Abstract
The phylogenetic position of two Aspergillus strains isolated from Australian soil and phenotypically resembling A. unilateralis was investigated by using multigene phylogeny based on β-tubulin (benA), calmodulin (CaM), actin (act), and RNA polymerase II second largest subunit (RPB2) genes. The analysis supported their placement into a separate lineage within a well-supported clade containing 10 other members of section Fumigati (“A. unilateralis clade”). Comparisons of extrolite profiles, taxonomically informative morphological and physiological characters were made, and it was discovered that the two strains can be differentiated from all relatives by their low maximum growth temperature, short stipes, and ornamentation of conidia. The data justified the proposal of a new species, A. tasmanius sp. nov. Amplification of mating-type genes showed that the A. unilateralis clade contains five heterothallic species. Only the MAT1-1-1 idiomorph was detected among isolates of A. unilateralis, A. tasmanicus, and A. marvanovae, while isolates having both opposite mating types were detected in A. turcosus and A. nishimurae. The sexual state of A. turcosus was induced by mating experiments and is described in this study. Ascospores of this species were unique by their smooth to finely verrucose convex surface and two well-visible equatorial crests. Some exometabolites detected in A. marvanovae and A. tasmanicus are also indicative of a perfect state, thus supporting the hypothesis that these species have cryptic sexual cycles. The epitype and ex-epitype culture is designated for A. nishimurae to facilitate further taxonomic work with this species.






Similar content being viewed by others
References
Alcazar-Fuoli L, Mellado E, Alastruey-Izquierdo A, Cuenca-Estrella M, Rodriguez-Tudela JL (2008) Aspergillus section Fumigati: antifungal susceptibility patterns and sequence-based identification. Antimicrob Agents Chemotherapy 52:1244–1251. doi:10.1128/AAC.00942-07
Anzawa K, Kawasaki M, Mochizuki T, Ishizaki H (2010) Successful mating of Trichophyton rubrum with Arthroderma simii. Med Mycol 48:629–634
Balajee SA, Gribskov J, Brandt M, Ito J, Fothergill A, Marr KA (2005a) Mistaken identity: Neosartorya pseudofischeri and its anamorph masquerading as Aspergillus fumigatus. J Clin Microbiol 43:5996–5999. doi:10.1128/JCM.43.12.5996-5999.2005
Balajee SA, Gribskov JL, Hanley E, Nickle D, Marr KA (2005b) Aspergillus lentulus sp. nov., a new sibling species of A. fumigatus. Eukar Cell 4:625–632. doi:10.1128/EC.4.3.625-632.2005
Balajee SA, Nickle D, Varga J, Marr KA (2006) Molecular studies reveal frequent misidentification of Aspergillus fumigatus by morphotyping. Eukar Cell 5:1705–1712. doi:10.1128/EC.00162-06
Balajee SA, Kano R, Baddley JW, Moser SA, Marr KA, Alexander BD, Andes D, Kontoyiannis DP, Perrone G, Peterson S, Brandt ME, Pappas PG, Chiller T (2009) Molecular identification of Aspergillus species collected for the transplant-associated infection surveillance network. J Clin Microbiol 47:3138–3141. doi:10.1128/JCM.01070-09
Barrs VR, van Doorn TM, Houbraken J, Kidd SE, Martin P, Pinheiro MD, Richardson M, Varga J, Samson RA (2013) Aspergillus felis sp. nov., an emerging agent of invasive aspergillosis in humans, cats and dogs. PLoS ONE 8:e64871. doi:10.1371/journal.pone.0064871
Capon RJ, Ratnayake R, Stewart M, Lacey E, Tennant S, Gill JH (2005) Aspergillazines A–E: novel heterocyclic dipeptides from an Australian strain of Aspergillus unilateralis. Organic Biomolec Chem 3:123–129. doi:10.1039/B413440K
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91:553–556. doi:10.2307/3761358
Chen A, Frisvad J, Sun B, Varga J, Kocsubé S, Dijksterhuis J, Kim D, Hong S-B, Houbraken J, Samson R (2016) Aspergillus section Nidulantes (formerly Emericella): polyphasic taxonomy, chemistry and biology. Stud Mycol 84:1–118. doi:10.1016/j.simyco.2016.10.001
Dethoup T, Gomes NGM, Chaopongpang S, Kijjoa A (2016) Aspergillus similanensis sp. nov. from a marine sponge in Thailand. Mycotaxon 131:7–15. doi:10.5248/131.7
Dyer PS, O’Gorman CM (2011) A fungal sexual revolution: Aspergillus and Penicillium show the way. Curr Opin Microbiol 14:649–654. doi:10.1016/j.mib.2011.10.001
Frisvad JC, Larsen TO (2015a) Extrolites of Aspergillus fumigatus and other pathogenic species in Aspergillus section Fumigati. Frontiers Microbiol 6:1485. doi:10.3389/fmicb.2015.01485
Frisvad JC, Larsen TO (2015b) Chemodiversity in the genus Aspergillus. Appl Microbiol Biotechnol 99:7859–7877. doi:10.1007/s00253-015-6839-z
Frisvad JC, Thrane U (1987) Standardized high-performance liquid chromatography of 182 mycotoxins and other fungal metabolites based on alkylphenone retention indices and UV–Vis spectra (diode array detection). J Chromatogr A 404:195–214
Frisvad JC, Thrane U (1993) Liquid column chromatography of mycotoxins. In: Betina V (ed) Chromatography of mycotoxins: techniques and applications. J Chromatogr Libr 54:253–372. doi:10.1016/S0301-4770(08)60568-0
Frisvad JC, Rank C, Nielsen KF, Larsen TO (2009) Metabolomics of Aspergillus fumigatus. Med Mycol 47:S53–S71. doi:10.1080/13693780802307720
Gautier M, Normand A-C, Ranque S (2016) Previously unknown species of Aspergillus. Clin Microbiol Infect 22:662–669. doi:10.1016/j.cmi.2016.05.013
Glass NL, Donaldson GC (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl Environm Microbiol 61:1323–1330
Guarro J, Kallas EG, Godoy P, Karenina A, Gene J, Stchigel A, Colombo AL (2002) Cerebral aspergillosis caused by Neosartorya hiratsukae, Brazil. Emerging Infect Dis 8:989–992
Han JI, Na KJ (2008) Dermatitis caused by Neosartorya hiratsukae infection in a hedgehog. J Clin Microbiol 46:3119–3123. doi:10.1128/JCM.00737-08
Hearn V, Moutaouakil M, Latgé J-P (1990) Analysis of components of Aspergillus and Neosartorya mycelial preparations by gel electrophoresis and Western blotting procedures. In: Samson RA, Pitt JI (eds) Modern concepts in Penicillium and Aspergillus classification. Springer, Berlin, pp 235–247. doi:10.1007/978-1-4899-3579-3_20
Hong SB, Go SJ, Shin HD, Frisvad JC, Samson RA (2005) Polyphasic taxonomy of Aspergillus fumigatus and related species. Mycologia 97:1316–1329
Hong SB, Shin HD, Hong J, Frisvad JC, Nielsen PV, Varga J, Samson RA (2008) New taxa of Neosartorya and Aspergillus in Aspergillus section Fumigati. Antonie van Leeuwenhoek J Microbiol Serol 93:87–98. doi:10.1007/s10482-007-9183-1
Hong S-B, Kim D-H, Park I-C, Samson RA, Shin H-D (2010) Isolation and identification of Aspergillus section Fumigati strains from arable soil in Korea. Mycobiology 38:1–6. doi:10.4489/MYCO.2010.38.1.001
Houbraken J, Spierenburg H, Frisvad JC (2012) Rasamsonia, a new genus comprising thermotolerant and thermophilic Talaromyces and Geosmithia species. Antonie van Leeuwenhoek J Microbiol Serol 101:403–421. doi:10.1007/s10482-011-9647-1
Houbraken J, Weig M, Groß U, Meijer M, Bader O (2016) Aspergillus oerlinghausenensis, a new mould species closely related to A. fumigatus. FEMS Microbiol Lett 363:fnv236. doi:10.1093/femsle/fnv236
Hubka V, Kolařík M (2012) β-tubulin paralogue tubC is frequently misidentified as the benA gene in Aspergillus section Nigri taxonomy: primer specificity testing and taxonomic consequences. Persoonia 29:1–10. doi:10.3767/003158512X658123
Hubka V, Kubatova A, Mallatova N, Sedlacek P, Melichar J, Skorepova M, Mencl K, Lyskova P, Sramkova B, Chudickova M, Hamal P, Kolarik M (2012) Rare and new aetiological agents revealed among 178 clinical Aspergillus strains obtained from Czech patients and characterised by molecular sequencing. Med Mycol 50:601–610. doi:10.3109/13693786.2012.667578
Hubka V, Peterson SW, Frisvad JC, Yaguchi T, Kubátová A, Kolařík M (2013) Aspergillus waksmanii sp. nov. and Aspergillus marvanovae sp. nov., two closely related species in section Fumigati. Int J Syst Evol Microbiol 63:783–789. doi:10.1099/ijs.0.047076-0
Hubka V, Lyskova P, Frisvad JC, Peterson SW, Skorepova M, Kolarik M (2014) Aspergillus pragensis sp. nov. discovered during molecular re-identification of clinical isolates belonging to Aspergillus section Candidi. Med Mycol 52:565–576. doi:10.1093/mmy/myu022
Hubka V, Nissen C, Jensen R, Arendrup M, Cmokova A, Kubatova A, Skorepova M (2015a) Discovery of a sexual stage in Trichophyton onychocola, a presumed geophilic dermatophyte isolated from toenails of patients with a history of T. rubrum onychomycosis. Med Mycol 53:798–809. doi:10.1093/mmy/myv044
Hubka V, Nováková A, Kolařík M, Jurjević Ž, Peterson SW (2015b) Revision of Aspergillus section Flavipedes: seven new species and proposal of section Jani sect. nov. Mycologia 107:169–208. doi:10.3852/14-059
Hubka V, Nováková A, Peterson SW, Frisvad JC, Sklenář F, Matsuzawa T, Kubátová A, Kolařík M (2016) A reappraisal of Aspergillus section Nidulantes with descriptions of two new sterigmatocystin producing species. Pl Syst Evol 302:1267–1299. doi:10.1007/s00606-016-1331-5
Jurjević Ž, Kubátová A, Kolařík M, Hubka V (2015) Taxonomy of Aspergillus section Petersonii sect. nov. encompassing indoor and soil-borne species with predominant tropical distribution. Pl Syst Evol 301:2441–2462. doi:10.1007/s00606-015-1248-4
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Molec Biol Evol 30:772–780. doi:10.1093/molbev/mst010
Kocsubé S, Perrone G, Magistà D, Houbraken J, Varga J, Szigeti G, Hubka V, Hong S-B, Frisvad J, Samson R (2016) Aspergillus is monophyletic: evidence from multiple gene phylogenies and extrolites profiles. Stud Mycol 85:199–213. doi:10.1016/j.simyco.2016.11.006
Kornerup A, Wanscher JH (1967) Methuen handbook of colour. Methuen & Co., Ltd., London
Koutroutsos K, Arabatzis M, Bougatsos G, Xanthaki A, Toutouza M, Velegraki A (2010) Neosartorya hiratsukae peritonitis through continuous ambulatory peritoneal dialysis. J Med Microbiol 59:862–865. doi:10.1099/jmm.0.019133-0
Kretzer A, Li Y, Szaro T, Bruns TD (1996) Internal transcribed spacer sequences from 38 recognized species of Suillus sensu lato: phylogenetic and taxonomic implications. Mycologia 88:776–785. doi:10.2307/3760972
Kwon-Chung K, Kim SJ (1974) A second heterothallic Aspergillus. Mycologia 66:628–638. doi:10.2307/3758166
Kwon-Chung KJ, Sugui JA (2009) Sexual reproduction in Aspergillus species of medical or economical importance: why so fastidious? Trends Microbiol 17:481–487. doi:10.1016/j.tim.2009.08.004
Lanfear R, Calcott B, Ho SY, Guindon S (2012) PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Molec Biol Evol 29:1695–1701. doi:10.1093/molbev/mss020
Liu YJ, Whelen S, Hall BD (1999) Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit. Molec Biol Evol 16:1799–1808. doi:10.1093/oxfordjournals.molbev.a026092
Malejczyk K, Sigler L, Gibas CFC, Smith SW (2013) Invasive sino-orbital mycosis in an aplastic anemia patient caused by Neosartorya laciniosa. J Clin Microbiol 51:1316–1319. doi:10.1128/JCM.02919-12
Matsuzawa T, Campos Takaki GM, Yaguchi T, Okada K, Gonoi T, Horie Y (2013) Two new species of Aspergillus section Fumigati isolated from caatinga soil in the State of Pernambuco, Brazil. Mycoscience 55:79–88. doi:10.1016/j.myc.2013.04.001
Matsuzawa T, Takaki GMC, Yaguchi T, Okada K, Abliz P, Gonoi T, Horie Y (2015) Aspergillus arcoverdensis, a new species of Aspergillus section Fumigati isolated from caatinga soil in State of Pernambuco, Brazil. Mycoscience 56:123–131. doi:10.1016/j.myc.2014.04.006
Mayamor S, Poeaim S (2014) Morphological and molecular phylogeny studies on Eurotiales isolated from soil. J Agr Tech 10:189–195
McLennan EI, Tucker SC, Thrower LB (1954) New soil fungi from Australian heathland: Aspergillus, Penicillium, Spegazzinia. Austral J Bot 2:355–364. doi:10.1071/BT9540355
Meyer V, Wu B, Ram AF (2011) Aspergillus as a multi-purpose cell factory: current status and perspectives. Biotechnol Lett 33:469–476. doi:10.1007/s10529-010-0473-8
Mirza J, Bajwa R (2005) New records of Fungi Imperfecti from Pakistan. Res Bull 1st Fung Cult Bank Pak 1:1–15
Mühlenfeld A, Achenbacht H (1988) Asperpentyn, a novel acetylenic cyclohexene epoxide from Aspergillus duricaulis. Phytochemistry 27:3853–3855. doi:10.1016/0031-9422(88)83031-0
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Molec Biol Evol 32:268–274. doi:10.1093/molbev/msu300
Nielsen KF, Månsson M, Rank C, Frisvad JC, Larsen TO (2011) Dereplication of microbial natural products by LC-DAD-TOFMS. J Nat Prod 74:2338–2348. doi:10.1021/np200254t
Nováková A, Hubka V, Dudová Z, Matsuzawa T, Kubátová A, Yaguchi T, Kolařík M (2014) New species in Aspergillus section Fumigati from reclamation sites in Wyoming (USA) and revision of A. viridinutans complex. Fungal Diversity 64:253–274. doi:10.1007/s13225-013-0262-5
O’Gorman CM, Fuller HT, Dyer PS (2009) Discovery of a sexual cycle in the opportunistic fungal pathogen Aspergillus fumigatus. Nature 457:471–474. doi:10.1038/nature07528
Olarte RA, Worthington CJ, Horn BW, Moore GG, Singh R, Monacell JT, Dorner JW, Stone EA, Xie DY, Carbone I (2015) Enhanced diversity and aflatoxigenicity in interspecific hybrids of Aspergillus flavus and Aspergillus parasiticus. Molec Ecol 24:1889–1909. doi:10.1111/mec.13153
Peterson SW (2008) Phylogenetic analysis of Aspergillus species using DNA sequences from four loci. Mycologia 100:205–226
Pitt JI, Hocking AD (2009a) Aspergillus and related teleomorphs. In: Pitt JI, Hocking AD (eds) Fungi and food spoilage. Springer, London, pp 275–337. doi:10.1007/978-0-387-92207-2_8
Pitt JI, Hocking AD (2009b) Spoilage of stored, processed and preserved foods. In: Pitt JI, Hocking AD (eds) Fungi and food spoilage. Springer, London, pp 401–421. doi:10.1007/978-0-387-92207-2_12
Réblová M, Hubka V, Thureborn O, Lundberg J, Sallstedt T, Wedin M, Ivarsson M (2016) From the tunnels into the treetops: new lineages of black yeasts from biofilm in the Stockholm metro system and their relatives among ant-associated fungi in the Chaetothyriales. PLoS ONE 11:e0163396. doi:10.1371/journal.pone.0163396
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. doi:10.1093/sysbio/sys029
Salomão BC, Slongo AP, Aragão GM (2007) Heat resistance of Neosartorya fischeri in various juices. LWT Food Sci Technol 40:676–680. doi:10.1016/j.lwt.2006.03.008
Samson RA, Hong S, Peterson SW, Frisvad JC, Varga J (2007) Polyphasic taxonomy of Aspergillus section Fumigati and its teleomorph Neosartorya. Stud Mycol 59:147–203. doi:10.3114/sim.2007.59.14
Samson RA, Houbraken J, Thrane U, Frisvad JC, Andersen B (2010) Food and indoor fungi. CBS KNAW Biodiversity Center, Utrecht
Samson RA, Visagie CM, Houbraken J, Hong SB, Hubka V, Klaassen CHV, Perrone G, Seifert KA, Tanney JB, Varga J, Yaguchi T, Frisvad JC (2014) Phylogeny, identification and nomenclature of the genus Aspergillus. Stud Mycol 78:141–173. doi:10.1016/j.simyco.2014.07.004
Santoianni J, Predari S, Verón D, Zucchini A, De Paulis A (2008) A 15 year-review of peritoneal dialysis-related peritonitis: microbiological trends and patterns of infection in a teaching hospital in Argentina. Revista Argent Microbiol 40:17–23
Sheng-Liang Z, Shu-Zhen Y, Zhen-Ying W, Shuang-Lin C (2014) Endophytic fungi associated with Macrosolen tricolor and its host Camellia oleifera. World J Microbiol Biotechnol 30:1775–1784. doi:10.1007/s11274-014-1600-9
Shivaprakash MR, Jain N, Gupta S, Baghela A, Gupta A, Chakrabarti A (2009) Allergic fungal rhinosinusitis caused by Neosartorya hiratsukae from India. Med Mycol 47:317–320. doi:10.1080/13693780802562977
Sugui JA, Vinh DC, Nardone G, Shea YR, Chang YC, Zelazny AM, Marr KA, Holland SM, Kwon-Chung KJ (2010) Neosartorya udagawae (Aspergillus udagawae), an emerging agent of aspergillosis: how different is it from Aspergillus fumigatus? J Clin Microbiol 48:220–228. doi:10.1128/JCM.01556-09
Sugui J, Peterson S, Figat A, Hansen B, Samson R, Mellado E, Cuenca-Estrella M, Kwon-Chung K (2014) Genetic relatedness versus biological compatibility between Aspergillus fumigatus and related species. J Clin Microbiol 52:3707–3721. doi:10.1128/JCM.01704-14
Sutton D et al (2002) First report of Neosartorya spinosa (Raper & Fennell) Kozakiewicz inciting human disease. American Society for Microbiology General Meeting, Salt Lake City, USA
Swilaiman SS, O’Gorman CM, Balajee SA, Dyer PS (2013) Discovery of a sexual cycle in Aspergillus lentulus, a close relative of A. fumigatus. Eukar Cell 12:962–969. doi:10.1128/EC.00040-13
Szewczyk E, Krappmann S (2010) Conserved regulators of mating are essential for Aspergillus fumigatus cleistothecium formation. Eukar Cell 9:774–783. doi:10.1128/EC.00375-09
Takada M, Horie Y, Abliz P (2001) Two new heterothallic Neosartorya from African soil. Mycoscience 42:361–367. doi:10.1007/BF02461219
Udagawa S, Tsubouchi H, Horie Y (1991) Neosartorya hiratsukae, a new species of food-borne Ascomycetes. Trans Mycol Soc Japan 32:23–29
Verdejo-Lucas S, Blanco M, Talavera M, Stchigel A, Sorribas F (2013) Fungi recovered from root-knot nematodes infecting vegetables under protected cultivation. Biocontrol Sci Technol 23:277–287. doi:10.1080/09583157.2012.756459
Visagie CM, Hirooka Y, Tanney JB, Whitfield E, Mwange K, Meijer M, Amend AS, Seifert KA, Samson RA (2014) Aspergillus, Penicillium and Talaromyces isolated from house dust samples collected around the world. Stud Mycol 78:63–139. doi:10.1016/j.simyco.2014.07.002
Wang Q, Gong J, Chisti Y, Sirisansaneeyakul S (2015) Fungal isolates from a pu-erh type tea fermentation and their ability to convert tea polyphenols to theabrownins. J Food Sci 80:M809–M817. doi:10.1111/1750-3841.12831
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA et al (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Yaguchi T, Someya A, Si Udagawa (1994) A new species of Neosartorya from Taiwan soil. Mycoscience 35:309–313. doi:10.1007/BF02268499
Yaguchi T, Horie Y, Tanaka R, Matsuzawa T, Ito J, Nishimura K (2007) Molecular phylogenetics of multiple genes on Aspergillus section Fumigati isolated from clinical specimens in Japan. Jap J Med Mycol 48:37–46. doi:10.3314/jjmm.48.37
Yaguchi T, Matsuzawa T, Tanaka R, Abliz P, Hui Y, Horie Y (2010) Two new species of Neosartorya isolated from soil in Xinjiang, China. Mycoscience 51:253–262. doi:10.1007/s10267-010-0037-8
Acknowledgements
This research was supported by the project of Charles University Grant Agency (GAUK 1130214), and the project BIOCEV (CZ.1.05/1.1.00/02.0109) provided by the Ministry of Education, Youth and Sports of CR and ERDF. Prof Karol Marhold’s consultation on nomenclatural issues is gratefully acknowledged. We thank Milada Chudíčková, Alena Gabrielová and Kyung-youn Hong for their invaluable assistance in laboratory and Veronica Jurjević for assistance with editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Handling editor: Stephen W. Peterson.
Information on Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Online Resource 1
Supplementary material 1 (DOC 47 kb)
Online Resource 2
Supplementary material 2 (PDF 21 kb)
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
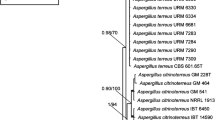
Online Resource 1. A concatenated alignment of β-tubulin (benA), calmodulin (CaM), actin (act) and RNA polymerase II second largest subunit (RPB2) genes used for construction of tree on Fig. 1: benA: positions 1–537; CaM: positions 538–1231; act: positions 1232–1669; RPB2: positions 1670–2667.
Online Resource 2. Secondary metabolites detected in species from Aspergillus unilateralis clade and their absorption maxima, and bracketed retention indexes (RI) based on a series of alkylphenones (Frisvad and Thrane 1987, 1993).
Rights and permissions
About this article
Cite this article
Hubka, V., Dudová, Z., Kubátová, A. et al. Taxonomic novelties in Aspergillus section Fumigati: A. tasmanicus sp. nov., induction of sexual state in A. turcosus and overview of related species. Plant Syst Evol 303, 787–806 (2017). https://doi.org/10.1007/s00606-017-1400-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-017-1400-4