Abstract
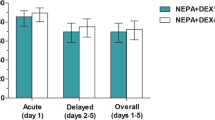
The prevention of post-chemotherapy symptoms such as delayed emesis, anorexia, and fatigue induced by irinotecan has not been studied. We compared the effects of dexamethasone (Dex) with those of a placebo on these symptoms in a randomized study. Seventy patients scheduled to receive irinotecan chemotherapy were enrolled in the study and randomly divided into a treatment or a placebo group. In the treatment group, 8 mg of Dex were administered on days 2–4 after the start of chemotherapy. All patients in both groups received Dex and granisetron for prophylaxis against acute emesis on day 1. We evaluated 68 patients (35 receiving Dex, 33 receiving the placebo). Although delayed emesis was completely prevented in most of patients in both groups (Dex, 82.9%; placebo, 78.8%), anorexia and fatigue were more completely prevented in those in the Dex group (Dex, 62.9% and 77.1%, placebo, 39.4% and 57.6%, respectively). The effect of Dex on improving simultaneous prophylaxis against all three symptoms was almost significant (Dex, 60.0%; placebo, 36.4%; P=0.058). The safety profiles of the two groups were not discernibly different. These results suggest that treatment with Dex may be beneficial to reduce post-chemotherapy symptoms induced by irinotecan, specifically anorexia and fatigue, with acceptable toxicities.

Similar content being viewed by others
Reference
Barbounis V, Koumakis G, Vassilomanolakis M, et al (2001) Control of irinotecan-induced diarrhea by octreotide after loperamide failure. Support Care Cancer 9:258–260
Boku N, Ohtsu A, Shimada Y, et al (1999) Phase II study of a combination of irinotecan and cisplatin against metastatic gastric cancer. J Clin Oncol 17:319–323
Burstein HJ, Manola J, Younger J, et al (2000) Docetaxel administration on a weekly basis for metastatic breast cancer. J Clin Oncol 18:1212–1219
Cunningham D, Pyrhšnen S, James RD, et al (1998) Randomized trial of irinotecan plus supportive care versus supportive care alone after fluorouracil failure for patients with metastatic colorectal cancer. Lancet 352:1413–1418
Gralla RJ, Osoba D, Kris MG, et al (1999) Recommendations for the Use of Antiemetics: Evidense-Based, Clinical Practice Guidelines. J Clin Oncol 17:2971–2994
Green MR, Harper M, Safa A, et al (2000) Irinotecan in the management of patients with pancreatic cancer. Oncology (Suppl 14):31–33
Hardy JR, Rees E, Ling J, et al (2001) A prospective survey of the use of dexamethasone on a palliative care unit. Palliat Med 15:3–8
Inoue A, Saijo N (2001) Recent advances in the chemotherapy of non-small cell lung cancer. Jpn J Clin Oncol 31:299–304
Kehrer DF, Sparreboom A, Verweij J, et al (2001) Modulation of irinotecan-induced diarrhea by cotreatment with neomycin in cancer patients. Clin Cancer Res 7:1136–1141
Noda K, Nishiwaki Y, Kawahara M, et al (2002) Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 346:85–91
Portenoy RK, Itri LM (1999) Cancer-related fatigue: guidelines for evaluation and management. Oncologist 4:1–10
Rougier P, Van Cutsem E, Bajetta E, et al (1998) Randomised trial of irinotecan versus fluorouracil by continuous infusion after fluorouracil failure in patients with metastatic colorectal cancer. Lancet 352:1407–1412
Saltz LB, Cox JV, Blanke C, et al (2000) Irinotecan plus fluorouracil and leucovorin for metastatic colorectal cancer. N Engl J Med 343:905–914
Seidman AD, Hudis CA, Albanel J, et al (1998) Dose-dense therapy with weekly 1-hour paclitaxel infusions in the treatment of metastatic breast cancer. J Clin Oncol 16:3353–3361
Stone P, Richards M, Hardy J (1998) Fatigue in patients with cancer. Eur J Cancer 34:1670–1676
Takeda Y, Kobayashi K, Akiyama Y, et al (2001) Prevention of irinotecan (CPT-11)-induced diarrhea by oral alkalization combined with control of defecation in cancer patients. Int J Cancer 92:269–275
The Italian group for antiemetic research (1995) Dexamethasone, granisetron, or both for the prevention of nausea and vomiting during chemotherapy for cancer. N Engl J Med 332:1–5
The Italian group for antiemetic research (1997) Ondansetron versus metoclopramide, both combined with dexamethasone, in the prevention of cisplatin-induced delayed emesis. J Clin Oncol 15:124–130
The Italian group for antiemetic research (2000) Dexamethasone alone or in combination with ondansetron for the prevention of delayed nausea and vomiting induced by chemotherapy. N Engl J Med 342:1554–1559
Yamada Y, Shirao K, Hyodo I, et al (2003) Phase II study of irinotecan plus mitomycin C in patients with fluoropyrimidine-resistant advanced colorectal cancer. Ann Oncol 13 (Suppl 5):86
Organization for pharmaceutical safety and research. Drug information of irinotecan (in Japanese). http://www.pharmasys.gr.jp/downfiles/PDF/400014_4240404A1032_1_06.pdf. Cited 17 Mar 2003)
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported in part by Grants-in-aid for Cancer Research from the Ministry of Health and Welfare and from the Second-term Comprehensive 10-year Strategy for Cancer Control.
Rights and permissions
About this article
Cite this article
Inoue, A., Yamada, Y., Matsumura, Y. et al. Randomized study of dexamethasone treatment for delayed emesis, anorexia and fatigue induced by irinotecan. Support Care Cancer 11, 528–532 (2003). https://doi.org/10.1007/s00520-003-0488-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-003-0488-y