Abstract
Key message
Tropical recalcitrant-seeded species, which disperse seeds at different seasons, provide evidence of the role of oxidative metabolism in seed bank dynamics. Our results indicate that CAT may regulate field germination.
Abstract
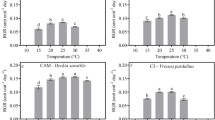
Oxidative metabolism during viability loss in recalcitrant seeds has been previously documented, but little is known about the role of ROS-scavenging enzymes in soil seed bank dynamics, especially in recalcitrant seeds. To address this, recalcitrant-seeded species were studied, Swartzia langsdorffii and Persea wildenovii. Diaspores were kept on the soil after natural dispersion, to simulate the seed bank, and were assessed for seed water content, viability, germination and changes in catalase (CAT), superoxide dismutase (SOD) and peroxidase (PRX) activity throughout the field experiment. These parameters were tested for correlations with climatic data. S. langsdorffii start to germinate after one month in soil, with an increase after two months, when CAT activity decreased and SOD activity increased. SOD was negatively correlated with CAT, and CAT was negatively correlated with germination. However, SOD and CAT were positively correlated for P. wildenovii, whose seeds did not germinate even after four months in the soil and with high activity of ROS-scavenging enzymes throughout the period in the soil. Seeds of these species remained viable during the period in the soil with no or little changes in seed water content. ROS-scavenging enzymes respond to environmental factors, adjusting their activity to maintain seed viability. P. wildenovii seeds did not germinate in the soil due to the timing of dispersal being at the end of rainy season, when additional water supply is limited; ROS-scavenging enzymes probably play a role in seed protection during this dry period. Relationship between CAT regulation and soil seed bank dynamics is discussed.




Similar content being viewed by others
References
Bailly C (2004) Active oxygen species and antioxidants in seed biology. Seed Sci Res 14:93–107
Bailly C, El-Maarouf-Bouteau H, Corbineau F (2008) From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. C R Biol 331:806–814
Berjak P, Pammenter NW (2008) From Avicennia to Zizania: seed recalcitrance in perspective. Ann Bot 101:213–228
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Davide AC, Carvalho LC, Carvalho MLM, Guimarães RM (2003) Classificação fisiológica de sementes de espécies florestais pertencentes à família Lauraceae quanto à capacidade de armazenamento. Cerne 9:29–35
El-Maarouf-Bouteau H, Bailly C (2008) Oxidative signaling in seed germination and dormancy. Plant Signal Behav 3:175–182
EMBRAPA (2006) Sistema Brasileiro de Classificação de Solos, 2nd edn. Embrapa-SPI, Rio de Janeiro
Fath A, Bethke PC, Jones RL (2001) Enzymes that scavenge reactive oxygen species are down-regulated prior to gibberellic acid-induced programmed cell death in barley aleurone. Plant Physiol 126:156–166
Finch-Savage WE, Hendry GAF, Atherton NM (1994) Free radical activity and loss of viability during drying of desiccation-sensitive tree seeds. Proc R Soc Edinb 102:257–260
Finkelstein R, Reeves W, Ariizumi T, Steber C (2008) Molecular aspects of seed dormancy. Ann Rev Plant Biol 59:387–415
Giannopolitis CN, Ries SK (1977) Superoxide dismutases I. Occurrence in higher plants. Plant Physiol 59:309–314
Gidrol X, Lin WS, Dégousée N, Yip SF, Kush A (1994) Accumulation of reactive oxygen species and oxidation of cytokinin in germinating soybean seeds. Eur J Biochem 224:21–28
Gomes MP, Garcia QS (2013) Reactive oxygen species and seed germination. Biologia 68:351–357
Hammerschmidt R, Nuckles EM, Kuc J (1982) Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol Plant Path 20:73–82
Hendricks SB, Taylorson RB (1975) Breaking of seed dormancy by catalase inhibition. Proc Natl Acad Sci 72:306–309
Hendry GAF, Finch-Savage WE, Thorpe PC, Atherton NM, Buckland SM, Nilsson KA, Seel WE (1992) Free radical processes and loss of seed viability during desiccation in the recalcitrant species Quercus robur L. New Phytol 122:273–279
ISTA (2004) International rules for seed testing. The International Seed Testing Association Bassesdorf, Switzerland
Leymarie J, Vitkauskaité G, Hoang HH, Gendreau E, Chazoule V, Meimoun P, Corbineau F, El-Maarouf-Bouteau H, Bailly C (2012) Role of reactive oxygen species in the regulation of Arabidopsis seed dormancy. Plant Cell Physiol 53:96–106
Li C, Sun WQ (1999) Desiccation sensitivity and activities of free radical-scavenging enzymes in recalcitrant Theobroma cacao seeds. Seed Sci Res 9:209–217
Liu Y, Ye N, Liu R, Chen M, Zhang J (2010) H2O2 mediates the regulation of ABA catabolism and GA biosynthesis in Arabidopsis seed dormancy and germination. J Exp Bot 61:2979–2990
Mansano VF, Pinto RB, Torke BM (2016) Swartzia in Lista de Espécies da Flora do Brasil. Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB19178. Accessed 10 July 2016
Marangon LC, Soares JJ, Feliciano ALP (2003) Florística arbórea da mata da pedreira, município de viçosa, Minas Gerais. Rev Árvore 27:207–215
Meinhard M, Rodriguez PL, Grill E (2002) The sensitivity of ABI2 to hydrogen peroxide links the abscisic acid-response regulator to redox signaling. Planta 214:775–782
Moothoo-Padayachie A, Varghese B, Pammenter NW, Govender P, Naidoo S (2016) Germination associated ROS production and glutathione redox capacity in two recalcitrant-seeded species differing in seed longevity. Botany 94:1103–1114
Morohashi Y (2002) Peroxidase activity develops in the micropylar endosperm of tomato seeds prior to radicle protrusion. J Exp Bot 53:1643–1650
Ogawa K, Iwabuchi M (2001) A mechanism for promoting the germination of Zinnia elegans seeds by hydrogen peroxide. Plant Cell Physiol 42:286–291
Oracz K, Karpiński S (2016) Phytohormones signaling pathways and ROS involvement in seed germination. Front Plant Sci 7:864. doi:10.3389/fpls.2016.00864
Oracz K, El-Maarouf-Bouteau H, Farrant JM, Cooper K, Belghazi M, Job C, Job D, Corbineau F, Bailly C (2007) ROS production and protein oxidation as a novel mechanism for seed dormancy alleviation. J Plant 50:452–465
Oracz K, El-Maarouf-Bouteau H, Kranner I, Bogatek R, Corbineau F, Bailly C (2009) The mechanisms involved in seed dormancy alleviation by hydrogen cyanide unravel the role of reactive oxygen species as key factors of cellular signaling during germination. Plant Physiol 150:494–505
Palmiano EP, Juliano BO (1973) Changes in the activity of some hydrolases, peroxidase, and catalase in the rice seed during germination. Plant Physiol 52:274–277
Pammenter NW, Berjak P (1999) A review of recalcitrant seed physiology in relation to desiccation-tolerance mechanisms. Seed Sci Res 9:13–37
Pammenter NW, Berjak P, Farrant JM, Smith MT, Ross G (1994) Why do stored hydrated recalcitrant seeds die? Seed Sci Res 4:187–191
Quinet A, Baitello JB, Moraes PLR, de Assis L, Alves FM (2016) Lauraceae in Lista de Espécies da Flora do Brasil. Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB8523. Accessed 15 Sept 2016
R Development Core Team (2011) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available at http://www.R-project.org. Accessed 22 Aug 2013
Santos N (1979) Fenologia. Rodriguésia 31:223–226
Scandalios JG, Guan L, Polidoros AN (1997) Catalases in plants: gene structure, properties, regulation, and expression. In: Scandalios JG (ed) Oxidative stress and the molecular biology of antioxidant defenses. Cold Spring Harbor Laboratory Press, New York, pp 343–406
Schopfer P, Plachy C, Frahry G (2001) Release of reactive oxygen intermediates (superoxide radicals, hydrogen peroxide, and hydroxyl radicals) and peroxidase in germinating radish seeds controlled by light, gibberellin, and abscisic acid. Plant Physiol 125:1591–1602
Siegel BZ (1993) Plant peroxidases—an organismic perspective. Plant Growth Regul 12:303–312
Tommasi F, Paciolla C, de Pinho MC, De Gara L (2006) Effects of storage temperature on viability, germination and antioxidant metabolism in Ginkgo biloba L. seeds. Plant Physiol Biochem 44:359–368
Varghese B, Sershen Berjak P, Varghese D, Pammenter NW (2011) Differential drying rates of recalcitrant Trichilia dregeana embryonic axes: a study of survival and oxidative stress metabolism. Physiol Plantarum 142:326–338
Vaz TAA, Davide AC, Rodrigues-Junior AG, Nakamura AT, Tonetti OAO, da Silva EAA (2016) Swartzia langsdorffii Raddi: morphophysiological traits of a recalcitrant seed dispersed during the dry season. Seed Sci Res 26:47–56
Veloso HP, Rangel Filho ALR, Lima JCA (1991) Classificação da vegetação brasileira adaptada a um sistema universal. FIBGE, Instituto Brasileiro de Geografia e Estatística, Rio de Janeiro
Acknowledgements
E.R. Marques wishes to thank CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for the scholarship received. T.A.A. Vaz wishes to thank CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and FAPEMIG (Fundação de Amparo à Pesquisa de Minas Gerais) for the scholarships received. A.G. Rodrigues-Junior wishes to thank CAPES for his scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by S.A. Merkle.
Rights and permissions
About this article
Cite this article
Marques, E.R., Vaz, T.A.A., Rodrigues-Junior, A.G. et al. In situ germination of two tropical recalcitrant seeds and changes in activity of ROS-scavenging enzymes. Trees 31, 1785–1792 (2017). https://doi.org/10.1007/s00468-017-1584-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-017-1584-9