Abstract
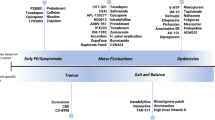
The contribution of non-dopaminergic degeneration to disability in Parkinson’s disease (PD) is still debated. It has been argued that no additional advance can be expected in the management of PD by the development of new dopaminergic agents and suggested that future research should mainly focus on therapies targeting the non-dopaminergic systems involved in the pathogenesis of levodopa resistant motor and non-motor symptoms. We believe this is only partially true and the achievement of a stable dopaminergic restoration and modulation of the dopaminergic system is still an important, unmet need of current pharmacological therapies in PD. Currently available oral levodopa and dopamine agonist medications provide insufficient benefit, as the therapeutic window progressively narrows and motor fluctuations eventually develop in most patients. Conversely, the application of infusion and surgical therapies is limited by selective indications and possible irreversible adverse events and device-related problems. Research of new, safer and less invasive strategies, able to modulate the dopaminergic circuits, would certainly improve the management of motor complications, and most importantly such treatments would be also beneficial to axial and non-motor symptoms, which are universally regarded as the major cause of PD functional disability. Indeed, gait and balance problems may improve with dopaminergic treatment in most patients and they become unresponsive only at the very late stages of the disease. Moreover, several non-motor disturbances, including cognition and depression are often linked to oscillation of dopamine concentrations, and are frequently relieved by treatments providing continuous dopaminergic delivery. Finally, drug trials testing non-dopaminergic treatments for motor and non-motor symptoms of PD provided so far disappointing results. Despite the impressive advances of PD therapeutic strategy, we think there is still need for safe, non-invasive and easily manageable dopaminergic treatments able to provide constant dopamine receptor stimulation and ensure a more stable control of dopamine responsive motor and non-motor symptoms at any stage of the disease.
Similar content being viewed by others
References
Ahlskog JE (2007) Beating a dead horse: dopamine and Parkinson disease. Neurology 69(17):1701–1711
Antonini A, Tolosa E (2009) Apomorphine and levodopa infusion therapies for advanced Parkinson’s disease: selection criteria and patient management. Expert Rev Neurother 9(6):859–867
Antonini A, Chaudhuri KR, Martinez-Martin P, Odin P (2010) Oral and infusion levodopa-based strategies for managing motor complications in patients with Parkinson’s disease. CNS Drugs 24(2):119–129
Antonini A, Isaias IU, Rodolfi G, Landi A, Natuzzi F, Siri C, Pezzoli G (2011) A 5-year prospective assessment of advanced Parkinson disease patients treated with subcutaneous apomorphine infusion or deep brain stimulation. J Neurol 258(4):579–585
Antonini A, Barone P, Marconi R et al (2012) The progression of non-motor symptoms in Parkinson’s disease and their contribution to motor disability and quality of life. J Neurol
Barone P, Scarzella L, Marconi R, Antonini A, Morgante L, Bracco F, Zappia M, Depression/Parkinson Italian Study Group (2006) Pramipexole versus sertraline in the treatment of depression in Parkinson’s disease: a national multicenter parallel-group randomized study. J Neurol 253(5):601–607
Bayulkem K, Lopez G (2010) Nonmotor fluctuations in Parkinson’s disease: clinical spectrum and classification. J Neurol Sci 289(1–2):89–92
Chapuis S, Ouchchane L, Metz O, Gerbaud L, Durif F (2005) Impact of the motor complications of Parkinson’s disease on the quality of life. Mov Disord 20(2):224–230
Coelho M, Marti MJ, Tolosa E, Ferreira JJ, Valldeoriola F, Rosa M, Sampaio C (2010) Late-stage Parkinson’s disease: the Barcelona and Lisbon cohort. J Neurol 257(9):1524–1532
Costa A, Peppe A, Dell’Agnello G et al (2003) Dopaminergic modulation of visual-spatial working memory in Parkinson’s disease. Dement Geriatr Cogn Disord 15:55–66
Czernecki V, Pillon B, Houeto JL, Pochon JB, Levy R, Dubois B (2002) Motivation, reward, and Parkinson’s disease: influence of dopatherapy. Neuropsychologia 40(13):2257–2267
Ferreira JJ, Rascol O, Poewe W, Sampaio C, Rocha JF, Nunes T, Almeida L, BIA-3202-202 Study Investigators (2010) A double-blind, randomized, placebo and active-controlled study of nebicapone for the treatment of motor fluctuations in Parkinson’s disease. CNS Neurosci Ther 16(6):337–347
Fox SH, Katzenschlager R, Lim SY, Ravina B, Seppi K, Coelho M, Poewe W, Rascol O, Goetz CG, Sampaio C (2011) The movement disorder society evidence-based medicine review update: treatments for the motor symptoms of Parkinson’s disease. Mov Disord 26(Suppl 3):S2–S41
Giladi N (2008) Medical treatment of freezing of gait. Mov Disord 23(Suppl 2):S482–S488
Hauser RA (2012) IPX066: a novel carbidopa-levodopa extended-release formulation. Expert Rev Neurother 12(2):133–140
Hely MA, Morris JG, Reid WG, Trafficante R (2005) Sydney multicenter study of Parkinson’s disease: non-l-dopa-responsive problems dominate at 15 years. Mov Disord 20:190–199
Hely MA, Reid WG, Adena MA, Halliday GM, Morris JG (2008) The Sydney multicenter study of Parkinson’s disease: the inevitability of dementia at 20 years. Mov Disord 23(6):837–844
Hilker R, Antonini A, Odin P (2011) What is the best treatment for fluctuating Parkinson’s disease: continuous drug delivery or deep brain stimulation of the subthalamic nucleus? J Neural Trans 118(6):907–914
Honig H, Antonini A, Martinez-Martin P, Forgacs I, Faye GC, Fox T, Fox K, Mancini F, Canesi M, Odin P, Chaudhuri KR (2009) Intrajejunal levodopa infusion in Parkinson’s disease: a pilot multicenter study of effects on nonmotor symptoms and quality of life. Mov Disord 24(10):1468–1474
Juri C, Rodriguez-Oroz M, Obeso JA (2010) The pathophysiological basis of sensory disturbances in Parkinson’s disease. J Neurol Sci 289(1–2):60–65
Lange KW, Robbins TW, Marsden CD et al (1992) l-Dopa withdrawal in Parkinson’s disease selectively impairs cognitive performance in tests sensitive to frontal lobe dysfunction. Psychopharmacology 107:394–404
Manca D, Cossu G, Murgia D, Molari A, Ferrigno P, Marcia E, Melis M (2009) Reversible encephalopathy and axonal neuropathy in Parkinson’s disease during duodopa therapy. Mov Disord 24(15):2293–2294
Menza MA, Sage J, Marshall E, Cody R, Duvoisin R (1990) Mood changes and “on–off” phenomena in Parkinson’s disease. Mov Disord 5:148–151
Molloy SA, Rowan EN, O’Brien JT, McKeith IG, Wesnes K, Burn DJ (2006) Effect of levodopa on cognitive function in Parkinson’s disease with and without dementia and dementia with Lewy bodies. J Neurol Neurosurg Psychiatry 77(12):1323–1328
Nakata Y, Yasuda T, Mochizuki H (2012) Recent progress in gene therapy for Parkinson’s disease. Curr Mol Med
Nissenbaum H, Quinn NP, Brown RG, Toone B, Gotham A-M, Marsden CD (1987) Mood swings associated with the “on–off” phenomenon in Parkinson’s disease. Psychol Med 17:899–904
Nutt JG, Bloem BR, Hallet M, Horak FB, Nieuboer A (2011) Freezing of gait: moving forward on a mysterious clinical phenomena. Lancet Neurol 10:734–744
Piacentino M, Pilleri M, Bartolomei L (2011) Hardware-related infections after deep brain stimulation surgery: review of incidence, severity and management in 212 single-center procedures in the first year after implantation. Acta Neurochir (Wien) 153(12):2337–2341
Remy P, Doder M, Lees A, Turjanski N, Brooks D (2005) Depression in Parkinson’s disease: loss of dopamine and noradrenaline innervation in the limbic system. Brain 128:1314–1322
Richard IH, Frank S, McDermott MP, Wang H, Justus AW, Ladonna KA et al (2004) The ups and downs of Parkinson’s disease: a prospective study of mood and anxiety fluctuations. Cogn Behav Neurol 17:201–207
Sakakibara R, Uchiyama T, Yamanishi T, Shirai K, Hattori T (2008) Bladder and bowel dysfunction in Parkinson’s disease. J Neural Trans 115(3):443–460
Seppi K, Weintraub D, Coelho M, Perez-Lloret S, Fox SH, Katzenschlager R, Hametner EM, Poewe W, Rascol O, Goetz CG, Sampaio C (2011) The movement disorder society evidence-based medicine review update: treatments for the non-motor symptoms of Parkinson’s disease. Mov Disord 26(Suppl 3):S42–S80
Stocchi F, Borgohain R, Onofrj M, Schapira AH, Bhatt M, Lucini V, Giuliani R, Study 015 Investigators (2012) A randomized, double-blind, placebo-controlled trial of safinamide as add-on therapy in early Parkinson’s disease patients. Mov Disord 27(1):106–112
Trenkwalder C, Kies B, Rudzinska M, Fine J, Nikl J, Honczarenko K, Dioszeghy P, Hill D, Anderson T, Myllyla V, Kassubek J, Steiger M, Zucconi M, Tolosa E, Poewe W, Surmann E, Whitesides J, Boroojerdi B, Recover Study Group (2011) Rotigotine effects on early morning motor function and sleep in Parkinson’s disease: a double-blind, randomized, placebo-controlled study (RECOVER). Mov Disord 26(1):90–99
Witt K, Daniels C, Reiff J, Krack P, Volkmann J, Pinsker MO, Krause M, Tronnier V, Kloss M, Schnitzler A, Wojtecki L, Bötzel K, Danek A, Hilker R, Sturm V, Kupsch A, Karner E, Deuschl G (2008) Neuropsychological and psychiatric changes after deep brain stimulation for Parkinson’s disease: a randomised, multicentre study. Lancet Neurol 7(7):605–614
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pilleri, M., Koutsikos, K. & Antonini, A. Is there room for new non-dopaminergic treatments in Parkinson’s disease?. J Neural Transm 120, 349–352 (2013). https://doi.org/10.1007/s00702-012-0947-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-012-0947-z