Abstract
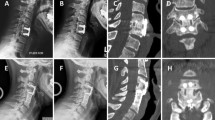
The use of bone morphogenetic protein-2 (rhBMP-2) in spinal fusion has increased dramatically since an FDA approval for its use in anterior lumbar fusion with the LT cage. There are several reports of its use in transforaminal lumbar interbody fusion, posterolateral fusion, and anterior cervical fusion. Reports on adverse effects of rhBMP-2 when used in spinal fusion are scarce in literature. An Institutional Review Board approved retrospective study was conducted in patients undergoing anterior spinal fusion and instrumentation following diskectomy at a single center. Forty-six consecutive patients were included. Twenty-two patients treated with rhBMP-2 and PEEK cages were compared to 24 in whom allograft spacers and demineralized bone matrix was used. Patients filled out Cervical Oswestry Scores, VAS for arm pain, neck pain, and had radiographs preoperatively as well at every follow up visit. Radiographic examination following surgery revealed end plate resorption in all patients in whom rhBMP-2 was used. This was followed by a period of new bone formation commencing at 6 weeks. In contrast, allograft patients showed a progressive blurring of end plate-allograft junction. Dysphagia was a common complication and it was significantly more frequent and more severe in patients in whom rhBMP-2 was used. Post operative swelling anterior to the vertebral body on lateral cervical spine X-ray was significantly larger in the rhBMP-2 group when measured from 1 to 6 weeks after which it was similar. These effects are possibly due to an early inflammatory response to rhBMP-2 and were observed to be dose related. With the parameters we used, there was no significant difference in the clinical outcome of patients in the two groups at 2 years. The cost of implants in patients treated with rhBMP-2 and PEEK spacers was more than three times the cost of allograft spacers and demineralized bone matrix in 1, 2, and 3-level cases. Despite providing consistently good fusion rates, we have abandoned using rhBMP-2 and PEEK cages for anterior cervical fusion, due to the side effects, high cost, and the availability of a suitable alternative.









Similar content being viewed by others
References
A guide from the The American Academy of Orthopedic Surgeons (AAOS) and American Association of Tissue Banks (AATB): What can you tell me about bone and tissue transplantation. Retrieved June 21, 2005 from http://www.aatb.org/aaosinfo.htm
Baskin DS, Ryan P, Sonntag V, Westmark R, Widmayer MA (2003) A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SRTM allograft ring and the ATLANTISTM anterior cervical plate. Spine 28(12):1219–1224
Boakye M, Mummaneni PV, Garrett M, Rodts G, Haid R (2005) Anterior cervical discectomy and fusion involving a polyetheretherketone spacer and bone morphogenetic protein. J Neurosurg Spine 2:521–525
Boden SD, Kang J, Sandhu H, Heller JG (2002) Use of recombinant human bone morphogenetic protein-2 to achieve posterolateral lumbar spine fusion in humans: a prospective, Randomized clinical pilot trial 2002 Volvo Award in clinical studies. Spine 27(23):2662–2673
Burkus JK (2004) Bone morphogenetic proteins in anterior lumbar interbody fusion: old techniques and new technologies. J Neurosurg Spine 3:254–260
Burkus JK, Heim SE, Gornet MF, Zdeblick TA (2004) The effectiveness of rhBMP-2 in replacing autograft: an integrated analysis of three human spine studies. Orthopedics 27(7):723–728
Celeste AJ, Iannazzi JA, Taylor RC, Hewick RM, Rosen V, Wang EA, Wozney JM (1990) Identification of transforming growth factor beta family members present in bone-inductive protein purified from bovine bone. Proc Natl Acad Sci USA 87(24):9843–9847
Hidehiro I, Sohei E, Mikio K, Yutaka T, Tetsuya K, Yohei Y, Kunio T (1999) Experimental spinal fusion with use of recombinant human bone morphogenetic protein 2. Spine 24(14):1402–1405
Johnson EE, Urist MR, Finerman GAM (1988) Bone morphogenetic protein augmentation grafting of resistant femoral nonunions. A preliminary report. Clin Orthop Relat Res 230:257–265
Johnson EE, Urist MR, Finerman GA (1988) Repair of segmental defects of the tibia with cancellous bone grafts augmented with human bone morphogenetic protein. A preliminary report. Clin Orthop Relat Res 236:249–257
Kleeman TJ, Michael Ahn U, Talbot-Kleeman A (2001) Laparoscopic anterior lumbar interbody fusion with rhBMP-2: a prospective study of clinical and radiographic outcomes. Spine 26(24):2751–2756
Mummaneni PV, Pan J, Haid RW, Rodts GE (2004) Contribution of recombinant human bone morphogenetic protein-2 to the rapid creation of interbody fusion when used in transforaminal lumbar interbody fusion. J Neurosurg Spine 1:19–23
Poynton AR, Lane JM (2002) Safety profile for the clinical use of bone morphogenetic proteins in the spine. Spine 27(16S):S40–S48
Sandhu HS (2003). Bone morphogenetic proteins and spinal surgery. Spine 28(15S):S64–S73
Sandhu HS, Khan SN (2003) Recombinant human bone morphogenetic protein-2: use in spinal fusion applications. J Bone Joint Surg 85-A(Suppl 3):89–95
Sandhu HS, Toth JM, Diwan AD, Seim HB, Kanim LEA, Kabo JM, Turner AS (2002) Histologic evaluation of the efficacy of rhBMP-2 compared with autograft bone in sheep spinal anterior interbody fusion. Spine 27(6):567–575
Schimandle JH, Boden SD, Hutton WC (1995) Experimental spinal fusion with recombinant human bone morphogenetic protein-2. Spine 20(12):1326–1337
Shields LB, Raque GH, Glassman SD, Campbell M, Vitaz T, Harpring J, Shields CB (2006) Adverse effects associated with high-dose recombinant human bone morphogenetic protein-2 use in anterior cervical spine fusion. Spine 31(5):542–547
Ullrich PF (1999) Anterior cervical spinal fusion surgery. Retrieved June 21, 2005 from http://www.spine-health.com/topics/surg/overview/cervical/cerv03.html
Urist MR (1965) Bone: formation by autoinduction. Science 150:893–899
Wang EA, Rosen V, D’Alessandro JS, Bauduy M, Cordes P, Harada T, Israel DI, Hewick RM, Kerns KM, LaPan P et al (1990) Recombinant human bone morphogenetic protein induces bone formation. Proc Natl Acad Sci USA 87(6):2220–2224
Wang EA, Rosen V, Cordes P, Hewick RM, Kriz MJ, Luxenberg DP, Sibley BS, Wozney JM (1988) Purification and characterization of other distinct bone-inducing factors. Proc Natl Acad Sci USA 85(24):9484–9488
Zdeblick TA, Ghanayem AJ, Rapoff AJ, Swain C, Bassett T, Cooke ME, Markel M (1998) Cervical interbody fusion cages: an animal model with and without bone morphogenetic protein. Spine 23(7):758–765
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vaidya, R., Carp, J., Sethi, A. et al. Complications of anterior cervical discectomy and fusion using recombinant human bone morphogenetic protein-2. Eur Spine J 16, 1257–1265 (2007). https://doi.org/10.1007/s00586-007-0351-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-007-0351-9