Abstract
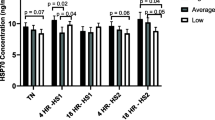
Adaptation is a relevant characteristic to be understood in livestock animals in order to maintain and raise productivity. In Brazil, the Nellore beef cattle are widely disseminated and well-adapted breed that present good thermoregulatory characteristics for tropical environment conditions. Conversely, the physiological and cellular mechanisms required for thermoregulation and thermotolerance in this breed are still limited. The aim of this study was to comprehend the heat loss efficiency at the whole animal level and heat shock response at the cellular level of Nellore cows in tropical climate conditions. Healthy purebred Nellore cows were classified according to their capacity to lose body heat as Efficient or Inefficient based on vaginal temperature which was continuously monitored by data-loggers. Rectal, tail, and ocular temperatures, sweating rate, and respiratory frequency were collected to assess other thermoregulatory responses. Peripheral mononuclear cells were used for gene expression of heat shock proteins 60, 70, and 90 induced by in vitro heat treatments at 38, 40, and 42 °C. In our findings, the Efficient cows presented higher sweating rates compared to Inefficient cows that presented higher rectal temperature with greater amplitude of vaginal temperature profile. Transcription of the HSP genes was stable at 38 and 40 °C and decreased for all HSP genes at 42 °C. In conclusion, the Nellore efficiency to lose heat was mainly associated with their sweating capacity and cellular thermotolerance confirmed by the maintenance of heat shock proteins transcripts under heat stress. Taken together, this knowledge contributes as a future key for genetic selection of adapted animals.


Similar content being viewed by others
Abbreviations
- cDNA:
-
Complementary DNA
- EF:
-
Efficient cows
- FZEA:
-
Faculty of Animal Science and Food Engineering
- HSPs:
-
Heat shock proteins
- NEF:
-
Inefficient cows
- IRT:
-
Infrared thermography trait
- L×C×W:
-
Length × circumference × weight
- m:
-
Meter
- PBMCs:
-
Peripheral blood mononuclear cells
- qPCR:
-
Quantitative polymerase chain reaction
- RBCs:
-
Red blood cells
- RF:
-
Respiratory frequency
- RH:
-
Relative humidity
- RT-qPCR:
-
Reverse transcription
- SR:
-
Sweating rate
- T a :
-
Air temperature
- T bg :
-
Black globe temperature
- T dp :
-
Dew point temperature
- THI:
-
Temperature humidity index
- T oc :
-
Ocular temperature
- T r :
-
Rectal temperature
- T t :
-
Tail temperature
- T vag :
-
Vaginal temperature
References
Agnew LL, Colditz IG (2008) Development of a method of measuring cellular stress in cattle and sheep. Vet Immunol Immunopathol 123:197–204. https://doi.org/10.1016/j.vetimm.2008.01.038
Alzahal O, Alzahal H, Steele MA et al (2011) The use of a radiotelemetric ruminal bolus to detect body temperature changes in lactating dairy cattle. J Dairy Sci 94:3568–3574. https://doi.org/10.3168/jds.2010-3944
Bañuelos-Valenzuela R, Sánchez-Rodríguez SH (2005) La proteína de estrés calórico Hsp70 funciona como un indicador de adaptación de los bovinos a las zonas áridas. Redvet VI:1–18
Baraja-Vásquez GE, Baldwin-Sevilla C, Barbosa Cisneros OY, Sánchez-Rodríguez SH (2005) Las proteínas de estrés calórico Hsp60, 70 y 90 participan en la adaptación de los caprinos a las zonas áridas (heat shock proteins 60, 70 and 90 participate in the adaptation of goats to the arid zones). Redvet VI:3–16
Barbosa BRP, Santos SA, UGP A, Egito AA, Comastri Filho JA, Juliano RS, Paiva SR, McManus C (2014) Tolerância ao calor em bovinos das raças Nelore branco, Nelore vermelho e Pantaneira. Rev Bras Saúde Prod Anim 15:854–865
Bettegowda A, Patel OV, Ireland JJ, Smith GW (2006) Quantitative analysis of messenger RNA abundance for ribosomal protein L-15, cyclophilin-A, phosphoglycerokinase, β-glucuronidase, glyceraldehyde 3-phosphate dehydrogenase, β-actin, and histone H2A during bovine oocyte maturation and early embryogenesis. Mol Reprod Dev 73:267–278. https://doi.org/10.1002/mrd.20333
Burdick NC, Carroll JA, Dailey JW, Randel RD, Falkenberg SM, Schmidt TB (2012) Development of a self-contained, indwelling vaginal temperature probe for use in cattle research. J Therm Biol 37:339–343. https://doi.org/10.1016/j.jtherbio.2011.10.007
Carvalho ME, Gasparin G, Poleti MD, Rosa AF, Balieiro JCC, Labate CA, Nassu RT, Tullio RR, Regitano LCA, Mourão GB, Coutinho LL (2014) Heat shock and structural proteins associated with meat tenderness in Nellore beef cattle, a Bos indicus breed. Meat Sci 96:1318–1324. https://doi.org/10.1016/j.meatsci.2013.11.014
Cheung SS, McLellan TM (1998) Heat acclimation, aerobic fitness, and hydration effects on tolerance during uncompensable heat stress. J Appl Physiol 84:1731–1739
Church JS, Cook NJ, Schaefer AL (2009) Recent applications of infrared thermography for animal welfare and veterinary research: everything from chicks to elephants. In: InfraMation Proceedings. p 10
Deb R, Sajjanar B, Singh U, Kumar S, Singh R, Sengar G, Sharma A (2013) Effect of heat stress on the expression profile of Hsp90 among Sahiwal (Bos indicus) and Frieswal (Bos indicus × Bos taurus) breed of cattle: a comparative study. Gene 536:435–440. https://doi.org/10.1016/j.gene.2013.11.086
Dobson H, Smith RF (2000) What is stress, and how does it affect reproduction? Anim Reprod Sci 60–61:743–752
Domiciano LF, Mombach MA, Carvalho P, da Silva NMF, Pereira DH, Cabral LS, Lopes LB, Pedreira BC (2018) Performance and behaviour of Nellore steers on integrated systems. Anim Prod Sci 58:920. https://doi.org/10.1071/AN16351
Finch VA (1985) Body temperature in beef cattle: its control and relevance to production in the tropics. J Anim Sci 62:531–542. https://doi.org/10.2134/jas1986.622531x
Finch VA, Bennett IL, Holmes CR (1984) Coat colour in cattle: effect on thermal balance, behaviour and growth, and relationship with coat type. J Agric Sci 102:141. https://doi.org/10.1017/S0021859600041575
Fisher AD, Morton R, Dempsey JMA, Henshall JM, Hill JR (2008) Evaluation of a new approach for the estimation of the time of the LH surge in dairy cows using vaginal temperature and electrodeless conductivity measurements. Theriogenology 70:1065–1074. https://doi.org/10.1016/j.theriogenology.2008.06.023
Gehring WJ, Wehner R (1995) Heat shock protein synthesis and thermotolerance in Cataglyphis, an ant from the Sahara desert. Proc Natl Acad Sci U S A 92:2994–2998
Hahn GL (1995) Environmental management for improved livestock performance, health and well-being. Japanese J Livest Manag 30:113–127. https://doi.org/10.20652/kachikukanri.30.3_113
Hahn GL (1999) Dynamic responses of cattle to thermal heat loads. J Anim Sci 77(Suppl 2):10–20
Hales JR, a a F, Bennett JW, Needham a D (1978) Thermal control of blood flow through capillaries and arteriovenous anastomoses in skin of sheep. Pflugers Arch 378:55–63. https://doi.org/10.1007/BF00581958
Hansen PJ (2004) Physiological and cellular adaptations of zebu cattle to thermal stress. Anim Reprod Sci 82–83:349–360. https://doi.org/10.1016/j.anireprosci.2004.04.011
Hillman PE, Gebremedhin KG, Willard ST et al (2009) Continuous measurements of vaginal temperature of female cattle using a data logger encased in a plastic anchor. Appl Eng Agric 25:291–296
Hooper HB, Gonella-Diaza ÂM, Binelli M, et al (2017a) Heat shock response in PBMCs of Nellore cows after in vitro thermal stress. In: VII Brazilian Congress of Biometeorology, Ambience, Behaviour and Animal Welfare (Conference proceeding)
Hooper HB, Henrique FL, Rodríguez LFP, et al (2017b) Heat loss efficiency and thermoregulation responses of Nellore cows. In: VII Brazilian Congress of Biometeorology, Ambience, Behaviour and Animal Welfare (Conference proceeding)
Hutchinson JC, Brown GD (1969) Penetrance of cattle coats by radiation. J Appl Physiol 26:454–464
Jessen C (2001) Temperature regulation in human and other mammals, 1st edn. Springer, Verlag, Berlin
Kastberger G, Stachl R (2003) Infrared imaging technology and biological applications. Behav Res Methods Instrum Comput 35:429–439
Kelly CF, Bond TE (1971) Bioclimatic factors and their measurement. Natl Acad Sci A Guid Environ Res Anim 123
Kishore A, Sodhi M, Kumari P, Mohanty AK, Sadana DK, Kapila N, Khate K, Shandilya U, Kataria RS, Mukesh M (2013) Peripheral blood mononuclear cells: a potential cellular system to understand differential heat shock response across native cattle (Bos indicus), exotic cattle (Bos taurus), and riverine buffaloes (Bubalus bubalis) of India. Cell Stress Chaperones 19:613–621. https://doi.org/10.1007/s12192-013-0486-z
Kolb E, Ketz AHG (1987) Fisiologia Veterinária, 4a. Rio de Janeiro
Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) World Map of the Köppen-Geiger climate classification updated. Meteorol Zeitschrift 15:259–263. https://doi.org/10.1127/0941-2948/2006/0130
Kumar A, Ashraf S, Goud TS, Grewal A, Singh SV, Yadav BR, Upadhyay RC (2015) Expression profiling of major heat shock protein genes during different seasons in cattle (Bos indicus) and buffalo (Bubalus bubalis) under tropical climatic condition. J Therm Biol 51:55–64. https://doi.org/10.1016/j.jtherbio.2015.03.006
Lepock JR (2005) How do cells respond to their thermal environment? Int J Hyperth 21:681–687. https://doi.org/10.1080/02656730500307298
Lyashko VN, Vikulova VK, Chernicov VG, Ivanov VI, Ulmasov KA, Zatsepina OG, Evgen’ev MB (1994) Comparison of the heat shock response in ethnically and ecologically different human populations. Proc Natl Acad Sci U S A 91:12492–12495
Mader TL, Davis MS, Brown-Brandl T (2006) Environmental factors influencing heat stress in feedlot cattle. J Anim Sci 84:712–719
Martello LS, da Luz e Silva S, da Costa Gomes R, da Luz e Silva S, da Costa Gomes R, da Silva Corte RRP, Leme PR (2016) Infrared thermography as a tool to evaluate body surface temperature and its relationship with feed efficiency in Bos indicus cattle in tropical conditions. Int J Biometeorol 60:173–181. https://doi.org/10.1007/s00484-015-1015-9
Mason IL, World Dictionary of Livestock Breeds (1988) Third Edition. C.A.B International
McClung JP, Hasday JD, He J-R et al (2008) Exercise-heat acclimation in humans alters baseline levels and ex vivo heat inducibility of HSP72 and HSP90 in peripheral blood mononuclear cells. Am J Physiol Regul Integr Comp Physiol 294:R185–R191. https://doi.org/10.1152/ajpregu.00532.2007
McManus C, Prescott E, Paludo GR, Bianchini E, Louvandini H, Mariante AS (2009) Heat tolerance in naturalized Brazilian cattle breeds. Livest Sci 120:256–264. https://doi.org/10.1016/j.livsci.2008.07.014
Mcmanus CM, Louvandini H, Paim TP, Silva FCP, Bernal FEM (2014) Factors affecting heat tolerance in crossbred cattle in central Brazil. Ciência Anim Bras 15:152–158. https://doi.org/10.1590/1809-6891v15i28726
Mishra A, Hooda OK, Singh G, Meur SK (2011) Influence of induced heat stress on HSP70 in buffalo lymphocytes. J Anim Physiol Anim Nutr (Berl) 95:540–544. https://doi.org/10.1111/j.1439-0396.2010.01082.x
Mohanarao GJ, Mukherjee A, Banerjee D et al (2014) HSP70 family genes and HSP27 expression in response to heat and cold stress in vitro in peripheral blood mononuclear cells of goat (Capra hircus). Small Rumin Res 116:94–99. https://doi.org/10.1016/j.smallrumres.2013.10.014
Montanholi YR, Odongo NE, Swanson KC, Schenkel FS, McBride BW, Miller SP (2008) Application of infrared thermography as an indicator of heat and methane production and its use in the study of skin temperature in response to physiological events in dairy cattle (Bos taurus). J Therm Biol 33:468–475. https://doi.org/10.1016/j.jtherbio.2008.09.001
Montanholi YR, Swanson KC, Schenkel FS, McBride BW, Caldwell TR, Miller SP (2009) On the determination of residual feed intake and associations of infrared thermography with efficiency and ultrasound traits in beef bulls. Livest Sci 125:22–30. https://doi.org/10.1016/j.livsci.2009.02.022
Moseley PL (1997) Invited review: heat shock proteins and heat adaptation of the whole organism. J Appl Physiol 83:1413–1417
Navarini FC, Klosowski ES, Campos AT, Teixeira RA, Almeida CP (2009) Conforto térmico de bovinos da raça nelore a pasto sob diferentes condições de sombreamento e a pleno sol. Eng Agríc [online] 29:508–517. https://doi.org/10.1590/S0100-69162009000400001
Nay T, Hayman R (1956) Sweat glands in Zebu (Bos indicus L.) and European ( B. taurus L.) cattle. I. Size of individual glands, the denseness of their population, and their depth below the skin surface. Aust J Agric Res 7:482. https://doi.org/10.1071/AR9560482
Pratt WB, Morishima Y, Peng H-M, Osawa Y (2010) Proposal for a role of the Hsp90/Hsp70-based chaperone machinery in making triage decisions when proteins undergo oxidative and toxic damage. Exp Biol Med 235:278–289. https://doi.org/10.1258/ebm.2009.009250
Reuter RR, Carroll JA, Hulbert LE, Dailey JW, Galyean ML (2010) Technical note: development of a self-contained, indwelling rectal temperature probe for cattle research. J Anim Sci 88:3291–3295. https://doi.org/10.2527/jas.2010-3093
Silva RG (2000) Introdução à bioclimatologia animal. Ed. Nobel, São Paulo
Sonna LA, Fujita J, Gaffin SL, Lilly CM (2002) Invited review: effects of heat and cold stress on mammalian gene expression. J Appl Physiol 92:1725–1742. https://doi.org/10.1152/japplphysiol.01143.2001
Thom EC (1958) Measuring the need for air conditioning. Air Cond Heat Vent 53:68–70
Titto EAL (1999) Estudo da tolerância ao calor em tourinhos das raças Marchigiana, Nelore e Simental. In: Congresso de Zooecnia. Porto, Portugal,
Titto CG, Titto EAL, Titto RM, Mourão GB (2011) Heat tolerance and the effects of shade on the behavior of Simmental bulls on pasture. Anim Sci J 82:591–600. https://doi.org/10.1111/j.1740-0929.2011.00872.x
Ulmasov KA, Shammakov S, Karaev K, Evgen’ev MB (1992) Heat shock proteins and thermoresistance in lizards. Proc Natl Acad Sci U S A 89:1666–1670
Vandesompele J, De Preter K, Pattyn F et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:34
Wang S, Diller KR, Aggarwal SJ (2003) Kinetics study of endogenous heat shock protein 70 expression. J Biomech Eng 125:794–797. https://doi.org/10.1115/1.1632522
West JW, Mullinix BG, Bernard JK (2003) Effects of hot, humid weather on milk temperature, dry matter intake, and milk yield of lactating dairy cows. J Dairy Sci 86:232–242. https://doi.org/10.3168/jds.S0022-0302(03)73602-9
Acknowledgements
This research was supported by São Paulo Research Foundation, no. 2011/09282-3. We would also like to show our gratitude to Prof. Ed Hoffman Madureira from University of São Paulo, who provided insight and expertise that greatly assisted the research. This article contains parts derived from two conference abstract presented in the VII Brazilian Congress of Biometeorology, Ambience, Behavior and Animal Welfare and are stated in Figshare proceedings (DOIs: https://doi.org/10.6084/m9.figshare.5176753; https://doi.org/10.6084/m9.figshare.5176756).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
All procedures performed in studies involving animals were in accordance with the current Federal Law no. 11.794, sanctioned by the President of the Republic on November 8, 2008. The law is available in full at http://planalto.gov.br/ccivil_03/_Ato2007-2010/2008/Lei/L11794.htm, and is known as the Arouca Law, which regulates the scientific use of animals in Brazil. The local committee certifies the protocol number CEUA 13.1.2186.74.2, which involves the production, maintenance and/or use of animals belonging to the phylum Chordata, subphylum Vertebrata (except human beings), for scientific research purposes (or teaching)—it is in accordance with Law 11.794, of October 8, 2008, Decree 6899, of July 15, 2009, with the rules issued by the National Council for Control of Animal Experimentation (CONCEA), and was approved by the Ethics Committee on Animal Use of the School of Animal Science and Food Engineering of São Paulo University.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. The animals used in the experiment had prior approval of the Ethics Faculty Committee, receiving the protocol number no. 13.1.2186.74.2.
Rights and permissions
About this article
Cite this article
Hooper, H.B., Titto, C.G., Gonella-Diaza, A.M. et al. Heat loss efficiency and HSPs gene expression of Nellore cows in tropical climate conditions. Int J Biometeorol 63, 1475–1486 (2019). https://doi.org/10.1007/s00484-018-1576-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-018-1576-5