Abstract
Background
IL-33 is a new tissue-derived cytokine constitutively expressed in epithelial cells and plays a role in sensing damage caused by inflammatory diseases. The function of IL-33 in the esophageal mucosa has not been previously described. Accordingly, we examined the expression of IL-33 and its role in the pathogenesis of reflux esophagitis (RE).
Methods
IL-33 in the esophageal mucosa of RE patients and in an in vitro stratified normal esophageal squamous epithelial model was examined at the messenger RNA and protein levels. The correlation of the level of IL-33 and IL-8 or IL-6 was examined. Cell layers were stimulated with bile acids and cytokines. IL-33 was knocked down by small interfering RNA (siRNA). Pharmacological inhibitors and signal transducer and activator of transcription 1 (STAT1) siRNA were used.
Results
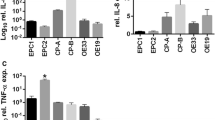
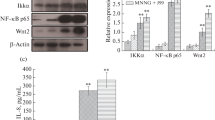
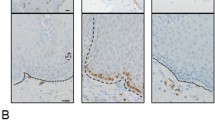
IL-33 was significantly upregulated in RE patients, and was located in the nuclei of basal and suprabasal layers. Upregulated IL-33 messenger RNA expression was correlated with IL-8 and IL-6 expression. In vitro, IL-33 was upregulated in the nuclei of basal and suprabasal layers by interferon-γ (IFNγ), and the upregulation was aggravated by the combination of deoxycholic acid (DCA) and IFNγ. IL-33 knockdown dampened IFNγ- and DCA-induced IL-8 and IL-6 production. IFNγ-induced IL-33 was inhibited by a Janus kinase inhibitor, a p38 mitogen-activated protein kinase inhibitor, and STAT1 siRNA.
Conclusions
Nuclear IL-33 is upregulated in erosive mucosa of RE patients and is correlated with IL-8 and IL-6 levels. The normal esophageal epithelial model enables us to show for the first time that epithelial-cell-derived nuclear but not exogenous IL-33 is located upstream of the production of inflammatory cytokines and can aggravate the inflammation.







Similar content being viewed by others
References
Souza RF, Huo X, Mittal V, et al. Gastroesophageal reflux might cause esophagitis through a cytokine-mediated mechanism rather than caustic acid injury. Gastroenterology. 2009;137:1776–84.
Rieder F, Biancani P, Harnett K, et al. Inflammatory mediators in gastroesophageal reflux disease: impact on esophageal motility, fibrosis, and carcinogenesis. Am J Physiol Gastrointest Liver Physiol. 2010;298:G571–81.
Shan J, Oshima T, Chen X, et al. Trypsin impaired epithelial barrier function and induced IL-8 secretion through basolateral PAR-2: a lesson from a stratified squamous epithelial model. Am J Physiol Gastrointest Liver Physiol. 2012;303:G1105–12.
Shan J, Oshima T, Fukui H, et al. Acidic deoxycholic acid and chenodeoxycholic acid induce interleukin-8 production through p38 mitogen-activated protein kinase and protein kinase A in a squamous epithelial model. J Gastroenterol Hepatol. 2013;28:823–8.
Ma J, Altomare A, Guarino M, et al. HCl-induced and ATP-dependent upregulation of TRPV1 receptor expression and cytokine production by human esophageal epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2012;303:G635–45.
Schmitz J, Owyang A, Oldham E, et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity. 2005;23:479–90.
Meephansan J, Tsuda H, Komine M, et al. Regulation of IL-33 expression by IFN-γ and tumor necrosis factor-α in normal human epidermal keratinocytes. J Invest Dermatol. 2012;132:2593–600.
Lin J, Zhang L, Zhao G, et al. A novel interleukin 33/ST2 signaling regulates inflammatory response in human corneal epithelium. PLoS One. 2013;8:e60963.
Carriere V, Roussel L, Ortega N, et al. IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proc Natl Acad Sci USA. 2007;104:282–7.
Ali S, Mohs A, Thomas M, et al. The dual function cytokine IL-33 interacts with the transcription factor NF-κB to dampen NF-κB-stimulated gene transcription. J Immunol. 2011;187:1609–16.
Pastorelli L, Garg RR, Hoang SB, et al. Epithelial-derived IL-33 and its receptor ST2 are dysregulated in ulcerative colitis and in experimental Th1/Th2 driven enteritis. Proc Natl Acad Sci USA. 2010;107:8017–22.
Kobori A, Yagi Y, Imaeda H, et al. Interleukin-33 expression is specifically enhanced in inflamed mucosa of ulcerative colitis. J Gastroenterol. 2010;45:999–1007.
Chen X, Oshima T, Tomita T, et al. Acidic bile salts modulate the squamous epithelial barrier function by modulating tight junction proteins. Am J Physiol Gastrointest Liver Physiol. 2011;301:G203–9.
Sakashita M, Yoshimoto T, Hirota T, et al. Association of serum interleukin-33 level and the interleukin-33 genetic variant with Japanese cedar pollinosis. Clin Exp Allergy. 2008;38:1875–81.
Oshima T, Gedda K, Koseki J, et al. Establishment of esophageal-like non-keratinized stratified epithelium using normal human bronchial epithelial cells. Am J Physiol Cell Physiol. 2011;300:C1422–9.
Oshima T, Koseki J, Chen X, et al. Acid modulates the squamous epithelial barrier function by modulating the localization of claudins in the superficial layers. Lab Invest. 2012;92:22–31.
Matsuda A, Okayama Y, Terai N, et al. The role of interleukin-33 in chronic allergic conjunctivitis. Invest Ophthalmol Vis Sci. 2009;50:4646–52.
Fitzgerald RC, Onwuegbusi BA, Bajaj-Elliott M, et al. Diversity in the oesophageal phenotypic response to gastro-oesophageal reflux: immunological determinants. Gut. 2002;50:451–9.
Miwa H, Koseki J, Oshima T, et al. Rikkunshito, a traditional Japanese medicine, may relieve abdominal symptoms in rats with experimental esophagitis by improving the barrier function of epithelial cells in esophageal mucosa. J Gastroenterol. 2010;45:478–87.
Jovov B, Que J, Tobey NA, et al. Role of E-cadherin in the pathogenesis of gastroesophageal reflux disease. Am J Gastroenterol. 2011;106:1039–47.
Oguro M, Koike M, Ueno T, et al. Dissociation and dispersion of claudin-3 from the tight junction could be one of the most sensitive indicators of reflux esophagitis in a rat model of the disease. J Gastroenterol. 2011;46:629–38.
Zhong C, Duan L, Wang K, et al. Esophageal intraluminal baseline impedance is associated with severity of acid reflux and epithelial structural abnormalities in patients with gastroesophageal reflux disease. J Gastroenterol. 2013;48:601–10.
Moussion C, Ortega N, Girard JP. The IL-1-like cytokine IL-33 is constitutively expressed in the nucleus of endothelial cells and epithelial cells in vivo: a novel ‘alarmin’? PLoS One. 2008;3:e3331.
Chen X, Oshima T, Shan J, et al. Bile salts disrupt human esophageal squamous epithelial barrier function by modulating tight junction proteins. Am J Physiol Gastrointest Liver Physiol. 2012;303:G199–208.
Rieder F, Cheng L, Harnett KM, et al. Gastroesophageal reflux disease-associated esophagitis induces endogenous cytokine production leading to motor abnormalities. Gastroenterology. 2007;132:154–65.
Seltmann J, Werfel T, Wittmann M. Evidence for a regulatory loop between IFN-γ and IL-33 in skin inflammation. Exp Dermatol. 2013;22:102–7.
Nomura K, Kojima T, Fuchimoto J, et al. Regulation of interleukin-33 and thymic stromal lymphopoietin in human nasal fibroblasts by proinflammatory cytokines. Laryngoscope. 2012;122:1185–92.
Zhong YQ, Lin Y, Xu Z. Expression of IFN-γ and IL-4 in the esophageal mucosa of patients with reflux esophagitis and Barrett’s esophagus and their relationship with endoscopic and histologic grading. Dig Dis Sci. 2011;56:2865–70.
Corrado G, Zicari A, Cavaliere M, et al. Increased release of interleukin-6 by oesophageal mucosa in children with reflux oesophagitis. Eur J Gastroenterol Hepatol. 1999;11:839–43.
Isomoto H, Wang A, Mizuta Y, et al. Elevated levels of chemokines in esophageal mucosa of patients with reflux esophagitis. Am J Gastroenterol. 2003;98:551–6.
Isomoto H, Inoue K, Kohno S. Interleukin-8 levels in esophageal mucosa and long-term clinical outcome of patients with reflux esophagitis. Scand J Gastroenterol. 2007;42:410–1.
Hurst SM, Wilkinson TS, McLoughlin RM, et al. IL-6 and its soluble receptor orchestrate a temporal switch in the pattern of leukocyte recruitment seen during acute inflammation. Immunity. 2001;14:705–14.
Moulin D, Donze O, Talabot-Ayer D, et al. Interleukin (IL)-33 induces the release of pro-inflammatory mediators by mast cells. Cytokine. 2007;40:216–25.
Sedhom MA, Pichery M, Murdoch JR, et al. Neutralisation of the interleukin-33/ST2 pathway ameliorates experimental colitis through enhancement of mucosal healing in mice. Gut. 2013;62:1714–23.
Lange A, Mills RE, Lange CJ, et al. Classical nuclear localization signals: definition, function, and interaction with importin α. J Biol Chem. 2007;282:5101–5.
Sharma S, Kulk N, Nold MF, et al. The IL-1 family member 7b translocates to the nucleus and down-regulates proinflammatory cytokines. J Immunol. 2008;180:5477–82.
Polumuri SK, Jayakar GG, Shirey KA, et al. Transcriptional regulation of murine IL-33 by TLR and non-TLR agonists. J Immunol. 2012;189:50–60.
Liu L, Wang Y, Fan Y, et al. IFN-γ activates cAMP/PKA/CREB signaling pathway in murine peritoneal macrophages. J Interferon Cytokine Res. 2004;24:334–42.
Acknowledgments
We thank Noriko Kamiya for excellent technical assistance. This study was supported by a Grant-in Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (25460939 to T.O.).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shan, J., Oshima, T., Muto, T. et al. Epithelial-derived nuclear IL-33 aggravates inflammation in the pathogenesis of reflux esophagitis. J Gastroenterol 50, 414–423 (2015). https://doi.org/10.1007/s00535-014-0988-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-014-0988-1