Abstract
Background
Oral chemotherapy regimens have emerged as comparably effective to intravenous regimens with the potential for less resource utilization, fewer treatment side effects and a better quality-of-life outcome. The objective of this retrospective chart review was to examine these issues among patients who received single-agent taxane therapy vs. single-agent capecitabine for first- or second-line treatment of metastatic breast cancer (MBC)
Methods
Data from the medical charts of 61 patients who received single-agent capecitabine, docetaxel, or paclitaxel therapy were supplemented with data from the 38-item Patient Care Monitor (PCM) survey of symptom burden and quality of life, prospectively collected during chemotherapy. The endpoints were PCM index scores, hospitalization during treatment, and the number of clinic visits during treatment.
Results
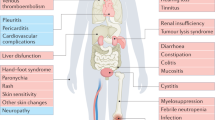
The sample was 75% Caucasian, 16% African–American, with a mean age of 59.4 years. Taxane-treated patients had more clinic visits than capecitabine patients, were more likely to have been hospitalized during treatment, and had worse treatment side effects. Controlling for depression, infusion-treated patients reported greater acute distress at the start of therapy than those on oral medication.
Conclusion
Capecitabine for MBC was associated with a more favorable outcome regarding treatment side effects and quality of life, with reduced resource burden to patients and providers. Physicians may have differentially selected patients with greater depressive symptoms for capecitabine therapy.




Similar content being viewed by others
References
Bigot A (1974) The relevance of American Life Satisfaction Indices for research on British subjects before and after retirement. Age Ageing 3:113–121. doi:10.1093/ageing/3.2.113
Borner M, Scheithauer W, Twelves C, Maroun J, Wilke H (2001) Answering patients’ needs: oral alternatives to intravenous therapy. Oncologist 6(Suppl 4):12–16. doi:10.1634/theoncologist.6-suppl_4-12
Chang VT, Hwang SS, Feuerman M, Kasimis BS, Thaler HT (2000) The memorial symptom assessment scale short form (MSAS-SF). Cancer 89:1162–1171. doi:10.1002/1097-0142(20000901)89:5<1162::AID-CNCR26>3.0.CO;2-Y
Cnaan A, Laird NM, Slasor P (1997) Using the general linear mixed model to analyse unbalanced repeated measures and longitudinal data. Stat Med 16:2349–2380. doi:10.1002/(SICI)1097-0258(19971030)16:20<2349::AID-SIM667>3.0.CO;2-E
Coates AS, Hürny C, Peterson HF et al (2000) Quality-of-life scores predict outcome in metastatic but not early breast cancer. J Clin Oncol 18:2768–3774
Derogatis LR, Melisaratos N (1983) The brief symptom inventory: an introductory report. Psychol Med 13:595–605
Diener E, Emmons RA, Larsen RJ, Griffin S (1985) The satisfaction with life scale. J Pers Assess 49:71–75. doi:10.1207/s15327752jpa4901_13
El Helw L, Coleman RE (2004) Reduced dose capecitabine is an effective and well-tolerated treatment in patients with metastatic breast cancer. Breast 14:368–374. doi:10.1016/j.breast.2004.12.005
Fortner B, Okon T, Schwartzberg L, Tauer K, Houts AC (2003) The cancer care monitor: psychometric content evaluation and pilot testing of a computer administered system for symptom screening and quality of life in adult cancer patients. J Pain Symptom Manage 26:1077–1092. doi:10.1016/j.jpainsymman.2003.04.003
Ghersi D, Wilcken N, Simes RJ (2005) A systematic review of taxane-containing regimens for metastatic breast cancer. Br J Cancer 93:293–301. doi:10.1038/sj.bjc.6602680
Gralow JR (2005) Optimizing the treatment of metastatic breast cancer. Breast Cancer Res Treat 89(Suppl 1):S9–S15. doi:10.1007/s10549-005-0143-z
Hays RD, Sherbourne CD, Mazel RM (1993) The RAND 36-item health survey 1.0. Health Econ 2:217–227. doi:10.1002/hec.4730020305
Jackisch C (2006) HER-2-positive metastatic breast cancer: optimizing trastuzumab-based therapy. Oncologist 11(Suppl 1):34–41. doi:10.1634/theoncologist.11-90001-34
Kopec JA, Yothers G, Ganz PA, Land SR, Cecchini RS, Wieand HS et al (2007) Quality of life in operable colon cancer patients receiving oral compared with intravenous chemotherapy: results from National Surgical Adjuvant Breast and Bowel Project Trial C-06. J Clin Oncol 25:424–430. doi:10.1200/JCO.2005.05.2597
Lembersky BC, Wieand HS, Petrelli NJ, O’Connell MJ, Colangelo LH, Smith RE et al (2006) Oral uracil and tegafur plus leucovorin compared with intravenous fluorouracil and leucovorin in stage II and III carcinoma of the colon: results from National Surgical Adjuvant Breast and Bowel Project Protocol C-06. J Clin Oncol 24:2059–2064. doi:10.1200/JCO.2005.04.7498
Littell RC, Pendergast J, Natarajan R (2000) Modelling covariance structure in the analysis of repeated measures data. Stat Med 19:1793–1819. doi:10.1002/1097-0258(20000715)19:13<1793::AID-SIM482>3.0.CO;2-Q
National Cancer Institute (2003) Common terminology criteria for adverse events v3.0 (CTC). National Cancer Institute
Navarro RP, Morrow T, Baran R (2002) Pharmacoeconomic and clinical outcomes in oncology using oral chemotherapy. Manag Care Interface 15:55–62
Neugarten BL, Havighurst RJ, Tobin SS (1961) The measurement of life satisfaction. J Gerontol 16:134–143
Portenoy RK, Thaler HT, Kornblith AB, Lepore JM, Friedlander-Klar H, Kiyasu E et al (1994) The memorial symptom assessment scale: an instrument for the evaluation of symptom prevalence, characteristics and distress. Eur J Cancer 30A:1326–1336. doi:10.1016/0959-8049(94)90182-1
Portenoy RK, Thaler HT, Kornblith AB, Lepore JM, Friedlander-Klar H, Coyle N et al (1994) Symptom prevalence, characteristics and distress in a cancer population. Qual Life Res 3:183–189. doi:10.1007/BF00435383
Roche Laboratories XELODA® (capecitabine) Tablets [package insert]. Roche Laboratories, Nutley, New Jersey, USA
Seidman AD, O’Shaughnessy J, Misset JL (2002) Single-agent capecitabine: a reference treatment for taxane-pretreated metastatic breast cancer? Oncologist 7(Suppl 6):20–28. doi:10.1634/theoncologist.7-suppl_6-20
Smorenburg CH, Bontenbal M, Verweij J (2001) Capecitabine in breast cancer: current status. Clin Breast Cancer 4:288–293. doi:10.3816/CBC.2001.n.003
Tubiana-Hulin M (2005) How to maximize the efficacy of taxanes in breast cancer. Cancer Treat Rev 31(Suppl 4):S3–S9. doi:10.1016/S0305-7372(05)80002-7
Verma S, Clemons M (2007) First-line treatment options for patients with HER-2 negative metastatic breast cancer: the impact of modern adjuvant chemotherapy. Oncologist 12:785–797. doi:10.1634/theoncologist.12-7-785
Walko CM, Lindley C (2005) Capecitabine: a review. Clin Ther 27:23–44. doi:10.1016/j.clinthera.2005.01.005
Ware JE, Snow KK, Kosinski M, Gandek B (1993) SF-36 health survey: manual and interpretation guide. The Health Institute New England Medical Center, Boston
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported in part by funding from Roche.
Rights and permissions
About this article
Cite this article
Schwartzberg, L.S., Cobb, P., Walker, M.S. et al. Patient and practice impact of capecitabine compared to taxanes in first-/second-line chemotherapy for metastatic breast cancer. Support Care Cancer 17, 1081–1088 (2009). https://doi.org/10.1007/s00520-008-0558-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-008-0558-2