Abstract
Key message
Combined application of ethylene and jasmonate is a strong signal that leads to an increased magnitude of defense induction in two Cupressaceae species.
Abstract
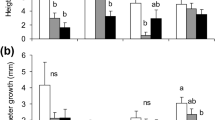
Ethrel (Et), methyl jasmonate (MJ), sodium salicylate (NS), and combinations of these compounds were applied to unwounded or wounded stems of Thujopsis dolabrata cuttings and Metasequoia glyptostroboides seedlings to clarify the mechanism of anatomical defense responses in Cupressaceae species. The application of MJ clearly induced the formation of abundant traumatic resin ducts (TRDs) in T. dolabrata, whereas the application of Et in M. glyptostroboides induced TRD formation rather than MJ. In both T. dolabrata and M. glyptostroboides, Et + MJ application induced massive tangential TRDs in the phloem or xylem. NS application did not significantly promote TRD formation in either species. Additionally, the application of NS with Et decreased resin duct formation in M. glyptostroboides compared with single Et application. These results indicate that Et and MJ application is a strong signal that leads to an increased magnitude of defense induction but no difference in species responsivity. Wounding increased TRD formation in the bark of T. dolabrata cuttings over that in unwounded cuttings under all treatments, whereas wounding in M. glyptostroboides showed little effect. These differences in anatomical responses may imply the strong constitutive defenses function by bark of T. dolabrata.




Similar content being viewed by others
Data availability
All experimental data and materials will be made available to third-party academic researchers upon reasonable request.
Code availability
Not applicable.
References
Alfaro RI (1995) An induced defense reaction in white spruce to attack by the white pine weevil, Pissodes strobi. Can J for Res 25:1725–1730. https://doi.org/10.1139/x95-186
Aloni R (2021) Vascular differentiation and plant hormones. Springer Nature, Switzerland, AG
Arango-Velez A, El Kayal W, Copeland CCJ et al (2016) Differences in defence responses of Pinus contorta and Pinus banksiana to the mountain pine beetle fungal associate Grosmannia clavigera are affected by water deficit. Plant Cell Environ 39:726–744. https://doi.org/10.1111/pce.12615
Berens ML, Berry HM, Mine A et al (2017) Evolution of hormone signaling networks in plant defense. Annu Rev Phytopathol 55:401–425. https://doi.org/10.1146/annurev-phyto-080516-035544
Brignolas F, Lieutier F, Sauvard D et al (1995) Changes in soluble-phenol content of Norway-spruce (Picea abies) phloem in response to wounding and inoculation with Ophiostoma polonicum. Eur J for Pathol 25:253–265. https://doi.org/10.1111/j.1439-0329.1995.tb01010.x
Cooper RJ (2003) World markets for coniferous forest products: recent trends and future prospects. Acta Horticulturae. International Society for Horticultural Science, pp 349–353
Danti R, Della RG (2017) Epidemiological history of cypress canker disease in source and invasion sites. Forests. https://doi.org/10.3390/f8040121
Drenkhan R, Ganley B, Martín-García J et al (2020) Global geographic distribution and host range of fusarium circinatum, the causal agent of pine pitch canker. Forests 11:1–40. https://doi.org/10.3390/F11070724
Evert RF, Eichhorn SE (2006) Esau’s plant anatomy: meristems, cells, and tissues of the plant body, 3rd edn. John Wiley & Sons
Eyles A, Bonello P, Ganley R, Mohammed C (2010) Induced resistance to pests and pathogens in trees. New Phytol 185:893–908
Fäldt J, Martin D, Miller B et al (2003) Traumatic resin defense in Norway spruce (Picea abies): methyl jasmonate-induced terpene synthase gene expression, and cDNA cloning and functional characterization of (+)-3-carene synthase. Plant Mol Biol 51:119–133. https://doi.org/10.1023/A:1020714403780
Farjon A (2018) The Kew review conifers of the world. Kew Bull 73:1–16. https://doi.org/10.1007/s12225-018-9738-5
Felicijan M, Kristl J, Krajnc AU (2016) Pre-treatment with salicylic acid induces phenolic responses of Norway spruce (Picea abies) bark to bark beetle (Ips typographus) attack. Trees - Struct Funct 30:2117–2129. https://doi.org/10.1007/s00468-016-1438-x
Franceschi VR, Krekling T, Christiansen E (2002) Application of methyl jasmonate on Picea abies (Pinaceae) stems induces defense-related responses in phloem and xylem. Am J Bot 89:578–586. https://doi.org/10.3732/ajb.89.4.578
Gartner H, Heinrich I (2009) The formation of traumatic rows of resin ducts in Larix decidua and Picea abies (Pinaceae) as a result of wounding experiments in the dormant season. IAWA J 30:199–215. https://doi.org/10.1163/22941932-90000215
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227. https://doi.org/10.1146/annurev.phyto.43.040204.135923
Hudgins JW, Franceschi VR (2004) Methyl jasmonate-induced ethylene production is responsible for conifer phloem defense responses and reprogramming of stem cambial zone for traumatic resin duct formation. Plant Physiol 135:2134–2149. https://doi.org/10.1104/pp.103.037929
Hudgins JW, Christiansen E, Franceschi VR (2003) Methyl jasmonate induces changes mimicking anatomical defenses in diverse members of the Pinaceae. Tree Physiol 23:361–371. https://doi.org/10.1093/treephys/23.6.361
Hudgins JW, Christiansen E, Franceschi VR (2004) Induction of anatomically based defense responses in stems of diverse conifers by methyl jasmonate: a phylogenetic perspective. Tree Physiol 24:251–264. https://doi.org/10.1093/treephys/24.3.251
Kozlowski G, Buchala A, Métraux JP et al (1999) Methyl jasmonate protects Norway spruce [Picea abies (L.) Karst.] Seedlings against Pythium ultimum Trow. Physiol Mol Plant Pathol 55:53–58. https://doi.org/10.1006/pmpp.1999.0205
Krajnc AU, Kristl J, Ivancic A (2011) Application of salicylic acid induces antioxidant defense responses in the phloem of Picea abies and inhibits colonization by Ips typographus. For Ecol Manag 261:416–426. https://doi.org/10.1016/j.foreco.2010.10.027
Krokene P (2015) Conifer defense and resistance to bark beetles. Bark beetles. Elsevier, pp 177–207
Krokene P, Nagy NE (2008) Traumatic resin ducts and polyphenolic parenchyma cells in conifers. Induced plant resistance to herbivory. Springer, Netherlands, pp 147–169
Kuroda K, Shimaji K (1983) Traumatic resin canal formation as a marker of xylem growth. For Sci 29:653–659. https://doi.org/10.1093/forestscience/29.3.653
Kusumoto D, Suzuki K (2001) Induction of traumatic resin canals in Cupressaceae by ethrel application. Mokuzaigakkaishi 47:1–6
Lawton KA, Potter SL, Uknes S, Ryals J (1994) Acquired resistance signal transduction in Arabidopsis is ethylene independent. Plant Cell 6:581–588. https://doi.org/10.1105/tpc.6.5.581
León J, Rojo E, Sánchez-Serrano JJ (2001) Wound signalling in plants. J Exp Bot 52:1–9. https://doi.org/10.1093/jexbot/52.354.1
López-Villamor A, Zas R, Pérez A et al (2021) Traumatic resin ducts induced by methyl jasmonate in Pinus spp. Trees - Struct Funct 35:557–567. https://doi.org/10.1007/s00468-020-02057-9
Matsumoto K, Chun JP, Takemura Y et al (2009) Gum accumulation of Japanese apricot fruit (Prunus mume Sieb. et Zucc.) was expedited by Jasmonate. J Jpn Soc Hortic Sci 78:273–278. https://doi.org/10.2503/jjshs1.78.273
Mayama S, Tosa Y (2020) Plant pathology, 2nd edn. Bun’eido Publishing (in Japanese)
McKay SAB, Hunter WL, Godard KA et al (2003) Insect attack and wounding induce traumatic resin duct development and gene expression of (-)-pinene synthase in Sitka spruce. Plant Physiol 133:368–378. https://doi.org/10.1104/pp.103.022723
Morris H, Hietala AM, Jansen S et al (2020) Using the CODIT model to explain secondary metabolites of xylem in defence systems of temperate trees against decay fungi. Ann Bot 125:701–720
Moungsrimuangdee B, Moriwaki H, Nakayama M et al (2011) Effects of injection of ethrel, methyl jasmonate, and salicylates and Raffaelea quercivora inoculation on sapwood discoloration in Quercus serrata. IAWA J 32:41–53. https://doi.org/10.1163/22941932-90000041
Nagy NE, Franceschi VR, Solheim H et al (2000) Wound-induced traumatic resin duct development in stems of Norway spruce (Pinaceae): anatomy and cytochemical traits. Am J Bot 87:302–313. https://doi.org/10.2307/2656626
Oven P, Torelli N (1994) Wound response of the bark in healthy and declining silver firs (Abies alba). IAWA J 15:407–415. https://doi.org/10.1163/22941932-90001375
Pallardy S (2008) Physiology of Woody Plants, 3rd edn. Academic Press
Penninckx IAMA, Eggermont K, Terras FRG et al (1996) Pathogen-induced systemic activation of a plant defensin gene in Arabidopsis follows a salicylic acid-independent pathway. Plant Cell 8:2309–2323. https://doi.org/10.1105/tpc.8.12.2309
Phillips MA, Croteau RB (1999) Resin-based defenses in conifers. Trends Plant Sci 4:184–190
Rodrigues KCS, Fett-Neto AG (2009) Oleoresin yield of Pinus elliottii in a subtropical climate: seasonal variation and effect of auxin and salicylic acid-based stimulant paste. Ind Crops Prod 30:316–320. https://doi.org/10.1016/j.indcrop.2009.06.004
Rodríguez-García A, Martín JA, López R et al (2016) Effect of four tapping methods on anatomical traits and resin yield in maritime pine (Pinus pinaster Ait.). Ind Crops Prod 86:143–154. https://doi.org/10.1016/j.indcrop.2016.03.033
Ruan J, Zhou Y, Zhou M et al (2019) Jasmonic acid signaling pathway in plants. Int J Mol Sci. https://doi.org/10.3390/ijms20102479
Schaller A (2008) Induced plant resistance to herbivory. Springer, Dordrecht
Schlink K (2011) Gene expression profiling in wounded and systemic leaves of Fagus sylvatica reveals up-regulation of ethylene and jasmonic acid signalling. Plant Biol 13:445–452. https://doi.org/10.1111/j.1438-8677.2010.00397.x
Schmidt A, Nagel R, Krekling T et al (2011) Induction of isoprenyl diphosphate synthases, plant hormones and defense signalling genes correlates with traumatic resin duct formation in Norway spruce (Picea abies). Plant Mol Biol 77:577–590. https://doi.org/10.1007/s11103-011-9832-7
Sharan M, Taguchi G, Gonda K et al (1998) Effects of methyl jasmonate and elicitor on the activation of phenylalanine ammonia-lyase and the accumulation of scopoletin and scopolin in tobacco cell cultures. Plant Sci 132:13–19. https://doi.org/10.1016/S0168-9452(97)00260-4
Telewski FW, Wakefield AH, Jaffe MJ (1983) Computer-assisted image analysis of tissues of ethrel-treated Pinus taeda seedlings. Plant Physiol 72:177–181. https://doi.org/10.1104/pp.72.1.177
Thaler JS, Humphrey PT, Whiteman NK (2012) Evolution of jasmonate and salicylate signal crosstalk. Trends Plant Sci 17:260–270. https://doi.org/10.1016/j.tplants.2012.02.010
Wainhouse D, Rose DR, Peace AJ (1997) The influence of preformed defences on the dynamic wound response in Spruce bark. Funct Ecol 11:564–572. https://doi.org/10.1046/j.1365-2435.1997.00125.x
Waterman JM, Cazzonelli CI, Hartley SE, Johnson SN (2019) Simulated herbivory: the key to disentangling plant defence responses. Trends Ecol Evol 34:447–458
Xu Y, Chang Pi Fang L, Liu D et al (1994) Plant defense genes are synergistically induced by ethylene and methyl jasmonate. Plant Cell 6:1077–1085. https://doi.org/10.1105/tpc.6.8.1077
Yamamoto F, Kozlowski T (1987) Effects of flooding, tilting of stems, and ethrel application on growth, stem anatomy and ethylene production of Pinus densiflora seedlings. J Exp Bot 38:293–310
Acknowledgements
This study was financially supported by the Joint Research Program of the Arid Land Research Center, Tottori University.
Funding
Partial financial support was received from the Joint Research Program of the Arid Land Research Center, Tottori University, because F Iwanaga, F Yamamoto and N Yamanaka are members of the joint research program of ALRC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest or competing interests.
Ethical approval
Not applicable.
Research involving human participants and/or animals
Not applicable.
Consent to participate
All authors consent to participate.
Consent for publication
All authors consent to publish.
Additional information
Communicated by Holger Gärtner.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Moungsrimuangdee, B., Iwanaga, F., Yamanaka, N. et al. Effects of applied ethrel, jasmonic acid, and salicylic acid on the formation of traumatic resin ducts in the bark of Thujopsis dolabrata cuttings and xylem of Metasequoia glyptostroboides seedlings. Trees 36, 793–801 (2022). https://doi.org/10.1007/s00468-021-02250-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-021-02250-4