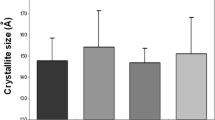
Abstract
This study was performed to characterize the bone metabolism in ten 6-month-old male Goto-Kakizaki (GK) rats, a spontaneous type 2 diabetic model, with ten age- and sex-matched non-diabetic Wistar rats as controls. The femora and the fifth lumbar vertebrae were analyzed by a dual energy X-ray absorptiometry for bone mineral density. Histomorphometrical analyses were performed on the sections from the tibia embedded in methylmethacrylate. Biomechanical characterizations were made by a three-point bending test and a compressive test on the femur and the fifth vertebral body respectively. Compared to the control rats, the bone mineral density was significantly deceased and the histomorphometrical studies showed significantly decreased trabecular bone volume, trabecular thickness and number, osteoid surface and thickness, mineralizing surface, mineral apposition rate and bone formation rate, and also a significant increase in mineralization lag time in the diabetic rats. Strength in both bones and elastic modulus of vertebral body significantly decreased in the diabetic rats as well. In addition, the serum osteocalcin levels were significantly decreased and the serum tartrate-resistant acid phosphatase activity was significantly increased. In conclusion, the 6-month-old GK diabetic rats developed osteopenia with an increased risk of fracture owing to the decreased bone formation, and might be a useful model for unraveling the effects of diabetes on bones independent of obesity frequently seen in the type 2 diabetic patients.


Similar content being viewed by others
References
Albright F, Reifenstein E (1948) Parathyroid glands and metabolic bone disease: selected studies. Williams and Wilkins, Baltimore
Lopez-Ibarra PJ, Pastor MM, Escobar-Jimenez F, Pardo MD, Gonzalez AG, Luna JD, Requena ME, Diosdado MA (2001) Bone mineral density at time of clinical diagnosis of adult-onset type 1 diabetes mellitus. Endocr Pract 7:346–351
Bauer DC, Browner WS, Cauley JA, Orwoll ES, Scott JC, Black DM, Tao JL, Cummings SR (1993) Factors associated with appendicular bone mass in older women. The Study of Osteoporotic Fractures Research Group. Ann Intern Med 118:657–665
Tuominen JT, Impivaara O, Puukka P, Ronnemaa T (1999) Bone mineral density in patients with type 1 and type 2 diabetes. Diabetes Care 22:1196–1200
Hanley DA, Brown JP, Tenenhouse A, Olszynski WP, Ioannidis G, Berger C, Prior JC, Pickard L, Murray TM, Anastassiades T, Kirkland S, Joyce C, Joseph L, Papaioannou A, Jackson SA, Poliquin S, Adachi JD (2003) Associations among disease conditions, bone mineral density, and prevalent vertebral deformities in men and women 50 years of age and older: cross-sectional results from the Canadian Multicentre Osteoporosis Study. J Bone Miner Res 18:784–790
Albala C, Yanez M, Devoto E, Sostin C, Zeballos L, Santos JL (1996) Obesity as a protective factor for postmenopausal osteoporosis. Int J Obes Relat Metab Disord 20:1027–1032
Ahmad T, Ohlsson C, Saaf M, Ostenson CG, Kreicbergs A (2003) Skeletal changes in type-2 diabetic Goto-Kakizaki rats. J Endocrinol 178:111–116
Östenson CG (2001) The Goto-Kakizaki rat. In: Sima A, Shafrir E (eds) Animal models in diabetes: a primer (Frontiers in Animal Diabetes Research). Harwood Academic Publishers, Amsterdam, pp 197–211
Xu J, Rong HQ, Ji H, Wang D, Liu CY (2006) Effects of human parathyroid hormone related protein on osteoporosis of variectomized rats. Chin J Rheumatol 10:154–157
Catala-Gregori P, Garcia V, Hernandez F, Madrid J, Ceron JJ (2006) Response of broilers to feeding low-calcium and phosphorus diets plus phytase under different environmental conditions: body weight and tibiotarsus mineralization. Poult Sci 85:1923–1931
Mattila P, Knuuttila M, Kovanen V, Svanberg M (1999) Improved bone biomechanical properties in rats after oral xylitol administration. Calcif Tissue Int 64:340–344
Turner CH, Burr DB (1993) Basic biomechanical measurements of bone: a tutorial. Bone 14:595–608
Erben RG (1997) Embedding of bone samples in methylmethacrylate: an improved method suitable for bone histomorphometry, histochemistry, and immunohistochemistry. J Histochem Cytochem 45:307–313
Miyakoshi N, Sato K, Abe T, Tsuchida T, Tamura Y, Kudo T (1999) Histomorphometric evaluation of the effects of ovariectomy on bone turnover in rat caudal vertebrae. Calcif Tissue Int 64:318–324
Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2:595–610
Botolin S, McCabe LR (2006) Chronic hyperglycemia modulates osteoblast gene expression through osmotic and non-osmotic pathways. J Cell Biochem 99:411–424
Gopalakrishnan V, Vignesh RC, Arunakaran J, Aruldhas MM, Srinivasan N (2006) Effects of glucose and its modulation by insulin and estradiol on BMSC differentiation into osteoblastic lineages. Biochem Cell Biol 84:93–101
Verhaeghe J, Suiker AM, Visser WJ, Van Herck E, Van Bree R, Bouillon R (1992) The effects of systemic insulin, insulin-like growth factor-I and growth hormone on bone growth and turnover in spontaneously diabetic BB rats. J Endocrinol 134:485–492
Ahmad T, Ugarph-Morawski A, Lewitt MS, Li J, Saaf M, Brismar K (2008) Diabetic osteopathy and the IGF system in the Goto-Kakizaki rat. Growth Horm IGF Res 18:404–411
Vestergaard P (2007) Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes: a meta-analysis. Osteoporos Int 18:427–444
Portha B (2005) Programmed disorders of beta-cell development and function as one cause for type 2 diabetes? The GK rat paradigm. Diabetes Metab Res Rev 21:495–504
Augat P, Reeb H, Claes LE (1996) Prediction of fracture load at different skeletal sites by geometric properties of the cortical shell. J Bone Miner Res 11:1356–1363
Fuse H, Fukumoto S, Sone H, Miyata Y, Saito T, Nakayama K, Takahashi H, Matsumoto T, Ogata E (1997) A new synthetic steroid, osaterone acetate (TZP-4238), increases cortical bone mass and strength by enhancing bone formation in ovariectomized rats. J Bone Miner Res 12:590–597
Kanter M, Altan MF, Donmez S, Ocakci A, Kartal ME (2007) The effects of quercetin on bone minerals, biomechanical behavior, and structure in streptozotocin-induced diabetic rats. Cell Biochem Funct 25:747–752
Verhaeghe J, Suiker AM, Einhorn TA, Geusens P, Visser WJ, Van Herck E, Van Bree R, Magitsky S, Bouillon R (1994) Brittle bones in spontaneously diabetic female rats cannot be predicted by bone mineral measurements: studies in diabetic and ovariectomized rats. J Bone Miner Res 9:1657–1667
Ortoft G, Oxlund H (1988) Reduced strength of rat cortical bone after glucocorticoid treatment. Calcif Tissue Int 43:376–382
(2000 March 27–29) Osteoporosis prevention, diagnosis, and therapy. NIH Consens Statement 17:1–36
Ostenson CG, Fiere V, Ahmed M, Lindstrom P, Brismar K, Brismar T, Kreicbergs A (1997) Decreased cortical bone thickness in spontaneously non-insulin-dependent diabetic GK rats. J Diabetes Complications 11:319–322
Rakic V, Davis WA, Chubb SA, Islam FM, Prince RL, Davis TM (2006) Bone mineral density and its determinants in diabetes: the fremantle diabetes study. Diabetologia 49:863–871
Perez-Castrillon JL, De Luis D, Martin-Escudero JC, Asensio T, del Amo R, Izaola O (2004) Non-insulin-dependent diabetes, bone mineral density, and cardiovascular risk factors. J Diabetes Complicat 18:317–321
Bouillon R (1991) Diabetic bone disease. Calcif Tissue Int 49:155–160
Lee AJ, Hodges S, Eastell R (2000) Measurement of osteocalcin. Ann Clin Biochem 37:432–446
Amir G, Rosenmann E, Sherman Y, Greenfeld Z, Ne’eman Z, Cohen AM (2002) Osteoporosis in the Cohen diabetic rat: correlation between histomorphometric changes in bone and microangiopathy. Lab Invest 82:1399–1405
Liu R, Bal HS, Desta T, Krothapalli N, Alyassi M, Luan Q, Graves DT (2006) Diabetes enhances periodontal bone loss through enhanced resorption and diminished bone formation. J Dent Res 85:510–514
Burkemper KM, Garris DR (2006) Influences of obese (ob/ob) and diabetes (db/db) genotype mutations on lumber vertebral radiological and morphometric indices: skeletal deformation associated with dysregulated systemic glucometabolism. BMC Musculoskelet Disord 7:10
Oz SG, Guven GS, Kilicarslan A, Calik N, Beyazit Y, Sozen T (2006) Evaluation of bone metabolism and bone mass in patients with type-2 diabetes mellitus. J Natl Med Assoc 98:1598–1604
Cutrim DM, Pereira FA, de Paula FJ, Foss MC (2007) Lack of relationship between glycemic control and bone mineral density in type 2 diabetes mellitus. Braz J Med Biol Res 40:221–227
Williams JP, Blair HC, McDonald JM, McKenna MA, Jordan SE, Williford J, Hardy RW (1997) Regulation of osteoclastic bone resorption by glucose. Biochem Biophys Res Commun 235:646–651
Morohoshi M, Fujisawa K, Uchimura I, Numano F (1996) Glucose-dependent interleukin 6 and tumor necrosis factor production by human peripheral blood monocytes in vitro. Diabetes 45:954–959
Miyata T, Notoya K, Yoshida K, Horie K, Maeda K, Kurokawa K, Taketomi S (1997) Advanced glycation end products enhance osteoclast-induced bone resorption in cultured mouse unfractionated bone cells and in rats implanted subcutaneously with devitalized bone particles. J Am Soc Nephrol 8:260–270
Acknowledgments
The authors gratefully acknowledge the financial support from the Shandong Provincial Department of Science and Technology, Jinan, China. The authors would like to thank Prof. Man-Chiu Poon, from University of Calgary, Canada, for his critical review of this manuscript and for his help with the language.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Liu, Y., Wang, D. et al. Bone biomechanical and histomorphometrical investment in type 2 diabetic Goto-Kakizaki rats. Acta Diabetol 46, 119–126 (2009). https://doi.org/10.1007/s00592-008-0068-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-008-0068-1