Abstract
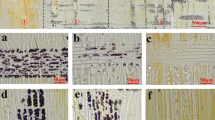
A living wood fiber (LWF) is one that retains the living protoplast. LWFs store numerous starch grains during the dormant period. In black locust (Robinia pseudoacacia), almost all wood fibers in the outer part of the annual ring are LWFs. In the outermost ring, starch is accumulated during the summer, retained in winter, and metabolized during spring. We determined the starch content of LWFs, ray parenchyma, and axial parenchyma using image analysis. More than 70% of the starch grains in the outermost ring were stored in LWFs during winter. After the breakdown of starch in spring, LWFs resulted in cell death. These results indicate that LWFs in black locust function as “single-use” large-capacity starch storage.







Similar content being viewed by others
References
Braun HJ (1963) Die Organisation des Stammes von Bäumen und Sträuchern. Wissenschaftliche Verlagsgesellschaft MBH, Stuttgart
Carlquist S (2001) Comparative wood anatomy, 2nd edn. Springer, Berlin
Derr WF, Evert RF (1967) The cambium and seasonal development of the phloem in Robinia pseudoacacia. Am J Bot 54:147–153
Dumbroff EB, Elmore HW (1977) Living fibres are a principal feature of the xylem in seedlings of Acer saccharum Marsh. Ann Bot 41:471–472
Essiamah S, Eschirich W (1985) Changes of starch content in the storage tissue of deciduous trees during winter and spring. IAWA Bull 6:97–106
Fahn A, Arnon N (1963) The living wood fibres of Tamarix aphylla and the changes occurring in them in transition from sapwood to heartwood. New Phytol 62:99–104
Fahn A, Leshem B (1963) Wood fibres with living protoplasts. New Phytol 62:91–98
Fischer C, Höll W (1992) Food reserves of scots pine (Pinus sylvestris L.). II. Seasonal changes and radial distribution of carbohydrate and fat reserves in pine wood. Trees 6:147–155
Fromard L, Babin V, Fleurat-Lessard P, Fromont JC, Serrano R, Bonnemain JL (1995) Control of vascular sap pH by the vessel-associated cells in woody species (physiological and immunological studies). Plant Physiol 108:913–918
Gregory RA (1978) Living elements of the conducting secondary xylem of sugar maple (Acer saccharum Marsh.). IAWA Bull 4:65–70
Hendrix DL (1993) Rapid extraction and analysis of nonstructural carbohydrates in plant tissues. Crop Sci 33:1306–1311
Höll W (2000) Distribution, fluctuation and metabolism of food reserves in the wood of trees. In: Savidge R, Barnett J, Napier R (eds) Cell and molecular biology of wood formation. BIOS Scientific Publishers Ltd., Oxford, pp 347–362
IAWA Committee on Nomenclature (1964) Multilingual glossary of terms used in wood anatomy. Konkordia, Winterthur, Switzerland
Krabel D (2000) Influence of sucrose on cambial activity. In: Savidge R, Barnett J, Napier R (eds) Cell and molecular biology of wood formation. BIOS Scientific Publishers Ltd., Oxford, pp 113–125
Lillie RD, Laskey A, Greco J, Jacquier H (1947) Reticulum staining with Schiff reagent after oxidation by acidified sodium periodate. J Lab Clin Med 32:910–912
Luft JH (1961) Improvements in epoxy resin embedding method. J Biophys Biochem Cytol 9:409–414
Magel E (2000) Biochemistry and physiology of heartwood formation. In: Savidge R, Barnett J, Napier R (eds) Cell and molecular biology of wood formation. BIOS Scientific Publishers Ltd., Oxford, pp 363–376
Magel EC, Jay-Allemand C, Ziegler H (1994) Formation of heartwood substances in the stem wood of Robinia pseudoacacia L. II. Distribution of nonstructural carbohydrates and wood extractives across the trunk. Trees 8:165–171
Sauter JJ (2000) Photosynthate allocation to the vascular cambium: facts and problems. In: Savidge R, Barnett J, Napier R (eds) Cell and molecular biology of wood formation. BIOS Scientific Publishers Ltd., Oxford, pp 71–83
Sauter JJ, van Cleve B (1994) Storage, mobilization and interrelations of starch, sugars, protein and fat in the ray storage tissue of poplar trees. Trees 8:297–304
Wargo PM (1979) Starch storage and radial growth in woody roots of sugar maple. Can J For Res 9:49–56
Witt W, Sauter JJ (1994) Starch metabolism in poplar wood ray cells during spring mobilization and summer deposition. Physiol Plant 92:9–16
Wolkinger F (1969) Morphology and systematic distribution of living wood fibres of shrubs and trees. I. Morphology and cytology. Holzforschung 23:135–144
Wolkinger F (1970a) Das Vorkommen lebender Holzfasern in Sträuchern un Bäumen. Phyton 14:55–67
Wolkinger F (1970b) Morphology and systematic distribution of living wood fibres of shrubs and trees. II. Histology. Holzforschung 24:141–147
Wong BL, Baggett KL, Rye AH (2003) Seasonal patterns of reserve and soluble carbohydrates in mature sugar maple (Acer saccharum). Can J Bot 81:780–788
Zhang C, Fujita M, Takabe K (2005) Comparison of contact and non-contact wood fibers in some hardwoods. IAWA J 26:457–467
Acknowledgments
The authors thank Dr. Koh Yasue (Shinshu University), Dr. Yoko Watanabe (Hokkaido University), and Dr. Kazuhiko Masaka (Hokkaido Forestry Research Institute) for their help in sample collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by U. Luettge.
Rights and permissions
About this article
Cite this article
Yamada, Y., Awano, T., Fujita, M. et al. Living wood fibers act as large-capacity “single-use” starch storage in black locust (Robinia pseudoacacia). Trees 25, 607–616 (2011). https://doi.org/10.1007/s00468-010-0537-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-010-0537-3