Abstract
Purpose
Cancer-related fatigue (CRF) is frequently overlooked. Adherence to treatment guidelines may be related to the patient’s views about illness. This study aimed at exploring patients’ views about CRF and determining whether they are congruent with best practice treatments.
Methods
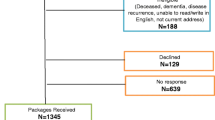
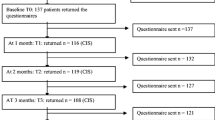
Data were collected in 160 consecutive patients hospitalized in a supportive care setting. Biological, clinical, and psychological variables were assessed using validated questionnaires. Patients were also asked to complete the Brief Fatigue Inventory (BFI) and a questionnaire investigating their main symptoms and views about CRF and its management.
Results
Patients were mainly men (60%); median age was 66 years. Various cancer diagnoses were represented; 17.5% had primary local diseases, 40% local recurrences, and 42.5% metastatic diseases. The majority of the patients experienced moderate or severe CRF (76.3%) on the BFI. Fatigue was the most frequently reported symptom (87.5%). Only anxiety, depression, and dimensions of quality of life were significantly related with CRF. Two thirds of the patients associated CRF with cancer-related morbidities. As for the best treatments, patients first stressed control of adverse effects. Over half of the patients were reluctant to report fatigue, mainly because they considered fatigue as an unavoidable side effect, but also because they feared a change towards less active/aggressive treatments.
Conclusion
Patients mostly consider that CRF must be tolerated. Guidelines emphasize activity enhancement strategies as beneficial. The patients’ preferences for rest rather than activity may be related to their high level of fatigue, which leads them to disregard activity as a possible treatment.
Similar content being viewed by others
References
Mock V, Atkinson A, Barsevick A et al (2000) NCCN practice guidelines for cancer-related fatigue. Oncology 14:151–161
National Comprehensive Cancer Network (NCCN) clinical practice guidelines in oncology – cancer-related fatigue, v.1.2009. http://www.nccn.org/professionals/physician_gls/PDF/fatigue.pdf. Accessed 7 Dec 2009
Stone PC, Minton O (2008) Cancer-related fatigue. Eur J Cancer 44:1097–1104
Ahlberg K, Ekman T, Gaston-Johansson F, Mock V (2003) Assessment and management of cancer-related fatigue in adults. Lancet 362:640–650
Dy SM, Lorenz KA, Naeim A, Sanati H, Walling A, Asch SM (2008) Evidence-based recommendations for cancer fatigue, anorexia, depression, and dyspnea. J Clin Oncol 26:3886–3895
Lesage P, Portenoy RK (2002) Management of fatigue in the cancer patient. Oncology 16:373–381
Flechtner H, Bottomley A (2003) Fatigue and quality of life: lessons from the real world. Oncologist 8:5–9
Stasi R, Abriani L, Beccaglia P, Terzoli E, Amadori S (2003) Cancer-related fatigue: evolving concepts in evaluation and treatment. Cancer 98:1786–1801
Morrow GR, Andrews PL, Hickok JT, Roscoe JA, Matteson S (2002) Fatigue associated with cancer and its treatment. Support Care Cancer 10:389–398
Hofman M, Ryan JL, Figueroa-Moseley CD, Jean-Pierre P, Morrow GR (2007) Cancer-related fatigue: the scale of the problem. Oncologist 12(Suppl 1):4–10
Stone P, Richardson A, Ream E, Smith AG, Kerr DJ, Kearney N (2000) Cancer-related fatigue: inevitable, unimportant and untreatable? Results of a multi-centre patient survey. Cancer fatigue forum. Ann Oncol 11:971–975
Minton O, Stone P, Richardson A, Sharpe M, Hotopf M (2008) Drug therapy for the management of cancer related fatigue. Cochrane Database Syst Rev 1:CD006704
Cramp F, Daniel J (2008) Exercise for the management of cancer-related fatigue in adults. Cochrane Database Syst Rev 2:CD006145
Goedendorp MM, Gielissen MF, Verhagen CA, Bleijenberg G (2009) Psychosocial interventions for reducing fatigue during cancer treatment in adults. Cochrane Database Syst Rev 1:CD006953
Donovan JL, Blake DR (1992) Patient non-compliance: deviance or reasoned decision-making? Soc Sci Med 34:507–513
Haynes RB, Yao X, Degani A, Kripalani S, Garg A, McDonald HP (2005) Interventions for enhancing medication adherence. Cochrane Database Syst Rev, Issue 4
Blank T, Graves K, Sepucha K, Llewellyn-Thomas H (2006) Understanding treatment decision making: contexts, commonalities, complexities, and challenges. Ann Behav Med 32:211–217
Piguet V, Cedraschi C, Dumont P, Desmeules J, Allaz AF, Dayer P (2007) Patients’ representations of antidepressants: a clue to nonadherence? Clin J Pain 23:669–675
Mendoza TR, Wang XS, Cleeland CS (1999) The rapid assessment of fatigue severity in cancer patients. Cancer 85:1186–1196
Whitehead L (2009) The measurement of fatigue in chronic illness: a systematic review of unidimensional and multidimensional fatigue measures. J Pain Symptom Manage 37:107–128
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. J Clin Oncol 5:649–655
Aaronson NK, Ahmedzai S, Bergman B et al (1993) The European organization for research and treatment of cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85:365–376
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67:361–370
Razavi D, Delvaux N, Farvacques C, Robaye E (1989) Validation de la version française du HADS dans une population de patients cancéreux hospitalisés. Rev Psychol App 39:295–308
Passik SD, Kirsh KL, Donaghy K, Holtsclaw E, Theobald D, Cella D, Breitbart W, Fatigue Coalition (2002) Patient-related barriers to fatigue communication: initial validation of the fatigue management barriers questionnaire. J Pain Symptom Manage 24:481–493
Escalante CP (2003) Treatment of cancer-related fatigue: an update. Support Care Cancer 11:79–83
Echteld MA, Passchier J, Teunissen S, Claessen S, de Wit R, van der Rijt CC (2007) Multidimensional fatigue and its correlates in hospitalised advanced cancer patients. Eur J Cancer 43:1030–1036
Cella D (2006) Quality of life and clinical decisions in chemotherapy-induced anemia. Oncology 20:25–28
Jacobsen PB, Garland LL, Booth-Jones M, Donovan KA, Thors CL, Winters E, Grendys E (2004) Relationship of hemoglobin levels to fatigue and cognitive functioning among cancer patients receiving chemotherapy. J Pain Symptom Manage 28:7–18
Romito F, Montanaro R, Corvasce C, Di Bisceglie M, Mattioli V (2008) Is cancer-related fatigue more strongly correlated to haematological or to psychological factors in cancer patients? Support Care Cancer 16:943–946
Collins S, de Vogel-Voogt E, Visser A, van der Heide A (2008) Presence, communication and treatment of fatigue and pain complaints in incurable cancer patients. Patient Educ Couns 72:102–108
Higginson IJ, Costantini M (2008) Dying with cancer, living well with advanced cancer. Eur J Cancer 44:1414–1424
Brown LF, Kroenke K (2009) Cancer-related fatigue and its associations with depression and anxiety: a systematic review. Psychosomatics 50:440–447
Hay MC, Weisner TS, Subramanian S, Duan N, Niedzinski EJ, Kravitz RL (2008) Harnessing experience: exploring the gap between evidence-based medicine and clinical practice. J Eval Clin Pract 14:707–713
Greenfield S, Kravitz R, Duan N, Kaplan SH (2007) Heterogeneity of treatment effects: implications for guidelines, payment, and quality assessment. Am J Med 120:S3–S9
Acknowledgment
The authors are grateful to Mrs. Wendy Pickering-Di Silvestro for language editing.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have full control of all primary data and agree to allow the journal to review their data if requested.
Rights and permissions
About this article
Cite this article
Luthy, C., Cedraschi, C., Pugliesi, A. et al. Patients’ views about causes and preferences for the management of cancer-related fatigue—a case for non-congruence with the physicians?. Support Care Cancer 19, 363–370 (2011). https://doi.org/10.1007/s00520-010-0826-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-010-0826-9