Abstract
Purposes
Outcomes following lung transplantation are limited by bronchiolitis obliterans syndrome (BOS). As the number of circulating regulatory T cells (Treg) is lower in lung recipients with BOS than in stable lung recipients, we hypothesized that Treg is also correlated with lung function in the early post-transplantation period.
Methods
This prospective study included 18 consecutive patients whose lung function parameters were recorded 3 weeks and 3 months after transplantation, between February and July 2007. Peripheral blood mononuclear cells were stained with anti-CD3, -CD4, -CD8, -CD19, -CD25, -CD28, -CD45RA, -CD45RO, -CD69, -CD127, -CTLA4, and -Foxp3 antibodies and FACS assays were performed. In addition, intracellular cytokines were stained for FACS.
Results
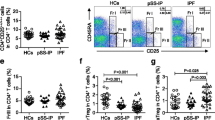
Treg-specific markers (Foxp3, CD127lo, and CTLA4) in the CD25+CD4+ population were correlated with both forced expiratory volume in 1 s and forced vital capacity. Th1-cytokine secretion was more dominant in CD4+CD25+ T cells than in CD4+CD25− T cells. In contrast, Th2 and Treg cytokine secretion was the dominant response in stable recipients.
Conclusions
The frequency of Treg cells was positively correlated with good lung function in the early period after lung transplantation.


Similar content being viewed by others
References
Haverich A. Experience with lung transplantation. Ann Thorac Surg. 1999;67:305–12.
Koop C, Haverich A, Fischer S. Immunosuppressive therapy after human lung transplantation. Eur Respir J. 2004;23:159–71.
Trulock EP, Edwards LB, Taylor DO, Boucek MM, Keck BM, Hertz MI. The registry of the international society for heart and lung transplantation: twenty-first official adult lung and heart–lung transplant report-2004. J Heart Lung Transplant. 2004;23:804–15.
Estenne M, Maurer JR, Boehler A, Egan JJ, Frost A, Hertz M, et al. Bronchiolitis Obliterans Syndrome 2001: an update of the diagnostic criteria. J Heart Lung Transplant. 2002;21:297–310.
Jackson CH, Sharples LD, MaNeil K, Stewart S, Wallwork J. Acute and chronic onset of bronchiolitis obliterans syndrome (BOS): are they different entities? J Heart Lung Transplant. 2002;21:658–66.
Meloni F, Vitulo P, Bianco AM, Paschetto E, Morosini M, Cascina A, et al. Regulatory CD4+CD25+ T cells in the peripheral blood of lung transplant recipients: correlation with transplant outcome. Transplantation. 2004;77:762–6.
Otulana BA, Higenbottam T, Scott J, Clelland C, Igboaka G, Wallwork J. Lung function associated with histologically diagnosed acute lung rejection and pulmonary infection in heart–lung transplant patients. Am Rev Respir Dis. 1990;142:329–32.
Reed AJ, Noorchashm H, Rostami SY, Zarrabi Y, Perate AR, Jeganathan AN, et al. Alloreactive CD4 T cell activation in vivo: an autonomous function of the indirect pathway of alloantigen presentation. J Immunol. 2003;171:6502–9.
Caton AJ, Swartzentruber JR, Kuhl AL, Carding SR, Stark SE. Activation and negative selection of functionally distinct subsets of antibody-secreting cells by influenza hemagglutinin as a vital and a neo-self antigen. J Exp Med. 1996;183:13–26.
Wood KJ, Sakaguchi S. Regulatory T cells in transplantation tolerance. Nat Rev Immunol. 2003;3(3):199–210.
Sakaguchi S, Sakaguchi N, Shimizu J, Yamazaki S, Sakihama T, Itoh M, et al. Immunologic tolerance maintained by CD25+CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol Rev. 2001;182:18–32.
Warnecke G, Avsar M, Morancho M, Peters C, Thissen S, Kruse B, et al. Preoperative low-dose irradiation promotes long-term alloantigen acceptance and induces regulatory T cells in a porcine model of pulmonary transplantation. Transplantation. 2006;82:93–101.
Christie JD, Carby M, Bag R, Corris P, Hertz M, Weill D. Report of the ISHLT working group on primary lung graft dysfunction part II: definition. A consensus statement of the international society for heart and lung transplantation. J Heart Lung Transplant. 2005;24:1454–9.
Harringer W, Haverich A. Heart and heart–lung transplantation: standards and improvement. World J Surg. 2002;26:218–25.
Kimura F, Shimizu H, Yoshidome H, Ohtsuka M, Miyazaki M. Immunosuppression following surgical and traumatic injury. Surg Today. 2010;40:793–808.
Satoda N, Shoji T, Wu Y, Fujinaga T, Chen F, Aoyama A, et al. Value of FOXP3 expression in peripheral blood as rejection marker after miniature swine lung transplantation. J Heart Lung Transplant. 2008;27:1293–301.
Moore KW, de Waal Malefyt R, Coffman RL, O’Garre A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765.
Read S, Malmström V, Powrie F, Cytotoxic T. lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J Exp Med. 2000;192:295–302.
Kingsley CI, Karim M, Bushell AR, Wood KJ. CD25+CD4+ regulatory T cells prevent graft rejection: CTLA-4- and IL-10-dependent immunoregulation of alloresponses. J Immunol. 2002;168:1080–6.
Liu W, Putnam AL, Xu-yu Z, Szot GL, Lee MR, Zhu S, Gottlieb PA, et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J Exp Med. 2006;203(7):1701–11.
Seddiki N, Santner-Nanan B, Martinson J, Zaunders J, Sasson S, Landay A, et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J Exp Med. 2006;203(7):1693–700.
Reynaud-Gaubert M, Thomas P, Badier M, Cau P, Giudicelli R, Fuentes P. Early detection of airway involvement in obliterative bronchiolitis after lung transplantation. Functional and bronchoalveolar lavage cell findings. Am J Respir Crit Care Med. 2000;161:1924–9.
Muylem AV, Mélot C, Antonie M, Knoop C, Estenne M. Role of pulmonary function in the detection of allograft dysfunction after heart–lung transplantation. Thorax. 1997;52:643–7.
Becker FS, Martinez FI, Brunsting LA, Deeb GM, Flint A, Lynch JP 3rd. Limitations of spirometry in detecting rejection after single-lung transplantation. Am J Respir Crit Care Med. 1994;150:159–66.
Testi R, Phillips JH, Lanier LL. T cell activation via Leu-23 (CD69). J Immunol. 1989;143:1123–8.
Tough DF, Sun S, Sprent J. T cell stimulation in vivo by lipopolysaccharide (LPS). J Exp Med. 1997;185:2089–94.
Bharat A, Fields RC, Steward N, Trulock EP, Patterson GA, Mohanakumar T. CD4+25+ regulatory T cells limit Th1-autoimmunity by inducing IL-10 producing T cells following human lung transplantation. Am J Transplant. 2006;6:1799–808.
Warnecke G, Bushell A, Nadig SN, Wood KJ. Regulation of transplant arteriosclerosis by CD25+CD4+ T cells generated to alloantigen in vivo. Transplantation. 2007;83:1459–65.
Acknowledgment
We thank Dr. Yuko Ohno of the Department of Health Promotion Science, Osaka University Graduate School of Medicine, Division of Health Sciences, for her critical review of the manuscript and advice on statistical issues.
Conflict of interest
Tomoyuki Nakagiri and his co-authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a grant from Osaka University and by the DFG (SFB 738).
Rights and permissions
About this article
Cite this article
Nakagiri, T., Warnecke, G., Avsar, M. et al. Lung function early after lung transplantation is correlated with the frequency of regulatory T cells. Surg Today 42, 250–258 (2012). https://doi.org/10.1007/s00595-011-0087-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-011-0087-3