Abstract
Background
Post-transplant diabetes mellitus (PTDM) is a major complication of immunosuppressive therapy, with many risk factors reported in adults with renal transplantation. The objective of this study was to investigate potential non-genetic and genetic risk factors of PTDM in children with renal transplantation treated with tacrolimus.
Methods
A national database was screened for patients developing PTDM within 4 years following tacrolimus introduction. PTDM was defined as glucose disorder requiring anti-diabetic treatment. PTDM patients were matched to “non-PTDM” control transplanted children according to age, gender, and duration of post-transplant follow-up. Patients were genotyped for six selected genetic variants in POR*28 (rs1057868), PPARa (rs4253728), CYP3A5 (rs776746), VDR (rs2228570 and rs731236), and ABCB1 (rs1045642) genes, implicated in glucose homeostasis and tacrolimus disposition.
Results
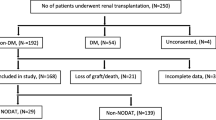
Among the 98 children with renal transplantation enrolled in this multicentre study, 18 developed PTDM. None of the clinical and biological parameters was significant between PTDM and control patients. Homozygous carriers of POR*28 or wild-type ABCB1 (rs1045642) gene variants were more frequent in PTDM than in control patients with differences close to significance (p = 0.114 and p = 0.066 respectively). A genetic score based on these variants demonstrated that POR*28/*28 and ABCB1 CC or CT genotype carriers were at a significantly higher risk of developing PTDM after renal transplantation.
Conclusion
Identification of PTDM risk factors should allow clinicians to allocate the best immunosuppressant for each patient with renal transplantation, and improve care for patients who are at a higher risk.



Similar content being viewed by others
References
Tonelli M, Wiebe N, Knoll G, Bello A, Browne S, Jadhav D, Klarenbach S, Gill J (2011) Systematic review: kidney transplantation compared with dialysis in clinically relevant outcomes. Am J Transplant 11:2093–2109
Assadi F (2013) Pediatric kidney transplantation: kids are different. Iran J Kidney Dis 7:429–431
Sharma A, Ramanathan R, Posner M, Fisher RA (2013) Pediatric kidney transplantation: a review. Transplantation 5:21–31
Jurewicz WA (2003) Tacrolimus versus cyclosporin immunosuppression: long-term outcome in renal transplantation. Nephrol Dial Transplant 18 [Suppl 1]:i7–11
Martins L, Ventura A, Branco A, Carvalho MJ, Henriques AC, Dias L, Sarmento AM, Amil M (2004) Cyclosporine versus tacrolimus in kidney transplantation: are there differences in nephrotoxicity? Transplant Proc 36:877–879
Kaufman DB, Shapiro R, Lucey MR, Cherikh WS, T Bustami R, Dyke DB (2004) Immunosuppression: practice and trends. Am J Transplant Suppl 9:38–53
Rodriguez-Rodriguez AE, Triñanes J, Velazquez-Garcia S, Porrini E, Vega Prieto MJ, Diez Fuentes ML, Arevalo M, Salido Ruiz E (2013) The higher diabetogenic risk of tacrolimus depends on pre-existing insulin resistance. A study in obese and lean Zucker rats. Am J Transplant 13:1665–1675
Vincenti F, Friman S, Scheuermann E, Rostaing L, Jenssen T, Campistol JM, Uchida K, Pescovitz MD, Marchetti P, Tuncer M, Citterio F, Wiecek A, Chadban S, El-Shahawi M, Budde K, Goto N (2007) Results of an international, randomized trial comparing glucose metabolism disorders and outcome with cyclosporine versus tacrolimus. Am J Transplant 7:1506–1514
Webster A, Woodroffe RC, Taylor RS, Chapman JR, Craig JC (2005) Tacrolimus versus cyclosporin as primary immunosuppression for kidney transplant recipients. Cochrane Database Syst Rev 19:CD003961
Kasiske BL, Snyder JJ, Gilbertson D, Matas AJ (2003) Diabetes mellitus after kidney transplantation in the United States. Am J Transplant 3:178–185
Cosio FG, Pesavento TE, Kim S, Osei K, Henry M, Ferguson RM (2002) Patient survival after renal transplantation. IV. Impact of post-transplant diabetes. Kidney Int 62:1440–1446
Cosio FG, Kudva Y, van der Velde M, Larson TS, Textor SC, Griffin MD, Stegal MD (2005) New onset hyperglycemia and diabetes are associated with increased cardiovascular risk after kidney transplantation. Kidney Int 67:2415–2421
Al-Uzri A, Stablein DM, Cohn RA (2001) Posttransplant diabetes mellitus in pediatric renal transplant recipients: a report of the north American Pediatric renal transplant cooperative study (NAPRTCS). Transplantation 72:1020–1024
Prokai A, Fekete A, Kis E, Reusz GS, Sallay P, Korner A, Wagner L, Tulassay T, Szabo AJ (2008) Post-transplant diabetes mellitus in children following renal transplantation. Pediatr Transplant 12:643–649
Burroughs TE, Swindle JP, Salvalaggio PR, Lentine KL, Takemoto SK, Bunnapradist S, Brennan DC, Schnitzler MA (2009) Increasing incidence of new-onset diabetes after transplant among pediatric renal transplant patients. Transplantation 88:367–373
Greenspan LC, Gitelman SE, Leung MA, Glidden DV, Mathias RS (2002) Increased incidence in post-transplant diabetes mellitus in children: a case-control analysis. Pediatr Nephrol 17:1–5
Sharif A, Baboolal K (2010) Risk factors for new-onset diabetes after kidney transplantation. Nat Rev Nephrol 6:415–423
Lancia P, Adam de Beaumais T, Jacqz-Aigrain E (2017) Pharmacogenetics of posttransplant diabetes mellitus. Pharm J 17:209–221
Sharif A, Hecking M, de Vries APJ, Porrini E, Hornum M, Rasoul-Rockenschaub S, Berlakovich G, Krebs M, Kautsky-Willer A, Schernthaner G, Marchetti P, Pacini G, Ojo A, Takahara S, Larsen JL, Budde K, Eller K, Pascual J, Jardine A, Bakker SJ, Valderhaug TG, Jenssen TG, Cohney S, Säemann MD (2014) Proceedings from an international consensus meeting on posttransplantation diabetes mellitus: recommendations and future directions. Am J Transplant 14:1992–2000
Cosio FG, Pesavento TE, Osei K, Henry ML, Ferguson RM (2001) Post-transplant diabetes mellitus: increasing incidence in renal allograft recipients transplanted in recent years. Kidney Int 59:732–737
Davidson J, Wilkinson A, Dantal J, Dotta F, Haller H, Hernandez D, Kasiske BL, Kiberd B, Krentz A, Legendre C, Marchetti P, Markell M, van der Woude FJ, Wheeler D (2003) Diabetes after transplantation: 2003 international consensus guidelines. Transplantation 75:SS3–SS24
Pham PT, Pham PM, Pham SV, Pham PA, Pham PC (2011) New onset diabetes after transplantation (NODAT): an overview. Diabetes Metab Syndr Obes 4:175–186
Garro R, Warshaw B, Felner E (2015) New-onset diabetes after kidney transplant in children. Pediatr Nephrol 30:405–416
Hamer RA, Chow CL, Ong ACM, McKane WS (2007) Polycystic kidney disease is a risk factor for new-onset diabetes after transplantation. Transplantation 83:36–40
Nesmith JD, Ellis E (2007) Childhood hemolytic uremic syndrome is associated with adolescent-onset diabetes mellitus. Pediatr Nephrol 22:294–297
Suri RS, Clark WF, Barrowman N, Mahon JL, Thiessen-Philbrook HR, Rosas-Arellano MP, Zarnke K, Garland JS, Garg AX (2005) Diabetes during diarrhea-associated hemolytic uremic syndrome: a systematic review and meta-analysis. Diabetes Care 28:2556–2562
Tobin JL, Beales PL (2007) Bardet-Biedl syndrome: beyond the cilium. Pediatr Nephrol 22:926–936
Sam WJ, Aw M, Quak SH, Lim SM, Charles BG, Chan SY, Ho PC (2000) Population pharmacokinetics of tacrolimus in Asian paediatric liver transplant patients. Br J Clin Pharmacol 50:531–541
Shishido S, Asanuma H, Tajima E, Honda M, Nakai H (2001) Pharmacokinetics of tacrolimus in pediatric renal transplant recipients. Transplant Proc 33:1066–1068
Luan FL, Steffick DE, Ojo AO (2011) New-onset diabetes mellitus in kidney transplant recipients discharged on steroid-free immunosuppression. Transplantation 91:334–341
Boots JMM, Christiaans MHL, Van Duijnhoven EM, Van Suylen R-J, Van Hooff JP (2002) Early steroid withdrawal in renal transplantation with tacrolimus dual therapy: a pilot study. Transplantation 74:1703–1709
Grenda R (2010) Effects of steroid avoidance and novel protocols on growth in paediatric renal transplant patients. Pediatr Nephrol 25:747–752
Sarwal MM, Vidhun JR, Alexander SR, Satterwhite T, Millan M, Salvatierra O (2003) Continued superior outcomes with modification and lengthened follow-up of a steroid-avoidance pilot with extended daclizumab induction in pediatric renal transplantation. Transplantation 76:1331–1339
Boots JMM, van Duijnhoven EM, Christiaans MHL, Wolffenbuttel BHR, van Hooff JP (2002) Glucose metabolism in renal transplant recipients on tacrolimus: the effect of steroid withdrawal and tacrolimus trough level reduction. J Am Soc Nephrol 13:221–227
Filler G, Neuschulz I, Vollmer I, Amendt P, Hocher B (2000) Tacrolimus reversibly reduces insulin secretion in paediatric renal transplant recipients. Nephrol Dial Transplant 15:867–871
Rathi M, Rajkumar V, Rao N, Sharma A, Kumar S, Ramachandran R, Kumar V, Kohli HS, Gupta KL, Sakhuja V (2015) Conversion from tacrolimus to cyclosporine in patients with new-onset diabetes after renal transplant: an open-label randomized prospective pilot study. Transplant Proc 47:1158–1161
Lorho R, Hardwigsen J, Dumortier J, Pageaux GP, Durand F, Bizollon T, Blanc AS, Di Giambattista F, Duvoux C (2011) Regression of new-onset diabetes mellitus after conversion from tacrolimus to cyclosporine in liver transplant patients: results of a pilot study. Clin Res Hepatol Gastroenterol 35:482–488
Van Duijnhoven EM, Christiaans MH, Boots JM, Nieman FH, Wolffenbutel BH, van Hooff JP (2002) Glucose metabolism in the first 3 years after renal transplantation in patients receiving tacrolimus versus cyclosporine-based immunosuppression. J Am Soc Nephrol 13:213–220
Tarnowski M, Słuczanowska-Głabowska S, Pawlik A, Mazurek-Mochol M, Dembowska E (2017) Genetic factors in pathogenesis of diabetes mellitus after kidney transplantation. Ther Clin Risk Manag 13:439–446
De Jonge H, Metalidis C, Naesens M, Lambrechts D, Kuypers DRJ (2011) The P450 oxidoreductase *28 SNP is associated with low initial tacrolimus exposure and increased dose requirements in CYP3A5-expressing renal recipients. Pharmacogenomics 12:1281–1291
Lunde I, Bremer S, Midtvedt K, Mohebi B, Dahl M, Bergan S, Asberg A, Christensen H (2014) The influence of CYP3A, PPARA, and POR genetic variants on the pharmacokinetics of tacrolimus and cyclosporine in renal transplant recipients. Eur J Clin Pharmacol 70:685–693
Klein K, Thomas M, Winter S, Nussler AK, Niemi M, Schwab M, Zanger UM (2012) PPARA: a novel genetic determinant of CYP3A4 in vitro and in vivo. Clin Pharmacol Ther 91:1044–1052
Chiu KC, Chu A, Go VLW, Saad MF (2004) Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am J Clin Nutr 79:820–825
Wang Z, Schuetz EG, Xu Y, Thummel KE (2013) Interplay between vitamin D and the drug metabolizing enzyme CYP3A4. J Steroid Biochem Mol Biol 136:54–58
Lindh JD, Björkhem-Bergman L, Eliasson E (2012) Vitamin D and drug-metabolising enzymes. Photochem Photobiol Sci 11:1797–1801
Hubbard PA, Shen AL, Paschke R, Kasper CB, Kim JJ (2001) NADPH-cytochrome P450 oxidoreductase. Structural basis for hydride and electron transfer. J Biol Chem 276:29163–29170
Miller WL, Agrawal V, Sandee D, Tee MK, Huang N, Choi JH, Morissey K, Giacomini KM (2011) Consequences of POR mutations and polymorphisms. Mol Cell Endocrinol 336:174–179
Oneda B, Crettol S, Jaquenoud Sirot E, Bochud M, Ansermot N, Eap CB (2009) The P450 oxidoreductase genotype is associated with CYP3A activity in vivo as measured by the midazolam phenotyping test. Pharmacogenet Genomics 19:877–883
Huang N, Agrawal V, Giacomini KM, Miller WL (2008) Genetics of P450 oxidoreductase: sequence variation in 842 individuals of four ethnicities and activities of 15 missense mutations. Proc Natl Acad Sci 105:1733–1738
Marzolini C, Paus E, Buclin T, Kim RB (2004) Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther 75:13–33
Hjelmesaeth J, Hartmann A, Kofstad J, Egeland T, Stenstrøm J, Fauchald P (2001) Tapering off prednisolone and cyclosporin the first year after renal transplantation: the effect on glucose tolerance. Nephrol Dial Transplant 16:829–835
Huang JW, Famure O, Li Y, Kim SJ (2016) Hypomagnesemia and the risk of new-onset diabetes mellitus after kidney transplantation. J Am Soc Nephrol 27:1793–1800
Hayes W, Boyle S, Carroll A, Bockenhauer D, Marks SD (2017) Hypomagnesemia and increased risk of new-onset diabetes mellitus after transplantation in pediatric renal transplant recipients. Pediatr Nephrol 32:879–884
Takaya J, Higashino H, Kobayashi Y (2004) Intracellular magnesium and insulin resistance. Magnes Res 17:126–136
Acknowledgements
The authors thank Dr MA Macher, Biomedicine Agency and Department of Paediatric Nephrology, Robert Debré Hospital, for her help in the study organisation, Yves Medard, Department of Paediatric Pharmacology and Pharmacogenetics, Robert Debré Hospital, for technical expertise in molecular biology and Professor M Tsimaratos, Department of Paediatric Nephrology, La Timone, Marseille, for his collaboration.
Funding
Funding was partially provided by the European Union Seventh Framework Programme FP7/2007–2013-Global Research in Paediatrics Network (grant no. 261060).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures were carried out in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The project fulfils the requirement of the “Commission Nationale Informatique et Liberté” (2014), and approval was obtained from the local Ethics Committee CEERB (2016/287). Informed consent was signed by the parents.
Rights and permissions
About this article
Cite this article
Lancia, P., Adam de Beaumais, T., Elie, V. et al. Pharmacogenetics of post-transplant diabetes mellitus in children with renal transplantation treated with tacrolimus. Pediatr Nephrol 33, 1045–1055 (2018). https://doi.org/10.1007/s00467-017-3881-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-017-3881-3