Abstract
Purpose
Chemotherapy-induced peripheral neuropathy (CIPN) is increasing with introduction of new and combination cancer pharmacotherapies. This study evaluated associations between clinical and self-report measurements and current perception threshold (CPT), a neuroselective measure of sensory nerve function that may detect asymptomatic CIPN damage.
Methods
Data for this secondary analysis were from a prospective, observational study using CPT to evaluate CIPN. Bivariate mixed models, accounting for the intraclass correlation between repeated patient assessments, were used to assess the relationship between CPT at each frequency (5, 250, and 2,000 Hz) and each subjective measure (Neuropathic Pain Scale, FACT-GOGntx) and objective measurement (quantitative sensory testing, deep tendon reflexes, and grip strength).
Results
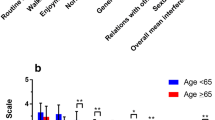
A total of 29 chemotherapy-naïve subjects with various cancer types had a mean age of 56.7 (SD 10.4); nine subjects developed CIPN grade >1 using NCI CTC-AE criteria. Cold detection thresholds were inversely associated with CPT 5 [b(95 % CI) = −2.5(−4.5, −0.5)] and CPT 2,000 [−7.5(−11.8, −3.3)] frequencies. FACT GOG-ntx quality of life (QoL) scale and neurotoxicity and function subscales were inversely associated with CPT 2,000 [−1.8 (−3.5, −0.05), −2.2 (−4.2, −0.2), and −5.4 (−9.8, −0.9), respectively], indicating worsening QoL, impairment, and function as hypoesthesia increases.
Conclusions
CPT 2,000 may identify impending worsening of patient-reported outcomes such as QoL.
Similar content being viewed by others
References
Edwards BK et al (2005) Annual report to the nation on the status of cancer, 1975-2002, featuring population-based trends in cancer treatment. J Natl Cancer Inst 97(19):1407–1427
World Health Organization (2001) International Classification of Functioning, Disability, and health (ICF)
Lee JJ, Swain SM (2006) Peripheral neuropathy induced by microtubule-stabilizing agents. J Clin Oncol 24(10):1633–1642
Mielke S, Sparreboom A, Mross K (2006) Peripheral neuropathy: a persisting challenge in paclitaxel-based regimes. Eur J Cancer 42(1):24–30
Postma TJ, Heimans JJ (2000) Grading of chemotherapy-induced peripheral neuropathy. Ann Oncol 11(5):509–513
Quasthoff S, Hartung HP (2002) Chemotherapy-induced peripheral neuropathy. J Neurol 249(1):9–17
Chaudhry V et al (1994) Inter- and intraexaminer reliability of nerve conduction measurements in patients with diabetic neuropathy. Neurology 44(8):1459–1462
Polomano RC et al (2001) A painful peripheral neuropathy in the rat produced by the chemotherapeutic drug, paclitaxel. Pain 94(3):293–304
Roytta M, Raine CS (1985) Taxol-induced neuropathy: further ultrastructural studies of nerve fibre changes in situ. J Neurocytol 14(1):157–175
Dougherty PM et al (2004) Taxol-induced sensory disturbance is characterized by preferential impairment of myelinated fiber function in cancer patients. Pain 109(1–2):132–142
Fischer SJ et al (2001) Alterations in cell cycle regulation underlie cisplatin induced apoptosis of dorsal root ganglion neurons in vivo. Neurobiol Dis 8(6):1027–1035
Andre T et al (2004) Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 350(23):2343–2351
Trotti A, Colevas AD, Setser A et al (2003) CTCAE v3.0: development of a comprehensive grading system for the adverse effects of cancer treatment. Semin Radiat Oncol 13(3):176–181
Oken MM et al (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5(6):649–655
Miller AB et al (1981) Reporting results of cancer treatment. Cancer 47(1):207–214
Cavaletti G et al (2006) Multi-center assessment of the Total Neuropathy Score for chemotherapy-induced peripheral neurotoxicity. J Peripher Nerv Syst 11(2):135–141
Wolf S et al (2008) Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. Eur J Cancer 44(11):1507–1515
Ma L et al (2012) Single application of A2 NTX, a botulinum toxin A2 subunit, prevents chronic pain over long periods in both diabetic and spinal cord injury-induced neuropathic pain models. J Pharmacol Sci 119(3):282–286
Matsumoto M et al (2006) Characterization of three different sensory fibers by use of neonatal capsaicin treatment, spinal antagonism and a novel electrical stimulation-induced paw flexion test. Mol Pain 2:16
Matsumoto M et al (2008) Pharmacological switch in Abeta-fiber stimulation-induced spinal transmission in mice with partial sciatic nerve injury. Mol Pain 4:25
Koga K et al (2005) Selective activation of primary afferent fibers evaluated by sine-wave electrical stimulation. Mol Pain 1:13
Katims JJ et al (1987) Constant current sine wave transcutaneous nerve stimulation for the evaluation of peripheral neuropathy. Arch Phys Med Rehabil 68(4):210–213
Takekuma K et al (2000) Age and gender differences in skin sensory threshold assessed by current perception in community dwelling Japanese. J Epidemiol 10(1):S33–S38
Rendell MS et al (1989) Mapping diabetic sensory neuropathy by current perception threshold testing. Diabetes Care 12(9):636–640
Rendell MS et al (1989) A comparison of nerve conduction velocities and current perception thresholds as correlates of clinical severity of diabetic sensory neuropathy. J Neurol Neurosurg Psychiatry 52(4):502–511
Masson EA, Boulton AJ (1991) The Neurometer: validation and comparison with conventional tests for diabetic neuropathy. Diabet Med 8(Spec No):S63–S66
Nishimura H et al (2004) Weekly paclitaxel therapy for locoregional lymph node recurrence of estrogen-receptor-negative breast cancer. Gan To Kagaku Ryoho 31(4):627–630
Tack CJ et al (1994) Comparison of clinical examination, current and vibratory perception threshold in diabetic polyneuropathy. Neth J Med 44(2):41–49
Doi D et al (2003) Evaluation of the neurotoxicity of paclitaxel and carboplatin by current perception threshold in ovarian cancer patients. J Nihon Med Sch 70(2):129–134
Rolke R, Baron R, Maier C et al (2006) Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): standardized protocol and reference values. Pain 123(3):231–243
Cavaletti G et al (2013) The chemotherapy-induced peripheral neuropathy outcome measures standardization study: from consensus to the first validity and reliability findings. Ann Oncol 24(2):454–462
Galer BS, Jensen MP (1997) Development and preliminary validation of a pain measure specific to neuropathic pain: the Neuropathic Pain Scale. Neurology 48(2):332–338
Calhoun EA et al (2003) Psychometric evaluation of the Functional Assessment of Cancer Therapy/Gynecologic Oncology Group-Neurotoxicity (Fact/GOG-Ntx) questionnaire for patients receiving systemic chemotherapy. Int J Gynecol Cancer 13(6):741–748
Huang HQ et al (2007) Validation and reduction of FACT/GOG-Ntx subscale for platinum/paclitaxel-induced neurologic symptoms: a gynecologic oncology group study. Int J Gynecol Cancer 17(2):387–393
Bell-Krotoski JA et al (1995) Threshold detection and Semmes-Weinstein monofilaments. J Hand Ther 8(2):155–162
de Sonnaville JJ et al (1997) The prevalence and determinants of foot ulceration in type II diabetic patients in a primary health care setting. Diabetes Res Clin Pract 35(2–3):149–156
Felix E, Widerstrom E (2009) Reliability and validity of quantitative sensory testing in persons with spinal cord injury and neuropathic pain. J Rehabil Res Dev 46:69–84
Hilz MJ, Dutsch M, Neundorfer B (1998) Autonomic disorders in polyneuropathies. Med Klin (Munich) 93(9):533–540
Martina IS et al (1998) Measuring vibration threshold with a graduated tuning fork in normal aging and in patients with polyneuropathy. European Inflammatory Neuropathy Cause and Treatment (INCAT) group. J Neurol Neurosurg Psychiatry 65(5):743–747
Merkies IS et al (2000) Reliability and responsiveness of a graduated tuning fork in immune mediated polyneuropathies. The Inflammatory Neuropathy Cause and Treatment (INCAT) Group. J Neurol Neurosurg Psychiatry 68(5):669–671
Smieja M et al (1999) Clinical examination for the detection of protective sensation in the feet of diabetic patients. International Cooperative Group for Clinical Examination Research. J Gen Intern Med 14(7):418–424
Hallett M (1993) NINDS myotatic reflex scale. Neurology 43(12):2723
Richardson JK (2002) The clinical identification of peripheral neuropathy among older persons. Arch Phys Med Rehabil 83(11):1553–1558
Fauci AS et al (ed) (2012) Harrison’s principles of internal medicine, 18ed.
Acknowledgments
The authors would like to acknowledge the contributions of Sharon Gordon, DDS, MPH, PhD, University of Maryland, Baltimore School of Dentistry; and Raymond Dionne, DDS, PhD, Food and Drug Administration for their critical input into study conceptualization and design. The views expressed are those of the authors and do not reflect the official policy or position of the Uniformed Services University of the Health Sciences, the Department of Defense, or the United States government. This study was funded by the National Institute of Health/National Institute of Nursing Research grant, P30 University of Maryland, Baltimore Center for Pain Studies to S.G. Dorsey (NRO11396-01).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Kathleen A. Griffith and Darren J. Couture are the co-first authors.
Rights and permissions
About this article
Cite this article
Griffith, K.A., Couture, D.J., Zhu, S. et al. Evaluation of chemotherapy-induced peripheral neuropathy using current perception threshold and clinical evaluations. Support Care Cancer 22, 1161–1169 (2014). https://doi.org/10.1007/s00520-013-2068-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-013-2068-0