Abstract
Introduction
This study aimed to determine the relative potency of direct ischemic preconditioning (DIPC) and remote ischemic preconditioning (RIPC) for protection against ischemic spinal cord injury in rabbits and to explore the mechanisms involved.
Methods
In experiment 1, we compared the neurological and histopathological outcomes of DIPC, kidney RIPC, and limb RIPC. The DIPC and kidney RIPC groups received two cycles of 5-min occlusion/15-min reperfusion of the abdominal aorta and left renal artery, respectively. The limb RIPC group received two cycles of 10-min occlusion/10-min reperfusion of the femoral arteries bilaterally. Thirty minutes after the conditioning ischemia, spinal cord ischemia was produced by occluding the abdominal aorta for 15 min. In experiments 2 and 3, we investigated whether pretreatment using a free-radical scavenger, dimethylthiourea (DMTU), an adenosine A1 receptor antagonist, 8-cyclopentyl-1,3-dipropylxanthine (DPCPX), or a mitochondrial ATP-sensitive potassium channel antagonist, 5-hydroxydecanoate (5HD), could attenuate the protective effects of DIPC. In experiment 4, comprehensive analysis of phosphorylated proteins in the spinal cord was performed using a Proteome Profiler Array followed by immunoblotting to elucidate the signal pathway activated by DIPC.
Results
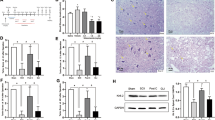
In experiment 1, DIPC improved the neurological and histopathological outcomes, whereas kidney and limb RIPC had no protective effects. In experiments 2 and 3, strong protective effects of DIPC were reconfirmed but were not attenuated by DMTU, DPCPX, or 5HD. In experiment 4, DIPC induced phosphorylation of Akt2.
Conclusions
DIPC, but not kidney or limb RIPC, protected against ischemic spinal cord injury in rabbits. Akt2 might contribute to this protective effect.





Similar content being viewed by others
References
Sivaraman V, Pickard JM, Hausenloy DJ. Remote ischaemic conditioning: cardiac protection from afar. Anaesthesia. 2015;70:732–48.
Meybohm P, Bein B, Brosteanu O, Cremer J, Gruenewald M, Stoppe C, Coburn M, Schaelte G, Boning A, Niemann B, Roesner J, Kletzin F, Strouhal U, Reyher C, Laufenberg-Feldmann R, Ferner M, Brandes IF, Bauer M, Stehr SN, Kortgen A, Wittmann M, Baumgarten G, Meyer-Treschan T, Kienbaum P, Heringlake M, Schon J, Sander M, Treskatsch S, Smul T, Wolwender E, Schilling T, Fuernau G, Hasenclever D, Zacharowski K, Collaborators RIS. A multicenter trial of remote ischemic preconditioning for heart surgery. N Engl J Med. 2015;373:1397–407.
Hausenloy DJ, Candilio L, Evans R, Ariti C, Jenkins DP, Kolvekar S, Knight R, Kunst G, Laing C, Nicholas J, Pepper J, Robertson S, Xenou M, Clayton T, Yellon DM, Investigators ET. Remote ischemic preconditioning and outcomes of cardiac surgery. N Engl J Med. 2015;373:1408–17.
Gurcun U, Discigil B, Boga M, Ozkisacik E, Badak MI, Yenisey C, Kurtoglu T, Meteoglu I. Is remote preconditioning as effective as direct ischemic preconditioning in preventing spinal cord ischemic injury? J Surg Res. 2006;135:385–93.
Dong HL, Zhang Y, Su BX, Zhu ZH, Gu QH, Sang HF, Xiong L. Limb remote ischemic preconditioning protects the spinal cord from ischemia-reperfusion injury: a newly identified nonneuronal but reactive oxygen species-dependent pathway. Anesthesiology. 2010;112:881–91.
Kakimoto M, Kawaguchi M, Sakamoto T, Inoue S, Furuya H, Nakamura M, Konishi N. Evaluation of rapid ischemic preconditioning in a rabbit model of spinal cord ischemia. Anesthesiology. 2003;99:1112–7.
Perez-Pinzon MA, Xu GP, Dietrich WD, Rosenthal M, Sick TJ. Rapid preconditioning protects rats against ischemic neuronal damage after 3 but not 7 days of reperfusion following global cerebral ischemia. J Cereb Blood Flow Metab. 1997;17:175–82.
Utada K, Ishida K, Tohyama S, Urushima Y, Mizukami Y, Yamashita A, Uchida M, Matsumoto M. The combination of insulin-like growth factor 1 and erythropoietin protects against ischemic spinal cord injury in rabbits. J Anesth. 2015;29:741–8.
Matsumoto M, Iida Y, Sakabe T, Sano T, Ishikawa T, Nakakimura K. Mild and moderate hypothermia provide better protection than a burst-suppression dose of thiopental against ischemic spinal cord injury in rabbits. Anesthesiology. 1997;86:1120–7.
Drummond JC, Moore SS. The influence of dextrose administration on neurologic outcome after temporary spinal cord ischemia in the rabbit. Anesthesiology. 1989;70:64–70.
Mizukami Y, Iwamatsu A, Aki T, Kimura M, Nakamura K, Nao T, Okusa T, Matsuzaki M, Yoshida K, Kobayashi S. ERK1/2 regulates intracellular ATP levels through alpha-enolase expression in cardiomyocytes exposed to ischemic hypoxia and reoxygenation. J Biol Chem. 2004;279:50120–31.
Haapanen H, Herajarvi J, Arvola O, Anttila T, Starck T, Kallio M, Anttila V, Tuominen H, Kiviluoma K, Juvonen T. Remote ischemic preconditioning protects the spinal cord against ischemic insult: an experimental study in a porcine model. J Thorac Cardiovasc Surg. 2016;151:777–85.
Herajarvi J, Anttila T, Sarja H, Mustonen C, Haapanen H, Makela T, Yannopoulos F, Starck T, Kallio M, Tuominen H, Puistola U, Karihtala P, Kiviluoma K, Anttila V, Juvonen T. Exploring spinal cord protection by remote ischemic preconditioning: an experimental study. Ann Thorac Surg. 2017;103:804–11.
Schulz R, Cohen MV, Behrends M, Downey JM, Heusch G. Signal transduction of ischemic preconditioning. Cardiovasc Res. 2001;52:181–98.
Lei C, Deng J, Wang B, Cheng D, Yang Q, Dong H, Xiong L. Reactive oxygen species scavenger inhibits STAT3 activation after transient focal cerebral ischemia-reperfusion injury in rats. Anesth Analg. 2011;113:153–9.
Hiraide T, Katsura K, Muramatsu H, Asano G, Katayama Y. Adenosine receptor antagonists cancelled the ischemic tolerance phenomenon in gerbil. Brain Res. 2001;910:94–8.
Nakamura M, Nakakimura K, Matsumoto M, Sakabe T. Rapid tolerance to focal cerebral ischemia in rats is attenuated by adenosine A1 receptor antagonist. J Cereb Blood Flow Metab. 2002;22:161–70.
Mehrjerdi FZ, Aboutaleb N, Pazoki-Toroudi H, Soleimani M, Ajami M, Khaksari M, Safari F, Habibey R. The protective effect of remote renal preconditioning against hippocampal ischemia reperfusion injury: role of KATP channels. J Mol Neurosci. 2015;57:554–60.
Yoshida M, Nakakimura K, Cui YJ, Matsumoto M, Sakabe T. Adenosine A1 receptor antagonist and mitochondrial ATP-sensitive potassium channel blocker attenuate the tolerance to focal cerebral ischemia in rats. J Cereb Blood Flow Metab. 2004;24:771–9.
Diez H, Garrido JJ, Wandosell F. Specific roles of Akt iso forms in apoptosis and axon growth regulation in neurons. PLoS One. 2012;7:e32715.
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research (C) (No. 25462437) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
About this article
Cite this article
Fukui, T., Ishida, K., Mizukami, Y. et al. Comparison of the protective effects of direct ischemic preconditioning and remote ischemic preconditioning in a rabbit model of transient spinal cord ischemia. J Anesth 32, 3–14 (2018). https://doi.org/10.1007/s00540-017-2420-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-017-2420-5