Abstract
Purpose
The effectiveness of laminectomy with fusion and laminectomy alone in degenerative cervical myelopathy was compared.
Methods
Individuals treated with laminectomy with fusion or laminectomy alone at or below the second cervical vertebra were identified in the Swedish spine registry. 66 individuals treated with laminectomy and instrumented fusion were age matched to 132 individuals treated with laminectomy alone. The European Myelopathy Scale (EMS), the Neck Disability Index (NDI), the Numeric Rating Scale (NRS) for neck pain and the EQ-5D index were available at baseline, and at 1 and 2 year follow-ups. Statistical analyses were performed with Mann–Whitney U tests and paired T tests. Effect sizes were described with Cohen’s D.
Results
Data at baseline did not differ significantly between the groups with the exception of a longer laminectomy in the fusion group (4.2 vs 3.4 vertebras; p < 0.001). Both groups improved EMS, NDI, NRS and EQ-5D from baseline to 1 year (p ≤ 0.011), with no additional improvement between 1 and 2 years (all p ≥ 0.09). Effect sizes for change from baseline were small to medium in both groups. At 2 years the laminectomy with fusion group and the laminectomy alone group had a median (25th;75th percentile) EMS of 13 (11;13) and 13 (11;15) (p = 0.77), NDI of 39 (24;54) and 27 (10;41) (p = 0.045), NRS of 4 (1;2) and 2 (0;5) (p = 0.048), and EQ-5D index of 0.67 (0.25;0.73) and 0.66 (0.17;0.76) (p = 0.96).
Conclusion
The results of this study suggest similar effectiveness of laminectomy with instrumented fusion and laminectomy alone in degenerative cervical myelopathy.
Level of evidence
III.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Degenerative cervical myelopathy, or cervical spondylotic myelopathy, is a common cause of spinal cord compromise and increase with age [1]. Clinical signs of myelopathy, together with magnetic resonance imaging, showing a compressed spinal cord with possible signal intensity changes, is used for diagnostics [1].
Failed non-surgical management or progressive myelopathy symptoms are reasons for surgery [2]. Choice of type of surgery is based on the extent and location of the pathology. Anterior cervical discectomy or corpectomy is often used for shorter and anteriorly located pathologies, while posterior cervical laminectomy is often chosen for longer or posteriorly located pathologies, but with no certain functional outcome advantage for any of the approaches [1, 3].
To date, there is no high level of evidence to support any benefit with the addition of posterior instrumented fusion as compared to laminectomy alone. The decision is made on a case-by-case basis based on patient characteristics such as age, severity of myelopathy, extent of the pathology, loss of cervical lordosis and spondylolisthesis [1, 2, 4].
Posterior instrumented fusion has theoretical advantages and may prevent the risk for postoperative kyphosis, but incurs a longer surgical time, a higher risk for infection, and costs for implants [4,5,6,7]. However, the correlation between changes in the sagittal alignment, neurologic deterioration and clinical outcome has not been established [4, 8, 9].
A recent observational study suggested that instrumented fusion was associated with a better result than laminectomy alone, but was hampered by a small cohort size for the group without fusion (n = 22) compared to the group with fusion (n = 186) making the study prone to chance findings and case ascertainment bias [5].
The purpose of this study was to compare the effectiveness of laminectomy alone and laminectomy and instrumented fusion on several important aspects of outcome such as function, health related quality of life and pain. We hypothesized that the two surgical methods had an equal effectiveness and similar outcome.
Materials and methods
The National Swedish Registry for Spine Surgery (Swespine) was used for the study. The study design was set after data collection, but before data retrieval from the registry. We considered the STROBE criteria when preparing this report [10].
Swespine was initiated for registration of cervical spine surgeries in 2006. The number of clinics reporting cervical spine surgeries is about 15. The patient response rate is about 75% at the 1 year follow-up [11]. Diagnosis, type of surgery, implant use, operated vertebras, presence of myelopathy and/or radiculopathy, length of inpatient stay and complications are registered by the treating clinic at the time of surgery. Questionnaire data answered by the patient preoperative and at 1, 2, and 5 years are reported here.
Study population and inclusion criteria
We included individuals in the Swespine registry according to the following criteria:
-
Degenerative cervical myelopathy,
-
Treatment with laminectomy with instrumented fusion or laminectomy alone, without foraminotomies,
-
Surgery at or below the second cervical vertebra,
-
No previous cervical spine surgeries,
-
Planned (elective) posterior-only surgery,
-
No signs of instability (as assessed by the surgeon),
-
Surgery until Dec, 2017,
-
Aged 40 years or older,
-
Outcome data at baseline and at the 2 year follow-up.
To achieve a similar age distribution, we matched the patients for age. Starting with the oldest patient in the laminectomy with fusion group, the two patients that were closest in age in the laminectomy alone group were selected. The process was repeated until all patients in the laminectomy with fusion group had been matched with two patients treated with laminectomy alone.
Data available in Swespine
The following data were collected at baseline: anthropometrics (body height, body weight), duration of any neck pain (no pain/ < 3 months/3–12 months/1–2 years/ > 2 years), duration of any arm pain (no pain/ < 3 months/3–12 months/1–2 years/ > 2 years), current smoking status (yes/no), use of analgesics (yes regularly, yes sometimes, no) and walking distance (< 100 m/100–500 m/0.5–1 km/ > 1 km).
The European Myelopathy Scale (EMS) was developed to assess cervical myelopathy and based upon the Japanese Orthopaedic Association (JOA) score [12,13,14]. The EMS has five domains with the following maximum points: gait impairment (five points), bowel and bladder dysfunction (three points), hand function impairment (four points, impairment with proprioception and coordination (three points), and paresthesia or dysesthesia (three points). The total EMS score runs from minimum 5 (severe disability) to 18 (normal). Grading of myelopathy according to the EMS has been suggested as follows; severe (5–8 points), moderate (9–12 points), mild (13–16 points) and normal (17–18 points) [12, 13]. EMS data was collected at baseline and at the follow-ups.
The Neck Disability Index (NDI) is a neck specific assessment tool concerning how neck problems are affecting the ability to manage everyday life and range between 0 (no disability) and 100 (maximum disability) [15]. NDI data was collected at baseline and at the follow-ups.
The Numeric Rating Scale (NRS) for neck pain runs from 0 (no pain) to 10 (worst imaginable pain). Previously the visual analogue scale was used in the Swespine Registry. Existing VAS data in the Swespine registry has been converted to NRS by dividing the VAS score by 10 with a stochastic approximation of decimals to the closest integer [16]. NRS data was collected at baseline and at the follow-ups.
The EQ-5D 3 level is a generic health related quality of life questionnaire. Individuals report their health in five dimensions: mobility, self-care, everyday activities, pain/discomfort, and anxiety/depression. The EQ-5D index used here is based on the British time trade-off data and ranges between − 0.59 (worst) and 1.00 (best) [17]. EQ-5D data was collected at baseline and at the follow-ups.
At the 1 and 2 year follow-up data on satisfaction with the surgical result (satisfied, uncertain, and dissatisfied), improvement in fine motor skills (completely recovered/much improved/somewhat improved/unchanged/worse), use of analgesics and walking distance was collected.
Complications
Surgeons and patients reported the occurrence of thrombosis/pulmonary embolism, urinary tract infection, bleeding/hematoma, wound infection, and iatrogenic dural tear within 3 months from surgery. Data on any re-operations was searched for up to 5 years after surgery in the Swespine registry.
Statistical analysis
Data are presented as mean with standard deviation (SD), median (25th; 75th percentile) or number (%). Data distribution was assessed with quantile–quantile plots. For group comparisons of normally distributed variables the Welch–Satterthwaite T test was used due to expected differences in sample size and variances. For non-normally distributed variables the Mann–Whitney U test was used. For group comparisons of categorical variables, the Pearson Chi-square test or the Fisher test (if observed or expected numbers were 5 or less) were used. For within group comparisons the paired T test or the McNemar test was used. Missing data was managed with pairwise deletion in the analyses. All tests were two sided.
Effect sizes were calculated with Cohen’s D for within group changes between baseline and the 2 year follow up for EMS, NDI, NRS and EQ-5D. Effect size was interpreted as small if D = 0.2, medium if D = 0.5 and large if D = 0.8 [18].
We performed additional comparisons to check for data robustness. Group comparisons were made based on the median number of vertebras included in the laminectomy (up to three, or four or more vertebras), and whether treatment took place before or after the median date of surgery, which was end of June 2014. We addressed a possible selection bias by comparing spinal units who had performed only one type of procedure and compared baseline and 2 year follow up data for EMS, NDI, NRS and EQ-5D.
All statistical analyses were performed in SPSS statistics software, version 27. p values less than 0.05 were considered significant.
Sample size
With a 1:2 distribution of cases, identification of a 10% difference in a patient reported outcome with a standard deviation twice this difference with 80% power and the probability of a type 1 error set to 0.05, require 48 and 96 patients in the different groups.
Ethical considerations
All patients undergoing spine surgery are informed of the quality registry Swespine, and that data may be used for research after ethical approval. The Regional Ethical Review Board in Stockholm has authorized the study and the use of the data collection, number 2012/206/31-1 and 2018/2746-32.
Results
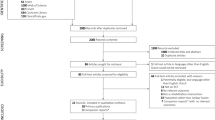
Figure 1 shows the flow chart of the study. 206 individuals fulfilled the inclusion criteria. After age matching 198 individuals remained; 66 individuals that had undergone laminectomy with fusion and 132 individuals that had undergone laminectomy alone.
Baseline data are shown in Table 1. These did not differ significantly between the groups, except that more vertebras were treated with laminectomy in the fusion group.
In the cohort of 198 patients, myelopathy grading according to the European Myelopathy Scale (EMS) was as follows: severe (5–8 points) in 9 individuals, moderate (9–12 points) in 69 individuals, mild (13–16 points) in 102 individuals and normal (17–18 points) in 18 individuals, with no difference in distribution between the treatment groups (p = 0.23).
At baseline, EMS, NDI, NRS neck pain and EQ-5D did not differ significantly between the groups (Table 2). A significant improvement in EMS, NDI, NRS and EQ-5D index was seen to the 1 year follow-up, but not between the 1 year and the 2 year follow-up (Fig. 2). In the group that underwent laminectomy with fusion the effect size for change was -0.27 for EMS, 0.40 for NDI, 0.27 for NRS, and − 0.57 for the EQ-5D index. In the group that underwent laminectomy alone the effect size for change was − 0.26 for EMS, 0.43 for NDI, 0.41 for NRS, and − 0.27 for the EQ-5D index. No significant changes occurred in the laminectomy with fusion and the laminectomy alone groups for self-assessed walking distance between baseline and the 1 year follow-up (p = 0.43 and p = 0.16), or between the 1 and 2 year follow-up (p = 0.97 and p = 0.59). No significant changes occurred in any of the groups between 1 and 2 years regarding improvement in fine motor skills (p = 0.14 and p = 0.13) or satisfaction with the surgical result (p = 0.67 and p = 0.35).
Mean preoperative, 1 and 2 year follow-up data for the different patient reported outcomes. The European Myelopathy Scale (EMS), the Neck Disability Index (NDI), the Numeric rating scale for neck pain (NRS), and the EQ-5D index all improved between baseline and the 1 year follow-up. No significant changes occurred between the 1 and 2 year follow-up. p values for paired t-tests for the difference between the time points are shown in the graph. The upper value is for the group that underwent laminectomy with fusion, while the lower value is for the group that underwent laminectomy alone without fusion. Solid blue line: individuals treated with laminectomy with fusion. Interrupted black line: individuals treated with laminectomy alone without fusion. Yr year, Fu follow-up
At 2 years, EMS and EQ-5D did not differ significantly between the groups, while NDI and NRS neck pain was significantly lower in the laminectomy alone group (Table 2). At 2 years, treatment satisfaction, walking distance and fine motor skills did not differ significantly between the groups, but the use of analgesics was significantly more common in the group that underwent laminectomy with fusion (Table 3).
At 5 years, EMS, NDI, NRS neck pain, EQ-5D did not differ significantly between the groups (Supplementary Table 1).
Complications
Wound infection was more common in the group treated with laminectomy and fusion (Table 4). Other complications, including reoperations did not differ between the groups (Table 4).
Additional analyses
When comparing individuals treated with laminectomy in up to three versus four or more vertebras, baseline variables, preoperative and 2 year follow-up EMS, NDI, NRS and EQ-5D index did not differ significantly (all p ≥ 0.34) (Supplementary Tables 2 and 3).
During the first time period, through June 2014, a higher proportion of laminectomies with fusion was performed (47 out of 99 surgeries; 48%) compared to the second time period, from July 2014, (19 out of 99 surgeries; 19%), p < 0.001. The baseline variables did not differ significantly between the time periods, with the exception of arm pain duration (Supplementary Table 4). EMS, NDI, NRS, and EQ-5D index did not differ significantly at baseline and the 2 year follow-up (Supplementary Table 5).
The baseline and 2 year EMS, NDI, NRS and EQ-5D for patients treated at spinal units who had performed only one type of procedure, laminectomy with fusion (n = 36) or laminectomy alone (n = 41), were without statistically significant differences (Supplementary Table 6).
Discussion
In summary, the results of this study suggest similar effectiveness of laminectomy with instrumented fusion and laminectomy alone in degenerative cervical myelopathy.
Guidelines suggest surgery for moderate and severe myelopathy and that it should be offered in mild myelopathy if non-surgical treatment fails, or symptoms progress [2]. Both disease specific and generic quality of life instruments showed an improvement after surgery, indicating that several aspects are affected by degenerative cervical myelopathy and that these also can be improved by surgery.
An improvement in the EMS after surgery has been reported also in other studies [14, 19]. The EMS was implemented in Swespine since one of the advantages was the capability for patient self-assessment. The EMS has not been used widely, and its response to change is probably smaller than the today widely used modified Japanese Orthopaedic Association score (mJOA) [14, 19]. Even though the composite EMS score include gait and hand function, our Swespine questions could not detect an improvement in walking distance and fine motor skills after surgery in this cohort.
The Neck Disability Index (NDI) is a composite variable directed towards neck pain, function in daily life and mobility. The 2 year result for both treatment groups was at a level which has been suggested to correspond to severe disability [15]. Neck pain was also measured with the Numeric Rating Scale, and a high level of pain was seen before surgery. Neck pain showed the same pattern of improvement as the disease specific and generic quality of life instruments and decreased after surgery, but remained high. Accordingly, analgesics were taken by more than half of the individuals also after surgery. Intake of analgesics was more common in the fusion group who also had slightly higher neck disability and pain.
The effect sizes for the changes between baseline and the 2 year follow-up were small to medium. The modest changes are also reflected in the fact that the suggested minimal clinical important differences were not reached at a group level. MCID has been suggested to be around 15 for NDI, 2.5 for NRS [20, 21], and 0.24 for EQ-5D [21]. MCID for EMS has not been established.
Improvement between 1 and 2 years after surgery was not seen in any of the tested variables. Therefore, either a 1 or 2 year follow-up seem sufficient after DCM surgery, as has been reported for degenerative lumbar disorders [22, 23]. We are unable to draw any firm conclusions about the importance of longer follow-ups due to the amount of missing data at 5 years in this study.
The advantages of the study design include the use of a well described registry with a high coverage and follow-up rate, a fairly large cohort size, treatment cohorts balanced for baseline variables and multiple questions covering various aspects of importance for the individual with degenerative cervical myelopathy.
At least two major limitations exist in the current study, the observational study design, and the lack of imaging data.
Due to the observational nature of the study, the data should be interpreted with some caution. High age could be one reason to defer from fusion. We chose a simple age matching design and avoided another currently popular study design, propensity score matching, which may inflate group imbalance [24]. The groups seemed well balanced on multiple variables. Even though a longer laminectomy was associated with the choice of instrumented fusion, it was not associated with any of the baseline variables or outcome.
Observational trials may give relevant results and comparable to randomized controlled trials within Scandinavian spine registries [25,26,27]. Registry data also represents the real life situation and have larger external validity than randomized controlled trials.
Data from magnetic resonance imaging and radiographs were lacking. We can therefore not determine if the choice of fusion was based on a kyphotic situation, a high degree of spinal cord compression, or any other condition seen on imaging. On the other hand, radiological findings have no clear association with patient reported outcome [8, 28], and the risk of reoperation was low after laminectomy alone, in concordance with other reports [6,7,8,9].
A change in type of surgery over time, and that use of only one type of procedure in a spinal unit were unrelated to significant outcome differences strengthens our results. It is evident that the choice of surgical method is based on treatment tradition: 33% of the individuals in this Swedish cohort were treated by instrumented fusion. This is far from the figure of 89% fused in a recently published similar sized cohort from 26 centers in North America and globally [5]. In this cohort, large regional differences in technique were found. Patients from European centers (n = 35) were only treated by instrumented fusion in 27% of the cases, a figure close to the data in our cohort.
Conclusion
The need for instrumented fusion in most cases with cervical degenerative myelopathy without signs of instability may not be needed. A randomized controlled study is needed to achieve a higher level of evidence. We welcome such efforts.
Code availability
No specific software codes used.
References
Badhiwala JH, Ahuja CS, Akbar MA, Witiw CD, Nassiri F, Furlan JC, Curt A, Wilson JR, Fehlings MG (2020) Degenerative cervical myelopathy—update and future directions. Nat Rev Neurol 16:108–124. https://doi.org/10.1038/s41582-019-0303-0
Fehlings MG, Tetreault LA, Riew KD, Middleton JW, Aarabi B, Arnold PM, Brodke DS, Burns AS, Carette S, Chen R, Chiba K, Dettori JR, Furlan JC, Harrop JS, Holly LT, Kalsi-Ryan S, Kotter M, Kwon BK, Martin AR, Milligan J, Nakashima H, Nagoshi N, Rhee J, Singh A, Skelly AC, Sodhi S, Wilson JR, Yee A, Wang JC (2017) A clinical practice guideline for the management of patients with degenerative cervical myelopathy: recommendations for patients with mild, moderate, and severe disease and nonmyelopathic patients with evidence of cord compression. Glob Spine J 7:70S-83S. https://doi.org/10.1177/2192568217701914
Ghogawala Z, Terrin N, Dunbar MR, Breeze JL, Freund KM, Kanter AS, Mummaneni PV, Bisson EF, Barker FG 2nd, Schwartz JS, Harrop JS, Magge SN, Heary RF, Fehlings MG, Albert TJ, Arnold PM, Riew KD, Steinmetz MP, Wang MC, Whitmore RG, Heller JG, Benzel EC (2021) Effect of ventral vs dorsal spinal surgery on patient-reported physical functioning in patients with cervical spondylotic myelopathy: a randomized clinical trial. JAMA 325:942–951. https://doi.org/10.1001/jama.2021.1233
Kim BS, Dhillon RS (2019) Cervical laminectomy with or without lateral mass instrumentation: a comparison of outcomes. Clin Spine Surg 32:226–232. https://doi.org/10.1097/BSD.0000000000000852
Kotter MRN, Tetreault L, Badhiwala JH, Wilson JR, Arnold PM, Bartels R, Barbagallo G, Kopiar B, Fehlings MG (2020) Surgical outcomes following laminectomy with fusion versus laminectomy alone in patients with degenerative cervical myelopathy. Spine 45:1696–1703. https://doi.org/10.1097/brs.0000000000003677
Ryken TC, Heary RF, Matz PG, Anderson PA, Groff MW, Holly LT, Kaiser MG, Mummaneni PV, Choudhri TF, Vresilovic EJ, Resnick DK (2009) Cervical laminectomy for the treatment of cervical degenerative myelopathy. J Neurosurg Spine 11:142–149. https://doi.org/10.3171/2009.1.Spine08725
Mikawa Y, Shikata J, Yamamuro T (1987) Spinal deformity and instability after multilevel cervical laminectomy. Spine 12:6–11. https://doi.org/10.1097/00007632-198701000-00002
Kaptain GJ, Simmons NE, Replogle RE, Pobereskin L (2000) Incidence and outcome of kyphotic deformity following laminectomy for cervical spondylotic myelopathy. J Neurosurg 93:199–204. https://doi.org/10.3171/spi.2000.93.2.0199
Lofgren H, Osman A, Blomqvist A, Vavruch L (2020) Sagittal alignment after laminectomy without fusion as treatment for cervical spondylotic myelopathy: follow-up of minimum 4 years postoperatively. Glob Spine J 10:425–432. https://doi.org/10.1177/2192568219858302
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, Initiative S (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ 335:806–808. https://doi.org/10.1136/bmj.39335.541782.AD
Fritzell P, Hägg O, Gerdhem P, Abbott A, Songsong A, Parai C, Thoreson O, Strömqvist B, Mellgren L, CB (2019) Swespine årsrapport 2019. Uppföljning av ryggkirurgi utförd i Sverige 2018. http://www.swespine.se/. Accessed 28 Feb 2021. (In Swedish)
Dvorak J, Sutter M, Herdmann J (2003) Cervical myelopathy: clinical and neurophysiological evaluation. Eur Spine J 12(Suppl 2):S181-187. https://doi.org/10.1007/s00586-003-0631-y
Herdmann J, Linzbach M, Krzan M, Dvorak J, Bock W (1994) The European Myelopathy Score. In: Bauer B, Brock M, Klinger M (eds) Advances in neurosurgery. Springer Berlin Heidelberg, Berlin Heidelberg, pp 266–268
Singh A, Crockard HA (2001) Comparison of seven different scales used to quantify severity of cervical spondylotic myelopathy and post-operative improvement. J Outcome Meas 5:798–818
Vernon H, Mior S (1991) The Neck Disability Index: a study of reliability and validity. J Manip Physiol Ther 14:409–415
Hjermstad MJ, Fayers PM, Haugen DF, Caraceni A, Hanks GW, Loge JH, Fainsinger R, Aass N, Kaasa S, European Palliative Care Research C (2011) Studies comparing Numerical Rating Scales, Verbal Rating Scales, and Visual Analogue Scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manag 41:1073–1093. https://doi.org/10.1016/j.jpainsymman.2010.08.016
Burstrom K, Johannesson M, Diderichsen F (2001) Swedish population health-related quality of life results using the EQ-5D. Qual Life Res 10:621–635
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum Associates, New York
Vitzthum HE, Dalitz K (2007) Analysis of five specific scores for cervical spondylogenic myelopathy. Eur Spine J 16:2096–2103. https://doi.org/10.1007/s00586-007-0512-x
Carreon LY, Glassman SD, Campbell MJ, Anderson PA (2010) Neck Disability Index, short form-36 physical component summary, and pain scales for neck and arm pain: the minimum clinically important difference and substantial clinical benefit after cervical spine fusion. Spine J 10:469–474. https://doi.org/10.1016/j.spinee.2010.02.007
Parker SL, Godil SS, Shau DN, Mendenhall SK, McGirt MJ (2013) Assessment of the minimum clinically important difference in pain, disability, and quality of life after anterior cervical discectomy and fusion: clinical article. J Neurosurg Spine 18:154–160. https://doi.org/10.3171/2012.10.SPINE12312
Elkan P, Lagerback T, Moller H, Gerdhem P (2018) Response rate does not affect patient-reported outcome after lumbar discectomy. Eur Spine J 27:1538–1546. https://doi.org/10.1007/s00586-018-5541-0
Endler P, Ekman P, Moller H, Gerdhem P (2017) Outcomes of posterolateral fusion with and without instrumentation and of interbody fusion for isthmic spondylolisthesis: a prospective study. J Bone Jt Surg Am 99:743–752. https://doi.org/10.2106/JBJS.16.00679
King G, Nielsen R (2019) Why propensity scores should not be used for matching. Polit Anal. https://doi.org/10.1017/pan.2019.11
Lonne G, Fritzell P, Hagg O, Nordvall D, Gerdhem P, Lagerback T, Andersen M, Eiskjaer S, Gehrchen M, Jacobs W, van Hooff ML, Solberg TK (2018) Lumbar spinal stenosis: comparison of surgical practice variation and clinical outcome in three national spine registries. Spine J. https://doi.org/10.1016/j.spinee.2018.05.028
Forsth P, Olafsson G, Carlsson T, Frost A, Borgstrom F, Fritzell P, Ohagen P, Michaelsson K, Sanden B (2016) A randomized, controlled trial of fusion surgery for lumbar spinal stenosis. N Engl J Med 374:1413–1423. https://doi.org/10.1056/NEJMoa1513721
Forsth P, Michaelsson K, Sanden B (2013) Does fusion improve the outcome after decompressive surgery for lumbar spinal stenosis? A 2-year follow-up study involving 5390 patients. Bone Jt J 95B:960–965. https://doi.org/10.1302/0301-620X.95B7.30776
Ninomiya K, Yamane J, Aoyama R, Suzuki S, Shiono Y, Takahashi Y, Fujita N, Okada E, Tsuji O, Yagi M, Watanabe K, Iga T, Nakamura M, Matsumoto M, Ishii K, Nagoshi N (2020) Clinical effects of anterior cervical spondylolisthesis on cervical spondylotic myelopathy after posterior decompression surgery: a retrospective multicenter study of 732 cases. Glob Spine J. https://doi.org/10.1177/2192568220966330
Acknowledgements
We acknowledge all the patients and surgeons contributing data to the Swedish Spine Registry, and Carina Blom, registry secretary for assistance with database extraction.
Funding
Open access funding provided by Karolinska Institute. Paul Gerdhem was supported by Region Stockholm in a clinical research appointment, and by CIMED, Karolinska Institutet. The funding sources had no role in the study design, analyses or interpretation of data, in the manuscript writing, or in the decision to submit the paper for publication. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
Author information
Authors and Affiliations
Contributions
DF: study design, data management, analysis, first draft of manuscript, manuscript comments. AC: study design, analysis, manuscript comments. PG: study design, data management, analysis, draft of manuscript, finalizing manuscript, corresponding author.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Availability of data
The datasets generated during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Revesz, D.F., Charalampidis, A. & Gerdhem, P. Effectiveness of laminectomy with fusion and laminectomy alone in degenerative cervical myelopathy. Eur Spine J 31, 1300–1308 (2022). https://doi.org/10.1007/s00586-022-07159-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-022-07159-1