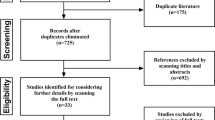
Abstract
Purpose
A depressed mood frequently occurs in perioperative patients, negatively impacting patient recovery. Recent studies suggested that ketamine has a rapid, obvious, and persistent antidepressant effect. The purpose of this study was to investigate the impact of intraoperative application of ketamine on postoperative depressive mood in patients undergoing elective orthopedic surgery.
Methods
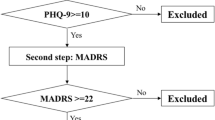
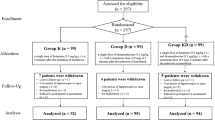
This was a randomized, double-blind, controlled study. A total of 120 patients (ASA grade I–II) undergoing elective orthopedic surgery were divided randomly into a ketamine group (group K) and a control group (group C). In the K group, 0.5 mg/kg (0.05 ml/kg) ketamine was given at induction of anesthesia, followed by 0.25 mg/kg/h (0.025 ml/kg/h) continuous infusion for 30 min. In the C group, 0.05 ml/kg 0.9 % saline was used at induction of anesthesia, followed by 0.025 ml/kg/h continuous infusion of saline for 30 min. PHQ-9 score was recorded preoperatively (1 day before surgery) and postoperatively (on day 1 and day 5 following surgery). Blood at these time points was drawn for serum brain-derived neurotrophic factor (BDNF) level analysis. Intraoperative blood loss, surgery time, postoperative visual analog scale pain scores and perioperative complications were also recorded.
Results
There were no differences in age, sex, surgery time, blood loss, and preoperative PHQ-9 scores between the two groups (P > 0.05). There were no differences in PHQ-9 scores preoperatively and postoperatively for the C group (P > 0.05); however, the PHQ-9 postoperative scores were lower than the preoperative PHQ-9 scores in the K group (P < 0.01). Postoperative PHQ-9 scores of K group were lower than those of C group (P < 0.05). There were no differences in serum BDNF levels in C group pre- to postoperatively (P > 0.05). Compared with the preoperative BDNF levels of K group, postoperative BDNF levels in K group increased significantly (P < 0.01). An inverse correlation between PHQ-9 score and serum BDNF level was shown.
Conclusion
Intraoperative application of ketamine was associated with improved scores for depressed mood and increased serum BDNF levels in patients undergoing elective orthopedic surgery.

Similar content being viewed by others
References
Aanhet Rot M, Collins KA, Murrough JW, Perez AM, Reich DL, Charney DS, Mathew SJ. Safety and efficacy of repeated-dose intravenous ketamine for treatment resistant depression. Biol Psychiatry. 2010;67(2):139–45.
Wang XB, Yang YL, Zhou X, Wu J, Li J, Jiang X, Qu Q, Ou C, Liu L, Zhou S. Propofol pretreatment increases antidepressant-like effects induced by acute administration of ketamine in rats receiving forced swimming test. Psychiatry Res. 2011;185(1–2):248–53.
Garcia LS, Comim CM, Valvassori SS, Réus GZ, Barbosa LM, Andreazza AC, Stertz L, Fries GR, Gavioli EC, Kapczinski F, Quevedo J. Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):140–4.
Murrough JW, Iosifescu DV, Chang LC, Al Jurdi RK, Green CE, Perez AM, Iqbal S, Pillemer S, Foulkes A, Shah A, Charney DS, Mathew SJ. Antidepressant efficacy of ketamine in treatment-resistant major depression: a two-site randomized controlled trial. Am J Psychiatry. 2013;170(10):1134–42.
Howland RH. Ketamine for the treatment of depression. J Psychosoc Nurs Ment Health Serv. 2013;51(1):11–4.
Aan Het Rot M, Zarate CA Jr, Charney DS, Mathew SJ. Ketamine for depression: where do we go from here? Biol Psychiatry. 2012;72(7):537–47.
Schatzberg AF. A word to the wise about ketamine. Am J Psychiatry. 2014;171(3):262–4.
Covvey JR, Crawford AN, Lowe DK. Intravenous ketamine for treatment-resistant major depressive disorder. Ann Pharmacother. 2012;46(1):117–23.
Kudoh A, Takahira Y, Katagai H, Takazawa T. Small-dose ketamine improves the postoperative state of depressed patients. Anesth Analg. 2002;95(1):114–8.
Wang X, Chen Y, Zhou X, Liu F, Zhang T, Zhang C. Effects of propofol and ketamine as the combined anesthesia for electroconvulsive therapy in patients with depressive disorder. J ECT. 2012;28(2):128–32.
Urban-Baeza A, Zárate-Kalfópulos B, Romero-Vargas S, Obil-Chavarría C, Brenes-Rojas L, Reyes-Sánchez A. Influence of depression symptoms on patient expectations and clinical outcomes in the surgical management of spinal stenosis. J Neurosurg Spine. 2015;22(1):75–9.
Warner-Schmidt JL, Duman RS. Hippocampal neurogenesis: opposing effects of stress and antidepressant treatment. Hippocampus. 2006;16(3):239–49.
Grah M, Mihanovic M, Ruljancic N, Restek-Petrovic B, Molnar S, Jelavic S. Brain-derived neurotrophic factor as a suicide factor in mental disorders. Acta Neuropsychiatr. 2014;26(6):356–63.
Cattaneo A, Bocchio-Chiavetto L, Zanardini R, Milanesi E, Placentino A, Gennarelli M. Reduced peripheral brain-derived neurotrophic factor mRNA levels are normalized by antidepressant treatment. Int J Neuropsychopharmacol. 2010;13(1):103–8.
Malpass A, Shaw A, Kessler D, Sharp D. Concordance between PHQ-9 scores and patientsʼ experiences of depression: a mixed methods study. Br J Gen Pract. 2010;60(575):231–8.
Chen S, Chiu H, Xu B, Ma Y, Jin T, Wu M, Conwell Y. Reliability and validity of the PHQ-9 for screening late-life depression in Chinese primary care. Int J Geriatr Psychiatry. 2010;25(11):1127–33.
Matthew M, Jeffrey B. Glioblastoma: preoperative depression may worsen outcomes of patients undergoing surgery. Oncol Times. 2008;30(12):49–51.
Alvin MD, Miller JA, Sundar S, Lockwood M, Lubelski D, Nowacki AS, Scheman J, Mathews M, McGirt MJ, Benzel EC, Mroz TE. The impact of preoperative depression on quality of life outcomes after posterior cervical fusion. Spine J. 2015;15(1):58–64.
Yang T, Liu XY, Wu XM. Incidence of peri-operative anxiety and depression in patients and of risk factors. Zhonghua yi xue za zhi. 2009;89(23):1597–601.
Dunkel A, Kendel F, Lehmkuhl E, Babitsch B, Oertelt-Prigione S, Hetzer R, Regitz-Zagrosek V. Predictors of preoperative depressive risk in patients undergoing coronary artery bypass graft surgery. Clin Res Cardiol. 2009;98(10):643–50.
Murrough JW, Soleimani L, DeWilde KE, Collins KA, Lapidus KA, Iacoviello BM, Lener M, Kautz M, Kim J, Stern JB, Price RB, Perez AM, Brallier JW, Rodriguez GJ, Goodman WK, Iosifescu DV, Charney DS. Ketamine for rapid reduction of suicidal ideation: a randomized controlled trial. Psychol Med. 2015;45(16):3571–80
Browne CA, Lucki I. Antidepressant effects of ketamine: mechanisms underlying fast-acting novel antidepressants. Front Pharmacol. 2013;4(161):1–18.
Zhang C, Li Z, Wu Z, Chen J, Wang Z, Peng D, Hong W, Yuan C, Wang Z, Yu S, Xu Y, Xu L, Xiao Z, Fang Y. A study of N-methyl-d-aspartate receptor gene (GRIN2B) variants as predictors of treatment-resistant major depression. Psychopharmacology. 2014;231(4):685–93.
Zhou W, Wang N, Yang C, Li XM, Zhou ZQ, Yang JJ. Ketamine-induced antidepressant effects are associated with AMPA receptors-mediated upregulation of mTOR and BDNF in rat hippocampus and prefrontal cortex. Eur Psychiatry. 2014;29(7):419–23.
Belujon P, Grace AA. Restoring mood balance in depression: ketamine reverses deficit in dopamine-dependent synaptic plasticity. Biol Psychiatry. 2014;76(12):927–36.
Xu SX, Zhou ZQ, Li XM, Ji MH, Zhang GF, Yang JJ. The activation of adenosine monophosphate-activated protein kinase in rat hippocampus contributes to the rapid antidepressant effect of ketamine. Behav Brain Res. 2013;253(4):305–9.
Stan TL, Alvarsson A, Branzell N, Sousa VC, Svenningsson P. NMDA receptor antagonists ketamine and Ro25-6981 inhibit evoked release of glutamate in vivo in the subiculum. Transl Psychiatry. 2014;4(6):e395.
Autry AE, Montoqqia LM. Brain-derived neurotrophic factor and neuropsychiatry disorders. Pharmacol Rev. 2012;64(2):238–58.
Yoshimura R, Kishi T, Hori H, Atake K, Katsuki A, Nakano-Umene W, Ikenouchi-Sugita A, Iwata N, Nakamura J. Serum proBDNF/BDNF and response to fluvoxamine in drug-naïve first-episode major depressive disorder patients. Ann Gen Psychiatry. 2014;13(19):1744–859.
Yang C, Hu YM, Zhou ZQ, Zhang GF, Yang JJ. Acute administration of ketamine in rats increases hippocampal BDNF and mTOR levels during forced swimming test. Ups J Med Sci. 2013;118(1):3–8.
Hu Y, Yu X, Yang F, Si T, Wang W, Tan Y, Zhou D, Wang H, Chen D. The level of serum brain-derived neurotrophic factor is associated with the therapeutic efficacy of modified electroconvulsive therapy in Chinese patients with depression. J ECT. 2010;26(2):121–5.
Nitta R, Goyagi T. NishikawaT. Combination of oral clonidine and intravenous low-dose ketamine reduces the consumption of postoperative patient-controlled analgesia morphine after spine surgery. Acta Anaesthesiol Taiwan. 2013;51(1):14–7.
Joly V, Richebe P, Guignard B, Fletcher D, Maurette P, Sessler DI, Chauvin M. Remifentanil-induced postoperative hyperalgesia and its prevention with small-dose ketamine. Anesthesiology. 2005;103(1):147–55.
Cagla Ozbakis Akkurt B, Inanoglu K, Kalaci A, Turhanoglu S, Asfuroglu Z, Tumkaya F. Effects of intravenous small dose ketamine and midazolam on postoperative pain following knee arthroscopy. Pain Pract. 2009;9(4):289–95.
Hang LH, Shao DH, Gu YP. The ED50 and ED95 of ketamine for prevention of postoperative hyperalgesia after remifentanil-based anaesthesia in patients undergoing laparoscopic cholecystectomy. Swiss Med Wkly. 2011;141(1):w13195.
Nelson AM, Battersby AS, Baghdoyan HA, Lydic R. Opioid-induced decreases in rat brain adenosine levels are reversed by inhibiting adenosine deaminase. Anesthesiology. 2009;111(6):1327–33.
Acknowledgments
This work was supported by the Projects of the National Natural Science Foundation of China (Grant No. 81271478), International Cooperation, Department of Science and Technology of Sichuan Province (Grant No. 2011HH0031), Department of Education of Sichuan Province (Grant No. 14106) and Innovative Team, Department of Education of Sichuan Province (Grant No. 14TD0018) to X. Wang.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no potential conflicts of interest to disclose.
Additional information
M. Jiang and M.-H. Wang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Jiang, M., Wang, MH., Wang, XB. et al. Effect of intraoperative application of ketamine on postoperative depressed mood in patients undergoing elective orthopedic surgery. J Anesth 30, 232–237 (2016). https://doi.org/10.1007/s00540-015-2096-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-015-2096-7