacillus casei phage PL-1 lysis genes. Arch. Virol. 145, 1521–1534 (2000). https://doi.org/10.1007/s007050070073
Issue Date:
DOI: https://doi.org/10.1007/s007050070073