Abstract
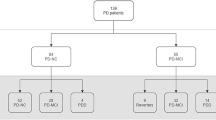
To determine whether systemic medical factors, such as vascular risk factors, metabolic and inflammatory markers contribute to cognitive decline in Parkinson’s disease (PD); if confirmed to determine whether a clinically applicable risk factor model can predict the conversion from normal cognition (NC) to mild cognitive impairment (MCI). 58 patients who met the UK Brain Bank Criteria for PD underwent clinical and laboratory assessment at study entry; 47 patients were re-assessed after 2 years. Medical history, vascular risk (QRISK2), blood metabolic and inflammatory factors, brain vessel examinations, activity of daily living, and neuropsychological testing were performed. Forty patients had NC and 18 patients had MCI at baseline. Patients with MCI had higher level of interleukin 6, folic acid below normal range and higher l-dopa equivalent dose compared to cognitive normal patients at baseline. Patients with NC at baseline were classified into two groups: patients who remained cognitively normal (non-converters, n = 23) and patients who progressed to MCI (converters, n = 11). MCI converters were older at baseline and had higher QRISK2 than the non-converters. Patients with higher QRISK2, lower uric acid level and lower activity of daily living scale at baseline had a higher risk of converting from NC to MCI with a sensitivity of 72.2%, a specificity of 87%, and an overall accuracy of 82.4%. Systemic medical factors are associated with cognitive impairment in PD both cross-sectionally and longitudinally. A risk factor model predicting the decline from NC to MCI could be constructed.

Similar content being viewed by others
Abbreviations
- ACE:
-
Addenbrooke’s cognitive examination
- ADL:
-
Activity of daily living
- AUC:
-
Area under the curve
- BI:
-
Barthel index
- BVRT:
-
Benton visual retention test
- CCA:
-
Common carotid artery
- CDT:
-
The clock drawing test
- CI:
-
Confidence intervals
- IL-6:
-
Interleukin 6
- IMT:
-
Intimal-medial thickness
- MCA:
-
Medial cerebral artery
- MCI:
-
Mild cognitive impairment
- MMSE:
-
Mini-mental state examination
- NC:
-
Normal cognition
- OR:
-
Odds ratio
- PD:
-
Parkinson’s disease
- PDD:
-
Parkinson’s disease dementia
- PD-MCI:
-
Parkinson’s disease mild cognitive impairment
- PI:
-
Pulsatility index
- RI:
-
Resistance index
- ROC:
-
Receiver operating characteristic
- UPDRS:
-
Unified Parkinson’s disease rating scale
- USG:
-
Ultrasonographic
- VFT:
-
The verbal fluency test (VFT),
- WMS:
-
Wechsler Memory Scale III
References
Annanmaki T, Pessala-Driver A, Hokkanen L, Murros K (2008) Uric acid associates with cognition in Parkinson’s disease. Parkinsonism Relat Disord 14(7):576–578
Annanmaki T, Pohja M, Parviainen T, Hakkinen P, Murros K (2011) Uric acid and cognition in Parkinson’s disease: a follow-up study. Parkinsonism Relat Disord 17(5):333–337
Araújo JR, Martel F, Borges N, Araújo JM, Keating E (2015) Folates and aging: role in mild cognitive impairment, dementia and depression. Ageing Res Rev 22:9–19
Auriacombe S, Grossman M, Carvell S, Gollomp S, Stern MB, Hurtig HI (1993) Verbal fluency deficits in Parkinson’s disease. Neuropsychology 7(2):182–192
Barrash J, Stillman A, Anderson SW, Uc EY, Dawson JD, Rizzo M (2010) Prediction of driving ability with neuropsychological tests: demographic adjustments diminish accuracy. J Int Neuropsychol Soc 16(4):679–686
Bonifati DM, Kishore U (2007) Role of complement in neurodegeneration and neuroinflammation. Mol Immunol 44(5):999–1010
Cao B, Wei QQ, Ou R, Yang J, Shang HF (2015) Association of serum uric acid level with cognitive function among patients with multiple system atrophy. J Neurol Sci 359(1–2):363–366
De Pandis MF, Galli M, Vimercati S, Cimolin V, De Angelis MV, Albertini G (2010) A New approach for the quantitative evaluation of the clock drawing test: preliminary results on subjects with Parkinson’s disease. Neurol Res Int. 2010:283890
Dimopoulos N, Piperi C, Salonicioti A et al (2006) Association of cognitive impairment with plasma levels of folate, vitamin B12 and homocysteine in the elderly. Vivo 20(6B):895–899
Dufek M, Hamanová M, Lokaj J et al (2009) Serum inflammatory biomarkers in Parkinson’s disease. Parkinsonism Relat Disord 15(4):318–320
Folstein MF, Folstein SE, McHugh PR (1975) Mini-mental state. A practical method forgrading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198
Grey MT, Veselý B, Gajdoš M, Klobušiaková P, Rektor I (2018) Contribution of white matter lesions on cognitive decline in Parkinson’s disease. Parkinsonism Relat Disord 61:248–249
Harrison MB, Wylie SA, Frysinger RC, Patrie JT, Huss DS, Currie LJ, Wooten GF (2009) UPDRS activity of daily living score as a marker of Parkinson’s disease progression. Mov Disord 24(2):224–230
Hsieh S, McGrory S, Leslie F et al (2015) The Mini-Addenbrooke’s Cognitive Examination: a new assessment tool for dementia. Dement Geriatr Cogn Disord 39(1–2):1–11
Kandiah N, Mak E, Ng A, Huang S, Au WL, Sitoh YY, Tan LC (2013) Cerebral white matter hyperintensity in Parkinson’s disease: a major risk factor for mild cognitive impairment. Parkinsonism Relat Disord 19(7):680–683
Lindqvist D, Hall S, Surova Y, Nielsen HM, Janelidze S, Brundin L, Hansson O (2013) Cerebrospinal fluid inflammatory markers in Parkinson’s disease—associations with depression, fatigue, and cognitive impairment. Brain Behav Immun 33:183–189
Litvan I, Goldman JG, Tröster AI et al (2012) Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: movement Disorder Society Task Force guidelines. Mov Disord 27(3):349–356
Litvan I, Kieburtz K, Tröster AI, Aarsland D (2018) Strengths and challenges in conducting clinical trials in Parkinson’s disease mild cognitive impairment: PD-MCI Clinical Trials. Mov Disord 33(4):520–527
Mahoney FI, Barthel DW (1965) Functional evaluation: the Barthel Index. Md State Med J 14:61–65
Malek N, Lawton MA, Swallow DM et al (2016) Vascular disease and vascular risk factors in relation to motor features and cognition in early Parkinson’s disease: vascular disease and vascular risk factors in PD. Mov Disord 31(10):1518–1526
Matias-Guiu JA, Cortés-Martínez A, Valles-Salgado M, Rognoni T, Fernández-Matarrubia M, Moreno-Ramos T, Matías-Guiu J (2017) Addenbrooke’s cognitive examination III: diagnostic utility for mild cognitive impairment and dementia and correlation with standardized neuropsychological tests. Int Psychogeriatr 29(1):105–113
Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease (2003) The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord 18(7):738–750
Pellecchia MT, Savastano R, Moccia M et al (2016) Lower serum uric acid is associated with mild cognitive impairment in early Parkinson’s disease: a 4-year follow-up study. J Neural Transm 123(12):1399–1402
QRISK®2-2016 risk calculator, downloaded on 31 Jan 2016. https://qrisk.org/2016/
Rektor I, Goldemund D, Sheardová K, Rektorová I, Michálková Z, Dufek M (2009) Vascular pathology in patients with idiopathic Parkinson’s disease. Parkinsonism Relat Disord 15(1):24–29
Rektor I, Goldemund D, Bednařík P et al (2012) Impairment of brain vessels may contribute to mortality in patients with Parkinson’s disease. Mov Disord 27(9):1169–1172
Schlesinger I, Schlesinger N (2008) Uric acid in Parkinson’s disease. Mov Disord 23(12):1653–1657
Swallow DM, Lawton MA, Grosset KA et al (2016) Statins are underused in recent-onset Parkinson’s disease with increased vascular risk: findings from the UK Tracking Parkinson’s and Oxford Parkinson’s Disease Centre (OPDC) discovery cohorts. J Neurol Neurosurg Psychiatry 87(11):1183–1190
Veselý B, Dufek M, Thon V et al (2018) Interleukin 6 and complement serum level study in Parkinson’s disease. J Neural Transm 125(5):875–881
Wechsler D (1997) Wechsler memory scale the third edition, WMS-III. The Psychological Corporation, San Antonio
Xie Y, Feng H, Peng S, Xiao J, Zhang J (2017) Association of plasma homocysteine, vitamin B12 and folate levels with cognitive function in Parkinson’s disease: a meta-analysis. Neurosci Lett 636:190–195
Acknowledgments
Support was provided by the project CEITEC 2020 (LQ1601) and grant AZV 16-33798A. We would like to thank Ján Necpal, M.D., from the Neurology Department, Zvolen, Slovak Republic for kindly referring patients for participation in this study. We also would like to thank to Anne Johnson for grammatical assistance and for reviewing the manuscript.
Funding
Support was provided by the project CEITEC 2020 (LQ1601) and grant Ministry of Health of Czech Republic AZV 16-33798A.
Author information
Authors and Affiliations
Contributions
(1) Research project: A. conception, B. organization, C. execution; (2) statistical analysis: A. design, B. execution, C. review and critique; (3) manuscript: A. writing of the first draft, B. review and critique. BV: 1A, 1B, 1C, 2C, 3A. EK: 2A, 2B, 2C. VD: 1C. SK: 1C. PV: 3B. EK: 3B. IR: 1A,1B, 1C, 2A, 3B.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors report any conflict of interest or financial disclosure.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Veselý, B., Koriťáková, E., Bohnen, N.I. et al. The contribution of cerebrovascular risk factors, metabolic and inflammatory changes to cognitive decline in Parkinson’s disease: preliminary observations. J Neural Transm 126, 1303–1312 (2019). https://doi.org/10.1007/s00702-019-02043-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-019-02043-7