Abstract
Purpose
Previous reviews of randomised controlled trials (RCTs) for low-back pain (LBP) have failed to identify any positive trend in study quality with more recent years of publication. This study aimed to identify and describe trends over time in the study design characteristics and risk of bias in chronic LBP trials performed over the past 30 years.
Methods
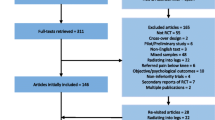
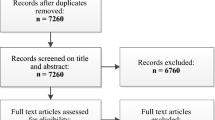
One fifty-seven randomised trials of interventions for chronic LBP were extracted from recently published systematic reviews. The reviews included RCTs on physical and rehabilitation interventions, injection therapy and denervation procedures, complementary and alternative therapies and pharmacological interventions for chronic LBP. Study level data were extracted and analysed for trends associated with year of publication.
Results
Overall, the mean sample size in the RCTs was 141 (median 70; range 17–3093). There was a slight increase in the median number of risk of bias criteria fulfilled from trials published prior to 1995 to those published after 1996. The analysis showed that in more recent years RCTs of medical interventions were more likely to be successfully blinded than RCTs of non-medical interventions.
Conclusions
The continuing uncertainty regarding the efficacy of many interventions for chronic LBP again stresses the need for large RCTs with low risk of bias. Further research is needed into specific risks of bias within the RCTs for chronic LBP and the effect they have on the plausibility of the results.




Similar content being viewed by others
References
Dagenais S, Caro J, Haldeman S (2008) A systematic review of low back pain cost of illness studies in the United States and internationally. Spine J 8(1):8–20
Freburger JK, Holmes GM, Agans RP, Jackman AM, Darter JD, Wallace AS, Castel LD, Kalsbeek WD, Carey TS (2009) The rising prevalence of chronic low back pain. Arch Intern Med 169(3):251–258
Koes BW, van Tulder M, Lin CW, Macedo LG, McAuley J, Maher C (2010) An updated overview of clinical guidelines for the management of non-specific low back pain in primary care. Eur Spine J [epub ahead of print]
Deyo RA, Mirza SK, Turner JA, Martin BI (2009) Overtreating chronic back pain: time to back off? J Am Board Fam Med 22(1):62–68
Koes BW, Malmivaara A, van Tulder MW (2005) Trend in methodological quality of randomised clinical trials in low back pain. Best Pract Res Clin Rheumatol 19(4):529–539
Moher D, Schulz KF, Altman DG (2001) The CONSORT statement: revised recommendations for improving the quality of reports of parallel-group randomised trials. Lancet 357(9263):1191–1194
Schulz KF, Altman DG, Moher D (2010) CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMJ 340:c332
Moher D, Jones A, Lepage L (2001) Use of the CONSORT statement and quality of reports of randomized trials: a comparative before-and-after evaluation. JAMA 285(15):1992–1995
van Tulder M, Furlan AD, Bombardier C, Bouter L (2003) Updated method guidelines for systematic reviews in the Cochrane Collaboration Back Review Group. Spine 28(12):1290–1299
Furlan AD, Pennick V, Bombardier C, van Tulder M (2009) 2009 updated method guidelines for systematic reviews in the Cochrane Back Review Group. Spine 34(18):1929–1941
Koes BW, Bouter LM, van der Heijden GJ (1995) Methodological quality of randomized clinical trials on treatment efficacy in low back pain. Spine 20(2):228–235
van Middelkoop M, Rubinstein SM, Kuijpers T, Verhagen AP, Ostelo R, Koes BW, van Tulder MW (2010) A systematic review on the effectiveness of physical and rehabilitation interventions for chronic non-specific low back pain. Eur Spine J [epub ahead of print]
Rubinstein SM, van Middelkoop M, Kuijpers T, Ostelo R, Verhagen AP, de Boer MR, Koes BW, van Tulder MW (2010) A systematic review on the effectiveness of complementary and alternative medicine for chronic non-specific low-back pain. Eur Spine J 19(8):1213–1228
Kuijpers T, van Middelkoop M, Rubinstein SM, Ostelo R, Verhagen A, Koes BW, van Tulder MW (2010) A systematic review on the effectiveness of pharmacological interventions for chronic non-specific low-back pain. Eur Spine J [epub ahead of print]
Henschke N, Kuijpers T, Rubinstein SM, van Middelkoop M, Ostelo R, Verhagen A, Koes BW, van Tulder MW (2010) Injection therapy and denervation procedures for chronic low-back pain: a systematic review. Eur Spine J 19(9):1425–1449
Haake M, Muller HH, Schade-Brittinger C, Basler HD, Schafer H, Maier C, Endres HG, Trampisch HJ, Molsberger A (2007) German Acupuncture Trials (GERAC) for chronic low back pain: randomized, multicenter, blinded, parallel-group trial with 3 groups. Arch Intern Med 167(17):1892–1898
Witt CM, Jena S, Selim D, Brinkhaus B, Reinhold T, Wruck K, Liecker B, Linde K, Wegscheider K, Willich SN (2006) Pragmatic randomized trial evaluating the clinical and economic effectiveness of acupuncture for chronic low back pain. Am J Epidemiol 164(5):487–496
Chan AW, Altman DG (2005) Epidemiology and reporting of randomised trials published in PubMed journals. Lancet 365(9465):1159–1162
van Tulder MW, Suttorp M, Morton S, Bouter LM, Shekelle P (2009) Empirical evidence of an association between internal validity and effect size in randomized controlled trials of low-back pain. Spine 34(16):1685–1692
Maher CG, Moseley AM, Sherrington C, Elkins MR, Herbert RD (2008) A description of the trials, reviews, and practice guidelines indexed in the PEDro database. Phys Ther 88(9):1068–1077
Acknowledgments
NH is supported by a fellowship from the National Health and Medical Research Council of Australia.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Henschke, N., Kuijpers, T., Rubinstein, S.M. et al. Trends over time in the size and quality of randomised controlled trials of interventions for chronic low-back pain. Eur Spine J 21, 375–381 (2012). https://doi.org/10.1007/s00586-011-2023-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-011-2023-z