Abstract
Background
The mechanisms of skin aging have not been completely elucidated. Anecdotal data suggests that EGFR inhibition accelerates aging-like skin changes.
Objective
The objective of the study was to evaluate the clinical characteristics and investigate the cellular and molecular mechanisms underlying skin changes associated with the use of EFGRIs.
Patients and methods
Patients during prolonged treatment with EGFRIs (>3 months) were analyzed for aging-like skin changes. Baseline EGFR expression was compared in young (<25 years old) vs. old (> 65 years old) skin. In addition, the regulation of extracellular matrix, senescence-associated genes, and cell cycle status was measured in primary human keratinocytes treated with erlotinib in vitro.
Results
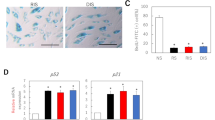
There were progressive signs of skin aging, including xerosis cutis, atrophy, rhytide formation, and/or actinic purpura in 12 patients. Keratinocytes treated with erlotinib in vitro showed a significant down-modulation of hyaluronan synthases (HAS2 and HAS3), whereas senescence-associated genes (p21, p53, IL-6, maspin) were upregulated, along with a G1 cell cycle arrest and stronger SA β-Gal activity. There was significantly decreased baseline expression in EGFR density in aged skin, when compared to young controls.
Conclusions
EGFR inhibition results in molecular alterations in keratinocytes that may contribute to the observed skin aging of patients treated with respective targeted agents.




Similar content being viewed by others
Abbreviations
- AE:
-
adverse event
- ECM:
-
extracellular matrix
- EGFRI:
-
epidermal growth factor (EGF) receptor (EGFR) inhibitor
- HAS:
-
hyaluronan (HA) synthase
- IL:
-
interleukin
- MSKCC:
-
Memorial Sloan Kettering Cancer Center
- PCR:
-
polymerase chain reaction
- ROS:
-
reactive oxygen species
- SA β-Gal:
-
senescence-associated β-galactosidase
- SASP:
-
senesence-associated secretory phenotype
- TGF:
-
transforming growth factor
- TNF:
-
tumor necrosis factor
References
Joshi SS, Ortiz S, Witherspoon JN, Rademaker A, West DP, Anderson R, Rosenbaum SE, Lacouture ME (2010) Effects of epidermal growth factor receptor inhibitor-induced dermatologic toxicities on quality of life. Cancer 116:3916–3923
Lacouture ME (2006) Mechanisms of cutaneous toxicities to EGFR inhibitors. Nat Rev Cancer 6:803–812
Gerber PA, Buhren BA, Cevikbas F, Bolke E, Steinhoff M, Homey B (2010) Preliminary evidence for a role of mast cells in epidermal growth factor receptor inhibitor-induced pruritus. J Am Acad Dermatol 63:163–165
Gerber PA, Buhren BA, Homey B (2011) More on aprepitant for erlotinib-induced pruritus. The New England journal of medicine 364:486–487
Gerber PA, Homey B (2008) Images in clinical medicine. Erlotinib-induced hair alterations. The New England journal of medicine 358: 1175
Segaert S, Van Cutsem E (2005) Clinical signs, pathophysiology and management of skin toxicity during therapy with epidermal growth factor receptor inhibitors. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO 16:1425–1433
Lichtenberger BM, Gerber PA, Holcmann M, Buhren BA, Amberg N, Smolle V, Schrumpf H, Boelke E, Ansari P, Mackenzie C, Wollenberg A, Kislat A, Fischer JW, Rock K, Harder J, Schroder JM, Homey B, Sibilia M (2013) Epidermal EGFR controls cutaneous host defense and prevents inflammation. Science translational medicine 5: 199ra111
Li Y, Liang J, Yang T, Monterrosa Mena J, Huan C, Xie T, Kurkciyan A, Liu N, Jiang D, Noble PW (2016) Hyaluronan synthase 2 regulates fibroblast senescence in pulmonary fibrosis. Matrix biology: journal of the International Society for Matrix Biology. doi:10.1016/j.matbio.2016.03.004
Tzellos TG, Klagas I, Vahtsevanos K, Triaridis S, Printza A, Kyrgidis A, Karakiulakis G, Zouboulis CC, Papakonstantinou E (2009) Extrinsic ageing in the human skin is associated with alterations in the expression of hyaluronic acid and its metabolizing enzymes. Exp Dermatol 18:1028–1035
Rufini A, Tucci P, Celardo I, Melino G (2013) Senescence and aging: the critical roles of p53. Oncogene 32:5129–5143
Childs BG, Durik M, Baker DJ, van Deursen JM (2015) Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med 21:1424–1435
Nickoloff BJ, Lingen MW, Chang BD, Shen M, Swift M, Curry J, Bacon P, Bodner B, Roninson IB (2004) Tumor suppressor maspin is up-regulated during keratinocyte senescence, exerting a paracrine antiangiogenic activity. Cancer Res 64:2956–2961
Garbers C, Kuck F, Aparicio-Siegmund S, Konzak K, Kessenbrock M, Sommerfeld A, Haussinger D, Lang PA, Brenner D, Mak TW, Rose-John S, Essmann F, Schulze-Osthoff K, Piekorz RP, Scheller J (2013) Cellular senescence or EGFR signaling induces interleukin 6 (IL-6) receptor expression controlled by mammalian target of rapamycin (mTOR). Cell Cycle 12:3421–3432
Kaya G, Saurat JH (2007) Dermatoporosis: a chronic cutaneous insufficiency/fragility syndrome. Clinicopathological features, mechanisms, prevention and potential treatments. Dermatology 215:284–294
Gerber PA, Enderlein E, Homey B (2008) The Koebner-phenomenon in epidermal growth factor receptor inhibitor-induced cutaneous adverse effects. J Clin Oncol Off J Am Soc Clin Oncol 26:2790–2792
Baselga J (2002) Targeting the epidermal growth factor receptor with tyrosine kinase inhibitors: small molecules, big hopes. J Clin Oncol Off J Am Soc Clin Oncol 20:2217–2219
Stern RS (2004) Clinical practice. Treatment of photoaging. The New England journal of medicine 350:1526–1534
Iwasa H, Yu S, Xue J, Driscoll M (2010) Novel EGF pathway regulators modulate C. elegans healthspan and lifespan via EGF receptor, PLC-gamma, and IP3R activation. Aging Cell 9:490–505
Rongo C (2011) Epidermal growth factor and aging: a signaling molecule reveals a new eye opening function. Aging 3:896–905
Yu S, Driscoll M (2011) EGF signaling comes of age: promotion of healthy aging in C. elegans. Exp Gerontol 46:129–134
Shurin GV, Yurkovetsky ZR, Chatta GS, Tourkova IL, Shurin MR, Lokshin AE (2007) Dynamic alteration of soluble serum biomarkers in healthy aging. Cytokine 39:123–129
Green MR, Basketter DA, Couchman JR, Rees DA (1983) Distribution and number of epidermal growth factor receptors in skin is related to epithelial cell growth. Dev Biol 100:506–512
Shiraha H, Gupta K, Drabik K, Wells A (2000) Aging fibroblasts present reduced epidermal growth factor (EGF) responsiveness due to preferential loss of EGF receptors. J Biol Chem 275:19343–19351
Jenkins G (2002) Molecular mechanisms of skin ageing. Mech Ageing Dev 123:801–810
Campisi J (2013) Aging, cellular senescence, and cancer. Annu Rev Physiol 75:685–705
Coppe JP, Desprez PY, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118
Yarden Y, Sliwkowski MX (2001) Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2:127–137
Rodriguez-Fragoso L, Melendez K, Hudson LG, Lauer FT, Burchiel SW (2009) EGF-receptor phosphorylation and downstream signaling are activated by benzo[a]pyrene 3,6-quinone and benzo[a]pyrene 1,6-quinone in human mammary epithelial cells. Toxicol Appl Pharmacol 235:321–328
Schmidt-Ullrich RK, Mikkelsen RB, Dent P, Todd DG, Valerie K, Kavanagh BD, Contessa JN, Rorrer WK, Chen PB (1997) Radiation-induced proliferation of the human A431 squamous carcinoma cells is dependent on EGFR tyrosine phosphorylation. Oncogene 15:1191–1197
Xu Y, Shao Y, Voorhees JJ, Fisher GJ (2006) Oxidative inhibition of receptor-type protein-tyrosine phosphatase kappa by ultraviolet irradiation activates epidermal growth factor receptor in human keratinocytes. J Biol Chem 281:27389–27397
Kang S, Chung JH, Lee JH, Fisher GJ, Wan YS, Duell EA, Voorhees JJ (2003) Topical N-acetyl cysteine and genistein prevent ultraviolet-light-induced signaling that leads to photoaging in human skin in vivo. The Journal of investigative dermatology 120:835–841
Wan YS, Wang ZQ, Shao Y, Voorhees JJ, Fisher GJ (2001) Ultraviolet irradiation activates PI 3-kinase/AKT survival pathway via EGF receptors in human skin in vivo. Int J Oncol 18:461–466
Xu YR, Fisher GJ (2005) Ultraviolet (UV) light irradiation induced signal transduction in skin photoaging. J Dermatol Sci: S1-S8
Peus D, Vasa RA, Meves A, Beyerle A, Pittelkow MR (2000) UVB-induced epidermal growth factor receptor phosphorylation is critical for downstream signaling and keratinocyte survival. Photochem Photobiol 72:135–140
Ikeyama S, Kusumoto K, Ogata S, Miyake H, Teshima S, Nikawa T, Rokutan K, Tashiro S (1999) A nontoxic heat shock protein 70 inducer, geranylgeranylacetone, prevents apoptosis of primary cultures of rat hepatocyted induced by hydrogen peroxide or ethanol. Gastroenterology 116:A1222–A1222
Davalos AR, Coppe JP, Campisi J, Desprez PY (2010) Senescent cells as a source of inflammatory factors for tumor progression. Cancer Metastasis Rev 29:273–283
Rock K, Meusch M, Fuchs N, Tigges J, Zipper P, Fritsche E, Krutmann J, Homey B, Reifenberger J, Fischer JW (2012) Estradiol protects dermal hyaluronan/versican matrix during photoaging by release of epidermal growth factor from keratinocytes. J Biol Chem 287:20056–20069
Haferkamp S, Borst A, Adam C, Becker TM, Motschenbacher S, Windhovel S, Hufnagel AL, Houben R, Meierjohann S (2013) Vemurafenib induces senescence features in melanoma cells. The Journal of investigative dermatology 133:1601–1609
Wang M, Morsbach F, Sander D, Gheorghiu L, Nanda A, Benes C, Kriegs M, Krause M, Dikomey E, Baumann M, Dahm-Daphi J, Settleman J, Willers H (2011) EGF receptor inhibition radiosensitizes NSCLC cells by inducing senescence in cells sustaining DNA double-strand breaks. Cancer Res 71:6261–6269
Schad K, Baumann Conzett K, Zipser MC, Enderlin V, Kamarashev J, French LE, Dummer R (2010) Mitogen-activated protein/extracellular signal-regulated kinase kinase inhibition results in biphasic alteration of epidermal homeostasis with keratinocytic apoptosis and pigmentation disorders. Clinical cancer research: an official journal of the American Association for Cancer Research 16:1058–1064
El-Abaseri TB, Putta S, Hansen LA (2006) Ultraviolet irradiation induces keratinocyte proliferation and epidermal hyperplasia through the activation of the epidermal growth factor receptor. Carcinogenesis 27:225–231
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
BAB, HS, PH, EB, DS, VRB, and CR have no conflicts of interest to declare; PAG has a speaking, consultant or advisory role with Amgen, Galderma, and Roche; MEL has a speaking, consultant or advisory role with Advancell, Amgen, AstraZeneca, Augmentium, Aveo, Bayer, Berg Pharma, Biopharm Communications, Boehringer Ingelheim, Brickell Biotech, Bristol-Myers Squibb, Clinical Assistance Programs, Clinical Care Options, EMD Serono, Envision Communications, Foamix, Galderma, Genentech, GlaxoSmithKline, Helsinn, Institute for Medical Education and Research, Integro-MC, Lindi Skin, Medscape, Medtrend International, Merck, Nerre Therapeutics, Novartis, Novocure, Oncology Specialty Group, OSI Pharmaceuticals, Permanyer, Physicians Education Resource, Pierre Fabre, Pfizer, Reata Pharmaceuticals, Roche, Sandoz, Sanofi Aventis, and Threshold Pharmaceuticals.
Funding
This work was supported by a grant of the Duesseldorf Science Commission and a grant by Roche Pharmaceuticals. MEL was funded in part through the NIH/Cancer Center Support Grant P30 CA008748.
Additional information
The contents of this manuscript have not been presented, and are not under consideration for publication elsewhere.
Rights and permissions
About this article
Cite this article
Gerber, P.A., Buhren, B.A., Schrumpf, H. et al. Mechanisms of skin aging induced by EGFR inhibitors. Support Care Cancer 24, 4241–4248 (2016). https://doi.org/10.1007/s00520-016-3254-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-016-3254-7