Abstract
Purpose
In order to determine the mechanism of the protective effect of a urinary trypsin inhibitor (UTI) on renal ischemic reperfusion injury, we measured the tissue oxygen partial pressure (\( p_{O_2 } \)) in both the renal cortex and medulla in rats, using electron paramagnetic resonance (EPR) oximetry.
Methods
We allocated the rats to three groups: normal saline (NS) group, a UTI 50 000 U·kg−1 (LD) group, and a UTI 150 000 U·kg−1 (HD) group, with the normal saline and UTI being administered 30 min before ischemia. Renal ischemia was achieved by inflating the balloon of a vascular occluder that had been placed around the abdominal aorta just above the bifurcation of the renal artery. Cortical and medullary \( p_{O_2 } \) were measured every 10 min during ischemia (30 min) and reperfusion (60 min) by EPR oximetry; also, systemic cardiopulmonary parameters were measured.
Results
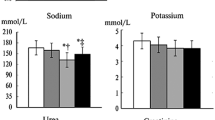
The \( p_{O_2 } \) in the cortex and medulla decreased to less than 2 mmHg during ischemia in all groups. At 60 min after reperfusion, the \( p_{O_2 } \) values in the NS group were not fully restored, whereas those in the LD and HD groups were completely restored to the pre-ischemic values. There were no significant differences between the HD and LD groups. There were no differences between any groups in cardiopulmonary parameters.
Conclusion
Because UTI improved renal oxygenation after reperfusion without changing cardiopulmonary parameters, the pharmacological properties of UTI, such as its renal protection and anti-shock activity, may be explained in part, by this improvement in tissue oxygenation.
Similar content being viewed by others
References
Cornard MF, Craeford RS, Davison JK, Cambria RPC. Thoracoabdominal aneurysm repair: a 20-year perspective. Ann Thorac Surg. 2007;83:S856–S861.
Bonventre JV, Weinberg JM. Recent advances in the pathophysiology of ischemic acute renal failure. J Am Soc Nephrol. 2003;14:2199–2210.
Nelimarkka O. Renal oxygen and lactate metabolism in hemorrhagic shock: an experimental study. Acta Chir Scand Suppl. 1984;518:1–44.
Binns OA, DeLima NF, Buchanan SA, Mauney MC, Cope JT, Thies SD, Shockey KS, Tribble CG, Kron IL. Neutrophil endopeptidase inhibitor improves pulmonary function during reperfusion after 18-hour preservation. J Thorac Cardiovasc Surg. 1996;112:607–613.
Hirose J, Ozawa T, Miura T, Isaji M, Nagao Y, Yamashiro K, Nii A, Kato K, Uemura A. Human neutrophil elastase degrades inter-α-trypsin inhibitor to liberate urinary trypsin inhibitor related proteins. Biol Pharm Bull. 1998;21:651–656.
Masuda T, Sato K, Noda C, Ikeda K, Matsunaga A, Ogura M, Shimizu K, Nagasawa H, Matsuyama N, Izumi T. Protective effect of urinary trypsin inhibitor on myocardial mitochondria during hemorrhagic shock and reperfusion. Crit Care Med. 2003;31: 1987–1992.
Komori M, Takada K, Tomizawa Y, Uezono S, Ozaki M. Urinary trypsin inhibitor improves peripheral microcirculation and bronchospasm associated with systemic anaphylaxis in rabbits in vivo. Shock. 2003;20:189–194.
Chen CC, Liu ZM, Wang HH, He W, Wang Y, Wu WD. Effects of ulinastatin on renal ischemia-reperfusion injury in rats. Acta Pharmacol Sin. 2004;25:1334–1340.
Nakahama H, Obata K, Sugita M. Ulinastatin ameliorates acute ischemic renal injury in rats. Ren Fail. 1996;18:893–898.
Taie S, Yokono S, Ueki M, Ogli K. Effects of ulinastatin (urinary trypsin inhibitor) on ATP, intracellular pH, and intracellular sodium transients during ischemia and reperfusion in the rat kidney in vivo. J Anesth. 2001;15:33–38.
Swartz HM, Clarkson RB. The measurement of oxygen in vivo using EPR techniques. Phys Med Biol. 1998;43:1957–1975.
Hou H, Grinberg OY, Williams B, Grinberg S, Yu H, Alvarenga DL, Wallach H, Buckey J, Swartz HM. The effect of oxygen therapy on brain damage and cerebral pO2 in transient focal cerebral ischemia in the rat. Physiol Meas. 2007;28:963–976.
Hou H, Grinberg OY, Taie S, Leichtweis S, Miyake M, Grinberg S, Xie H, Csete M, Swartz HM. Electron paramagnetic resonance assessment of brain tissue oxygen tension in anesthetized rats. Anesth Analg. 2003;96:1467–1472.
Nakajima K, Goto Y. Differentiation of the anti-shock effect of ulinastatin from steroid hormone, by the continuous observation of microcirculation dynamics. Circ Shock. 1992;36:284–289.
Kawamura T, Inada K, Akasaka N, Wakusawa R. Ulinastatin reduces elevation of cytocines and soluble adhesion molecules during cardiac surgery. Can J Anaesth. 1996;43:456–460.
Aosasa S, Ono S, Seki S, Takayama E, Tadakuma T, Hiraide H, Mochizuki H. Inhibitory effect of protease inhibitor on endothelial cell activation. J Surg Res. 1998;80:182–187.
Inoue K, Takano H, Shimada A, Yanagisawa R, Sakurai M, Yoshino S, Sato H, Yoshikawa T. Urinary trypsin inhibitor protects against systemic inflammation induced by lipopolysaccharide. Mol Pharmacol. 2005;67:673–680.
Yano T, Anraku S, Nakayama R, Ushijima K. Neuroprotective effect of urinary trypsin inhibitor against focal cerebral ischemiareperfusion injury in rats. Anesthesiology. 2003;98:465–473.
Ueki M, Taie S, Chujo K, Asaga T, Iwanaga Y, Ono J, Maekawa N. Urinary trypsin inhibitor reduces inflammatory response in kidney induced by lipopolysaccharide. J Biosci Bioeng. 2007;104: 315–320.
Brezis M, Rosen S. Hypoxia of the renal medulla—its implications for disease. N Engl J Med. 1995;332:647–655.
Juillard L, Lerman LO, Kruger DG, Haas JA, Rucker BC, Polzin JA, Riederer SJ, Romero JC. Blood oxygen leveldependent measurement of acute intra-renal ischemia. Kidney Int. 2004;65:944–950.
Whitehouse T, Stotz M, Taylor V, Stidwill R, Singer M. Tissue oxygen and hemodynamics in renal medulla, cortex, and corticomedullary junction during hemorrhage-reperfusion. Am J Physiol Renal Physiol. 2006;291:F647–F653.
Endo S, Inada K, Taki K, Hoshi S, Yoshida M. Inhibitory effects of ulinastatin on the production of cytokines: implications for the prevention of septicemic shock. Clin Ther. 1990;12:323–326.
Author information
Authors and Affiliations
About this article
Cite this article
Taie, S., Ueki, M., Chujo, K. et al. Urinary trypsin inhibitor ameliorates renal tissue oxygenation after ischemic reperfusion in rats. J Anesth 22, 149–154 (2008). https://doi.org/10.1007/s00540-007-0602-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-007-0602-2