Abstract
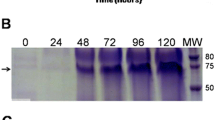
Viral protein 2 (VP2) of porcine parvovirus (PPV) is the major viral structural protein and is responsible for eliciting neutralizing antibodies in immunized animals. In this study, we constructed and characterized a recombinant yeast vector encoding the VP2 protein, designated as pGAPZαA-VP2. The construct was confirmed by restriction enzyme digestion, PCR, and sequencing and then introduced into P. pastoris strain SMD1168 by electroporation. The expressed VP2 protein was analyzed by SDS-PAGE and western blot. Immunization of mice with the VP2 protein elicited a PPV-specific humoral immune response. Notably, a preparation of VP2 protein containing adjuvant induced a much better antibody response than VP2 alone. Clearly, the adjuvant strongly enhanced the immunogenicity of VP2. This study provides a foundation for the application of the VP2 protein in the clinical diagnosis of PPV and in vaccination against PPV in the future.





Similar content being viewed by others
References
Allan GM, Kennedy S, McNeilly F, Foster JC, Ellis JA, Krakowka SJ, Meehan BM, Adair BM (1999) Experimental reproduction of severe wasting disease by co-infection of pigs with porcine circovirus and porcine parvovirus. J Comp Pathol 121:1–11
Cadar D, Dan A, Tombacz K, Lorincz M, Kiss T, Becskei Z, Spinu M, Tuboly T, Csagola A (2012) Phylogeny and evolutionary genetics of porcine parvovirus in wild boars. Infect Genet Evol: J Mol Epidemiol Evol Genet Infect Dis 12:1163–1171
Casal JI (1996) Parvovirus diagnostics and vaccine production in insect cells. Cytotechnology 20:261–270
Chen H-Y, Zhao L, Wei Z-Y, Cui B-A, Wang Z-Y, Li X-S, Xia P-A, Liu J-P (2010) Enhancement of the immunogenicity of an infectious laryngotracheitis virus DNA vaccine by a bicistronic plasmid encoding glycoprotein B and interleukin-18. Antivir Res 87:235–241
Corbett EM, Grooms DL, Bolin SR (2012) Evaluation of skin samples for bovine viral diarrhea virus by use of reverse transcriptase polymerase chain reaction assay after vaccination of cattle with a modified-live bovine viral diarrhea virus vaccine. Am J Vet Res 73:319–324
Fisher S, Genbacev O, Maidji E, Pereira L (2000) Human cytomegalovirus infection of placental cytotrophoblasts in vitro and in utero: Implications for transmission and pathogenesis. J Virol 74:6808–6820
Gardner I, Carpenter T, Leontides L, Parsons T (1996) Financial evaluation of vaccination and testing alternatives for control of parvovirus-induced reproductive failure in swine. J Am Vet Med Assoc 208:863–869
Ha Y, Shin JH, Chae C (2010) Colostral transmission of porcine circovirus 2 (PCV-2): reproduction of post-weaning multisystemic wasting syndrome in pigs fed milk from PCV-2-infected sows with post-natal porcine parvovirus infection or immunostimulation. J Gen Virol 91:1601–1608
Hao X, Lu Z, Sun P, Fu Y, Cao Y, Li P, Bai X, Bao H, Xie B, Chen Y, Li D, Liu Z (2011) Phylogenetic analysis of porcine parvoviruses from swine samples in China. Virol J 8:320
Hu H, Huang X, Tao L, Huang Y, Cui B-A, Wang H (2009) Comparative analysis of the immunogenicity of SARS-CoV nucleocapsid DNA vaccine administrated with different routes in mouse model. Vaccine 27:1758–1763
Jóźwik A, Manteufel J, Selbitz H-J, Truyen U (2009) Vaccination against porcine parvovirus protects against disease, but does not prevent infection and virus shedding after challenge infection with a heterologous virus strain. J Gen Virol 90:2437–2441
Johnson SC, Yang MM, Murthy PPN (2010) Heterologous expression and functional characterization of a plant alkaline phytase in Pichia pastoris. Protein Expr Purif 74:196–203
Kamstrup S, Langeveld J, Bøtner A, Nielsen J, Schaaper WM, Boshuizen RS, Casal JI, Højrup P, Vela C, Meloen R (1998) Mapping the antigenic structure of porcine parvovirus at the level of peptides. Virus Res 53:163–173
Kim J, Chae C (2004) A comparison of virus isolation, polymerase chain reaction, immunohistochemistry, and in situ hybridization for the detection of porcine circovirus 2 and porcine parvovirus in experimentally and naturally coinfected pigs. J Vet Diagn Invest 16:45–50
Krakowka S, Ellis JA, Meehan B, Kennedy S, McNeilly F, Allan G (2000) Viral wasting syndrome of swine: experimental reproduction of postweaning multisystemic wasting syndrome in gnotobiotic swine by coinfection with porcine circovirus 2 and porcine parvovirus. Vet Pathol 37:254–263
López DTJ, Cortés E, Ranz A, García J, Sanz A, Vela C, Casal JI (1991) Fine mapping of canine parvovirus B cell epitopes. J Gen Virol 72:2445–2456
Lima KM, dos Santos SA, Rodrigues JM, Silva CL (2004) Vaccine adjuvant: it makes the difference. Vaccine 22:2374–2379
Lo-Man R, Rueda P, Sedlik C, Deriaud E, Casal I, Leclerc C (1998) A recombinant virus-like particle system derived from parvovirus as an efficient antigen carrier to elicit a polarized Th1 immune response without adjuvant. Eur J Immunol 28:1401–1407
Luckow VA, Lee S, Barry G, Olins P (1993) Efficient generation of infectious recombinant baculoviruses by site-specific transposon-mediated insertion of foreign genes into a baculovirus genome propagated in Escherichia coli. J Virol 67:4566–4579
Mølhøj M, Ulvskov P, Dal Degan F (2001) Characterization of a functional soluble form of a Brassica napus membrane-anchored endo-1, 4-β-glucanase heterologously expressed in Pichia pastoris. Plant Physiol 127:674–684
Martínez C, Dalsgaard K, López de Turiso J, Cortés E, Vela C, Casal J (1992) Production of porcine parvovirus empty capsids with high immunogenic activity. Vaccine 10:684–690
Meyer D, Aebischer A, Muller M, Grummer B, Greiser-Wilke I, Moennig V, Hofmann MA (2012) New insights into the antigenic structure of the glycoprotein E-rns of classical swine fever virus by epitope mapping. Virology 433:45–54
Niewold TA, Kerstens HHD, van der Meulen J, Smits MA, Hulst MM (2005) Development of a porcine small intestinal cDNA micro-array: characterization and functional analysis of the response to enterotoxigenic E-coli. Vet Immunol Immunop 105:317–329
O’dea MA, Kabay MJ, Carr J, Wilcox GE, Richards RB (2012) Porcine circovirus diseases: a review of PMWS (vol 59, p. 60, 2012). Transbound Emerg Dis 59:376
Opriessnig T, Shen HG, Pal N, Ramamoorthy S, Huang YW, Lager KM, Beach NM, Halbur PG, Meng XJ (2011) A live-attenuated chimeric porcine circovirus type 2 (PCV2) vaccine is transmitted to contact pigs but is not upregulated by concurrent infection with porcine parvovirus (PPV) and porcine reproductive and respiratory syndrome virus (PRRSV) and Is efficacious in a PCV2b-PRRSV-PPV challenge model. Clin Vaccine Immunol 18:1261–1268
Parke C, Burgess G (1993) An economic assessment of porcine parvovirus vaccination. Aust Vet J 70:177–180
Pena L, Yanez R, Revilla Y, Vinuela E, Salas M (1993) African swine fever virus guanylyltransferase. Virology 193:319–328
Peres MDS, Silva VC, Valentini SR, Gattas EAD (2010) Recombinant expression of glycerol-3-phosphate dehydrogenase using the Pichia pastoris system. J Mol Catal B Enzym 65:128–132
Qi T, Cui S (2009) Expression of porcine parvovirus VP2 gene requires codon optimized E. coli cells. Virus Genes 39:217–222
Qing L, Lv J, Li H, Tan Y, Hao H, Chen Z, Zhao J, Chen H (2006) The recombinant nonstructural polyprotein NS1 of porcine parvovirus (PPV) as diagnostic antigen in ELISA to differentiate infected from vaccinated pigs. Vet Res Commun 30:175–190
Rueda P, Fominaya J, Langeveld JP, Bruschke C, Vela C, Casal JI (2000) Effect of different baculovirus inactivation procedures on the integrity and immunogenicity of porcine parvovirus-like particles. Vaccine 19:726–734
Sahmel J, Devlin K, Paustenbach D, Hollins D, Gaffney S (2010) The role of exposure reconstruction in occupational human health risk assessment: current methods and a recommended framework. Crit Rev Toxicol 40:799–843
Shangjin C, Cortey M, Segalés J (2009) Phylogeny and evolution of the NS1 and VP1/VP2 gene sequences from porcine parvovirus. Virus Res 140:209–215
Si Y-H, Fang M-G, Wang H-Z (2006) Expression of porcine parvovirus vp2 gene and construction of virus like particles. Virologica Sinica 21:148
Simpson AA, Hébert Bt, Sullivan GM, Parrish CR, Zádori Z, Tijssen P, Rossmann MG (2002) The structure of porcine parvovirus: comparison with related viruses. J Mol Biol 315:1189–1198
Tijssen P, Bergeron J, Dubuc R, Hébert BT (1995) Minor genetic changes among porcine parvovirus groups are responsible for major distinguishing biological properties. In: Seminars in virology. Elsevier, pp 319–328
Valore EV, Ganz T (1997) Laboratory production of antimicrobial peptides in native conformation. Methods Mol Biol: Clifton Totowa 78:115–132
Wang JL, Liu MQ, Han J, Chen WZ, Cong W, Cheng G, Gao YH, Lu YG, Chen JL, Zuo XP, Yan WY, Zheng ZX (2007) A peptide of foot-and-mouth disease virus serotype Asia1 generating a neutralizing antibody response, and an immunostimulatory peptide. Vet Microbiol 125:224–231
Xu G, Xu X, Li Z, He Q, Wu B, Sun S, Chen H (2004) Construction of recombinant pseudorabies virus expressing NS1 protein of Japanese encephalitis (SA14-14-2) virus and its safety and immunogenicity. Vaccine 22:1846–1853
Zeeuw E, Leinecker N, Herwig V, Selbitz H-J, Truyen U (2007) Study of the virulence and cross-neutralization capability of recent porcine parvovirus field isolates and vaccine viruses in experimentally infected pregnant gilts. J Gen Virol 88:420–427
Zhou HC, Yao GZ, Cui SJ (2010) Production and purification of VP2 protein of porcine parvovirus expressed in an insect-baculovirus cell system. Virol J 7:366
Acknowledgment
This work was supported by Guangdong Province Live Pig Industry Technology System of Modern Agriculture (F10021).
Conflict of interest
The authors declare that they have no competing interests. None of the authors has any financial or personal relationships that could inappropriately influence or bias the content of the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, C., Zhong, Z. & Huang, Y. Production and immunogenicity of VP2 protein of porcine parvovirus expressed in Pichia pastoris . Arch Virol 159, 963–970 (2014). https://doi.org/10.1007/s00705-013-1907-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-013-1907-0