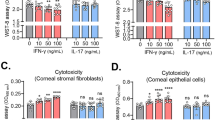
Abstract
Angiogenesis and inflammatory mediators are critical pathogenic factors in herpetic stromal keratitis (HSK). Since disease progresses without infectious virus, HSV-DNA and HSV-IgG complexes (HSV-IC) may contribute to HSK by triggering these factors. Production of VEGF and MMP-9 was studied in vitro using corneal epithelial cells (HCE), fibroblasts (HCRF) and macrophages (THP-1). VEGF was elevated in HCRF and THP-1 following treatment with HSV-DNA and HSV-IC. MMP-9 was elevated in THP-1 but not in corneal cells. When anti-HSV-IgG(Fab′)2 complexes stimulated THP-1, MMP-9 was reduced to control levels. Pretreatment of THP-1 with anti-TLR-2 and -3 inhibited MMP-9 production. Thus, HSV-IC may stimulate THP-1 through the Fc receptor and TLRs. Proinflammatory cytokines (IL-1b, IL-6, and TNF-α) increased VEGF and MMP-9 in corneal cells and macrophages. These studies indicate that the continued presence of HSV-DNA and HSV-IC contribute to angiogenesis and inflammation in HSK. Thus, cytokines and TLRs may be potential targets for intervention.






Similar content being viewed by others
References
Akira S, Takeda K (2004) Toll like receptor signaling. Nat Rev Immunol 4:499–511
Akira S, Uematsu S et al (2006) Pathogen recognition and innate immunity. Cell 124:783–880
Banerjee K, Biswas PS et al (2005) Elucidating the protective and pathologic T cell species in the virus-induced corneal immunoinflammatory condition herpetic stromal keratitis. J Leukocyte Biol 77:24–32
Biswas PS, Banerjee K et al (2006) Involvement of IL-6 in the paracrine production of VEGF in ocular HSV-1 infection. Exp Eye Res 82:46–54
Biswas PS, Rouse BT (2005) Early events in HSV keratitis-setting the stage for a blinding disease. Microbes Infect 7:799–810
Blander JM, Medzhitov R (2004) Regulation of phagosome maturation by signals from toll-like receptors. Science 304:1014–1018
Blander JM (2007) Coupling Toll-like receptor signaling with phagocytosis: potentiation of antigen presentation. Trends Immunol 28:19–25
Cox D, Tseng CC et al (1997) A requirement for phosphoinositol 3-kinase in pseodopod extension. J Biol Chem 274:1240–1247
Hayashi K, Hooper LC et al (2006) Herpes simplex virus 1(HSV-1) DNA and immune complex (HSV-1-human IgG) elicit vigorous interleukin 6 release from infected corneal cells via Toll-like receptors. J Gen Virol 87:2161–2169
Kwong AD, Kruper JA et al (1988) Herpes simplex virus virion host shut off function. J Virol 62:912–921
Lee S, Zheng M et al (2002) Role of matrix metalloproteinase-9 in angiogenesis caused by ocular infection with herpes simplex virus. J Clin Invest 110:1105–1111
Li X, Tupper JC et al (2003) Phosphoinositide-3 kinase mediate Toll-like receptor 4-induced activation of NF-kappa B in endothelial cells. Infect Immun 71:4414–4420
Liesengang TS (2001) Herpes simplex virus epidemiology and ocular importance. Cornea 20:1–13
Matsumoto M, Kikkawa S et al (2002) Establishment of a monoclonal antibody against human Toll-like receptor 3 that blocks double-stranded RNA-mediated signaling. Biochem Biophys Res Commun 293:1364–1369
Meyers-Elliott RH, Pettit TH et al (1980) Viral antigens in the immune ring of herpes simplex stromal keratits. Arch Ophthalmol 98:897–904
Opremcak EM, Rice BA et al (1990) Histology and immunohistology of Ig-h-1-restricted herpes simplex keratitisin Balb/c congenic mice. Invest Ophthalmol Vis Sci 31:305–312
Sciortino M-T, Suzuki M et al (2001) RNAs extracted from herpes simplex virus 1 virions: apparent selectivity of viral but not cellular RNAs packaged in virions. J Virol 75:8105–8116
Sarkar SN, Peters KL et al (2004) Novel roles of TLR3 tyrosine phosphorylation and PI Kinase in double strand RNA signaling. Nat Struct Mol Biol 11:1060–1067
Shimomura Y, Deai T et al (2007) Corneal buttons obtained from HSK patients harbor high copy number of the HSV genome. Cornea 26:190–193
Staats HF, Lausch RN (1993) Cytokine expression in vivo during murine herpetic stromal keratits. Effect of protective antibody therapy. J Immunol 151:277–283
Toker A, Cantley LC (1997) Signaling through the lipid products of phosphoinositide-3-OH-kinase. Nature 387:673–676
Tsuchiya S, Kobayashi Y et al (1982) Induction of maturation in cultured human monocytic leukemia cells by a phorbol diester. Cancer Res 42:1530–1536
Ueta M, Hamuro J et al (2005) Triggering of TLR3 by poly I:C in human corneal epithelial cells to induce inflammatory cytopkines. Biochem Biophys Res Commun 331:285–294
Underhill DM, Gantner B (2004) Integration of Toll-like receptor and phagocytic signaling for tailored immunity. Microbes Infect 6:1368–1373
Underhill DM, Ozinsky A et al (1999) The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401:811–815
Viahos CJ, Matter WF et al (1994) A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)-8 phenyl-4H-I-benzopyran-4-one (LY294002). J Boiol Chem 269:5241–5248
Yang Y-N, Bauer D et al (2003) Matrix metalloproteases (MMP-2 and 9) and tissue inhibitors of matrix metalloproteinases (TIMP-1 and 2) during the course of experimental necrotizing herpetic keratitis. Exp Eye Res 77:227–237
Zarember KA, Godowski PJ (2002) Tissue expression of human Toll-like receptors and differential regulation of Toll-like receptor mRNAs in leukocutes in response to microbes, their products, and cytokines. J Immunol 168:554–561
Zheng M, Deshpande S et al (2001) Contribution of vascular endothelial growth factor in the neovacularization process during the pathogenesisof herpetic stromal keratitis. J Virol 75:9828–9835
Zheng M, Schwarz MA et al (2001) Control of stromal keratits by inhibition of neovascularization. Am J Pathol 159:1021–1029
Acknowledgments
We thank Dr. Chandraskharam Naginini for providing us HCRFs. This research was supported (in part) by the Intramural Research program of NIH National Eye Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hayashi, K., Hooper, L.C., Detrick, B. et al. HSV immune complex (HSV-IgG: IC) and HSV-DNA elicit the production of angiogenic factor VEGF and MMP-9. Arch Virol 154, 219–226 (2009). https://doi.org/10.1007/s00705-008-0303-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-008-0303-7